Oncology Biostatistics Design strategies to assess benefit for

Oncology Biostatistics Design strategies to assess benefit for biomarker sub-populations in Phase III clinical trials Bharani Dharan, Ekkehard Glimm Joint Statistical Meeting, Denver July 31, 2019

Outline 2 • Background • Factors impacting biomarker study designs • Types of biomarker designs • Simulation Results • Summary Oncology Analytics - Biostatistics

Background: Biomarker based Phase III designs • A confirmatory trial with a biomarker subpopulation • Multiple populations of interest : All patients, Biomarker positive (B+), Biomarker negative (B-) • Multiple statistical comparisons involved due to multiple populations – Overall type 1 error rate needs to be controlled at one-sided 2. 5% level • Role of B+ subgroup is key to assess any differential effect between the biomarker subgroups Freidlin B, Sun Z, Gray R, et al: Phase III clinical trials that integrate treatment and biomarker evaluation. JCO 31: 3158 -3161, 2013 Goteti S, Hirawat S, Massacesi C, Dharan B. Some practical considerations for phase III studies with biomarker evaluations. JCO 32: 854– 855, 2014 3 Oncology Analytics - Biostatistics

Factors that can impact the study design Biomarker prevalence, targeted effect, biomarker assay • Prevalence of the biomarker subgroup – If B+ comprises a higher percentage of patient population, it is expected that treatment effect in “all patients” will be influenced by treatment effect in B+ • Targeted treatment effect in biomarker subgroups and all patients – A conservative targeted effect can result in over-powering of the endpoints. • Assay to determine the biomarker status – Treatment effect needs to be demonstrated by CDx assay in addition to clinical trial assay – Assay sensitivity and specificity is crucial – Minimize unknown status or randomize only the known biomarker status patients 4 Oncology Analytics - Biostatistics
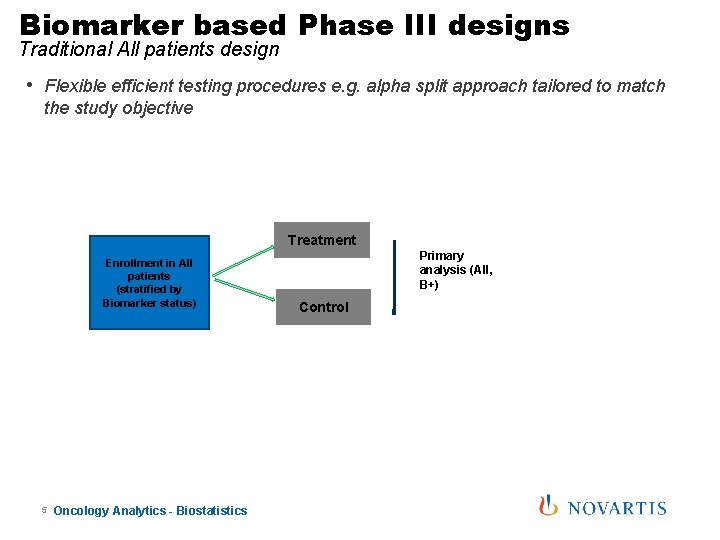
Biomarker based Phase III designs Traditional All patients design • Flexible efficient testing procedures e. g. alpha split approach tailored to match the study objective Treatment Enrollment in All patients (stratified by Biomarker status) 5 Oncology Analytics - Biostatistics Primary analysis (All, B+) Control
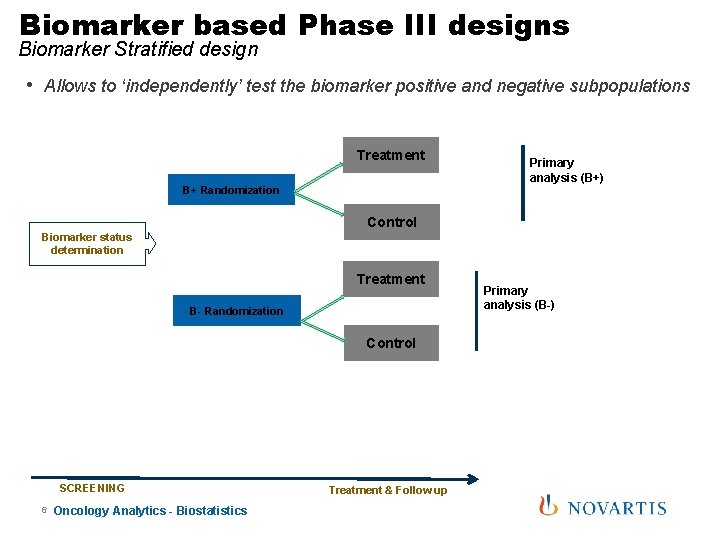
Biomarker based Phase III designs Biomarker Stratified design • Allows to ‘independently’ test the biomarker positive and negative subpopulations Treatment B+ Randomization Primary analysis (B+) Control Biomarker status determination Treatment B- Randomization Control SCREENING 6 Oncology Analytics - Biostatistics Treatment & Follow up Primary analysis (B-)
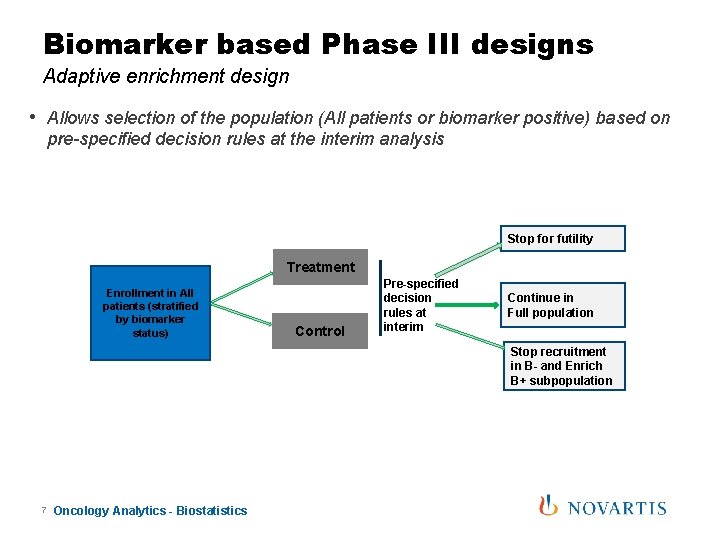
Biomarker based Phase III designs Adaptive enrichment design • Allows selection of the population (All patients or biomarker positive) based on pre-specified decision rules at the interim analysis Stop for futility Treatment Enrollment in All patients (stratified by biomarker status) Control Pre-specified decision rules at interim Continue in Full population Stop recruitment in B- and Enrich B+ subpopulation 7 Oncology Analytics - Biostatistics

Traditional All patient design: Simulation set-up • Study enrolled ‘all patients’ which include biomarker positive and negative sub-populations – Primary endpoints: Progression-free survival (PFS) in All patients and biomarker positive – Key secondary endpoints: Overall survival (OS) in All patients and biomarker positive • Gatekeeping approach to test the multiple endpoints • Assumptions: – Target hazard ratio for PFS: 0. 67 in all patients and 0. 60 in B+ sub-population – Target hazard ratio for OS: 0. 75 in all patients and 0. 625 in B+ sub-population – Accrual rate: 30 patients/month with ~40% B+ 8 Oncology Analytics - Biostatistics

Traditional design: Statistical testing approach Alpha split approach tailored to the hypotheses of interest • Several testing strategies are explored. – Scenario 1: More alpha for overall and less alpha for biomarker +: 2%, 0. 5% – Scenario 2: Equal alpha split between overall and biomarker +: 1. 25%, 1. 25% – Scenario 3: Less alpha for overall and more alpha for biomarker +: 0. 5%, 2% Scenario 1: 9 Oncology Analytics - Biostatistics
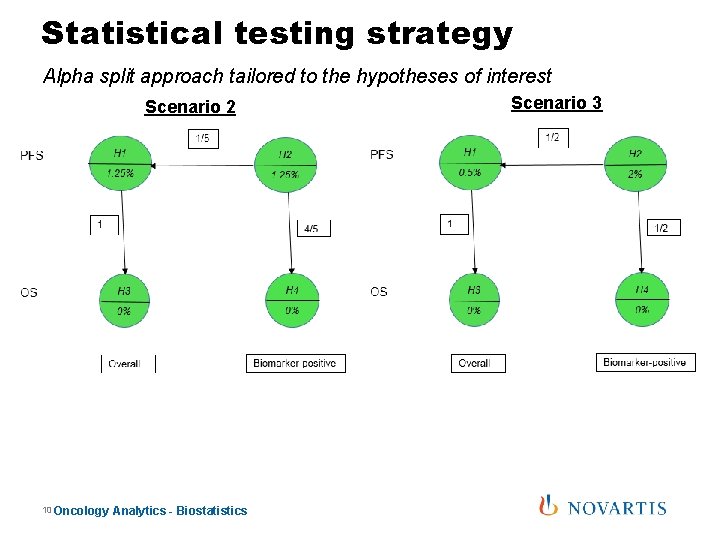
Statistical testing strategy Alpha split approach tailored to the hypotheses of interest Scenario 2 10 Oncology Analytics - Biostatistics Scenario 3
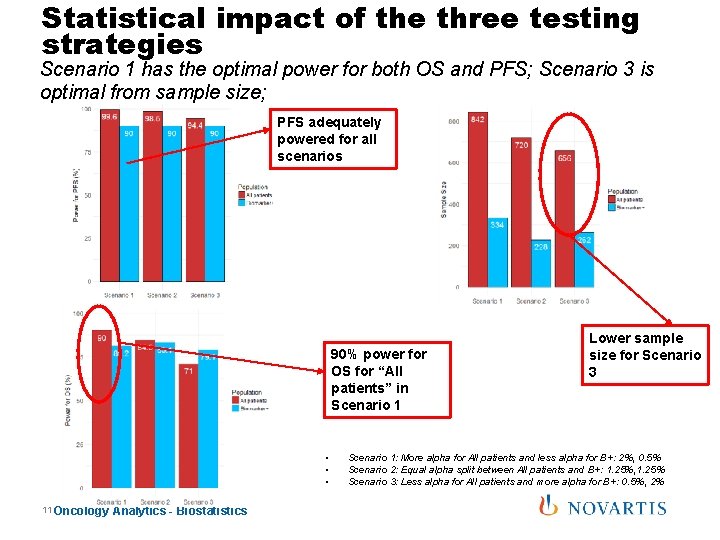
Statistical impact of the three testing strategies Scenario 1 has the optimal power for both OS and PFS; Scenario 3 is optimal from sample size; PFS adequately powered for all scenarios 90% power for OS for “All patients” in Scenario 1 • • • 11 Oncology Analytics - Biostatistics Lower sample size for Scenario 3 Scenario 1: More alpha for All patients and less alpha for B+: 2%, 0. 5% Scenario 2: Equal alpha split between All patients and B+: 1. 25%, 1. 25% Scenario 3: Less alpha for All patients and more alpha for B+: 0. 5%, 2%

Testing strategy for biomarker negative Decision rule for B- • Biomarker negative sub-population will be assessed to determine whether the treatment effect in All patients is driven solely by biomarker positive subpopulation or by both positive and negative subpopulation • Should there an alpha allocated to test the biomarker negative? or • Will a pre-defined clinically relevant threshold based on a decision rule suffice? – E. g. Hazard ratio <0. 67 and probability(HR<1)>97. 5% 12 Oncology Analytics - Biostatistics
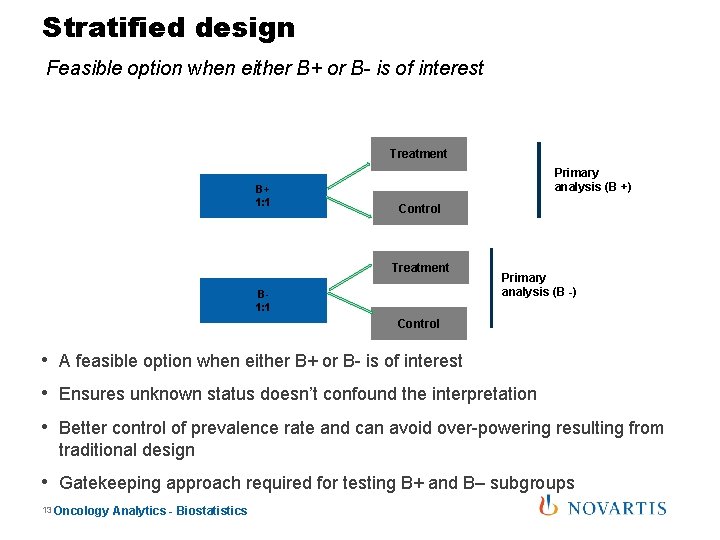
Stratified design Feasible option when either B+ or B- is of interest Treatment B+ 1: 1 Primary analysis (B +) Control Treatment B 1: 1 Primary analysis (B -) Control • A feasible option when either B+ or B- is of interest • Ensures unknown status doesn’t confound the interpretation • Better control of prevalence rate and can avoid over-powering resulting from traditional design • Gatekeeping approach required for testing B+ and B– subgroups 13 Oncology Analytics - Biostatistics
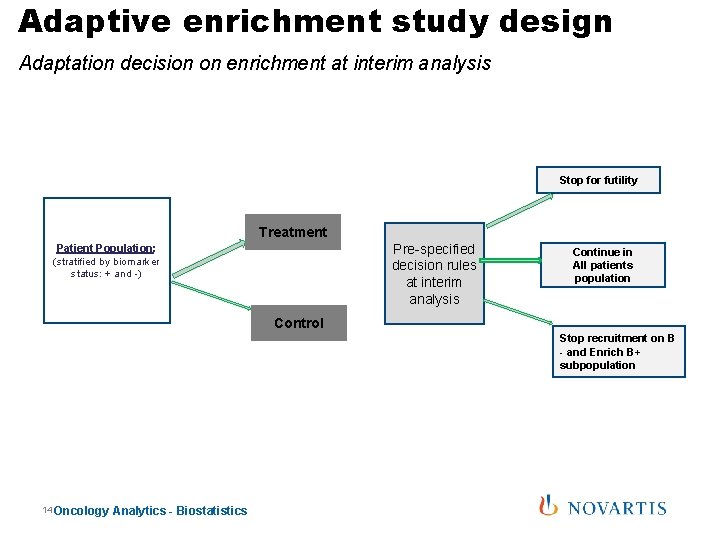
Adaptive enrichment study design Adaptation decision on enrichment at interim analysis Stop for futility Treatment Pre-specified decision rules at interim analysis Patient Population: (stratified by biomarker status: + and -) Continue in All patients population Control Stop recruitment on B - and Enrich B+ subpopulation 14 Oncology Analytics - Biostatistics
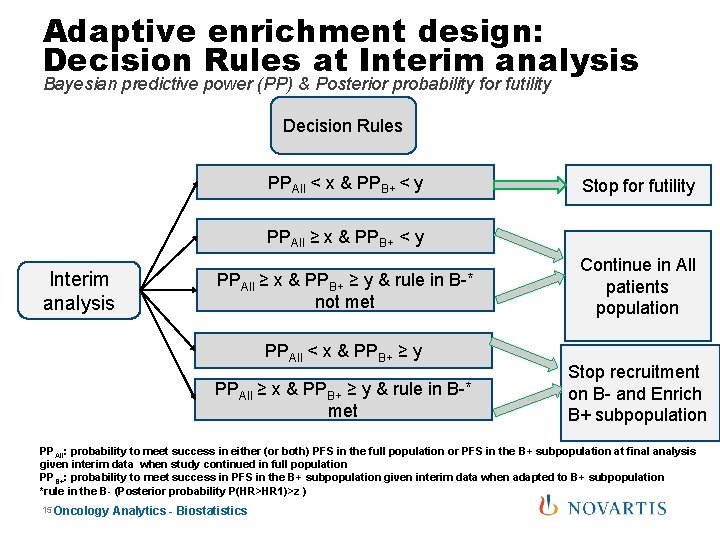
Adaptive enrichment design: Decision Rules at Interim analysis Bayesian predictive power (PP) & Posterior probability for futility Decision Rules PPAll < x & PPB+ < y Stop for futility PPAll ≥ x & PPB+ < y Interim analysis PPAll ≥ x & PPB+ ≥ y & rule in B-* not met PPAll < x & PPB+ ≥ y PPAll ≥ x & PPB+ ≥ y & rule in B-* met Continue in All patients population Stop recruitment on B- and Enrich B+ subpopulation PPAll: probability to meet success in either (or both) PFS in the full population or PFS in the B+ subpopulation at final analysis given interim data when study continued in full population PPB+: probability to meet success in PFS in the B+ subpopulation given interim data when adapted to B+ subpopulation *rule in the B- (Posterior probability P(HR>HR 1)>z ) 15 Oncology Analytics - Biostatistics

Adaptive enrichment design: Methodology • Multiplicity issues – Testing in All patients & B+ populations – Two looks; Interim -Adaptation, Futility; Final - Efficacy analysis • Closed testing procedure – Hochberg p-value qi for intersection hypothesis H 0 F ∩ H 0 S at stage i qi=min{2 min(pi. S, pi. F), max(pi. S, pi. F)} • At final analysis – If trial continues in full population – Tests in F and S of equal interest (H 0 F & H 0 S co-primary) – Elementary hypotheses at stage 2: – Reject H 0 F ∩ H 0 S if C(q 1, q 2)≥c 2 then: – reject H 0 F if C(p 1 F, p 2 F) ≥c 2 & reject H 0 S if C(p 1 S, p 2 S) ≥c 2 – If trials is adapted to the B+ subpopulation – Reject H 0 F ∩ H 0 S if C(q 1, p 2 S)≥c 2 then – Reject H 0 S if C(p 1 S, p 2 S) ≥c 2 *F refers to “All patients” & S refers to biomarker positive subpopulation 16 Oncology Analytics - Biostatistics

Adaptive enrichment design: Methodology* • 17 Oncology Analytics - Biostatistics

Adaptive enrichment design Simulation set-up • Prevalence of biomarker positive – 30% – 40% – 50% • Posterior probability in the biomarker negative subpopulation: – Probability cutoff: 0. 75 – Hazard Ratio cutoff: 0. 80 • Predictive power cutoff (Full population, biomarker positive subpopulation): (0. 35, 0. 35) • Hazard ratio scenarios for biomarker positive and negative • Sample size of 524 patients; Interim analysis at 125 PFS events; Final analysis at 374 events in the All patients population and 281 events if enriched to the B+ subpopulation 18 Oncology Analytics - Biostatistics
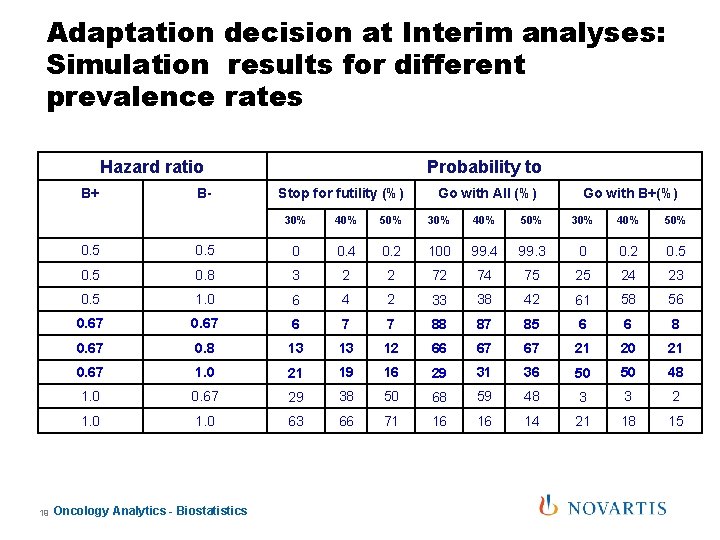
Adaptation decision at Interim analyses: Simulation results for different prevalence rates Hazard ratio B+ 19 B- Probability to Stop for futility (%) Go with All (%) Go with B+(%) 30% 40% 50% 0. 5 0 0. 4 0. 2 100 99. 4 99. 3 0 0. 2 0. 5 0. 8 3 2 2 72 74 75 25 24 23 0. 5 1. 0 6 4 2 33 38 42 61 58 56 0. 67 6 7 7 88 87 85 6 6 8 0. 67 0. 8 13 13 12 66 67 67 21 20 21 0. 67 1. 0 21 19 16 29 31 36 50 50 48 1. 0 0. 67 29 38 50 68 59 48 3 3 2 1. 0 63 66 71 16 16 14 21 18 15 Oncology Analytics - Biostatistics
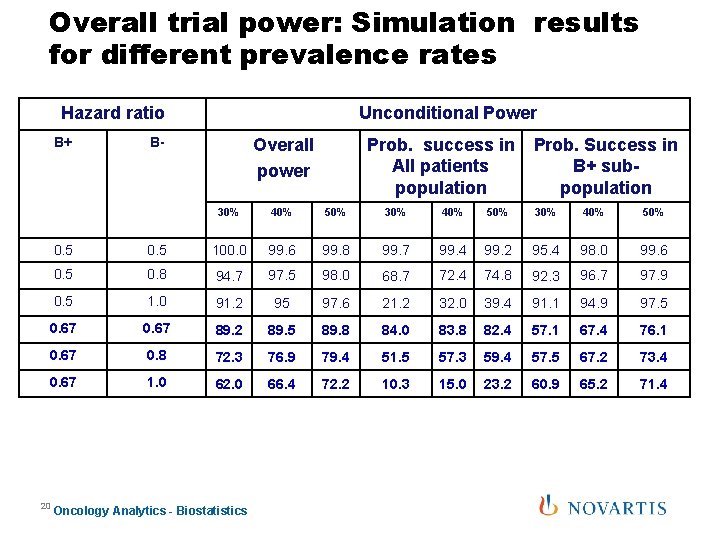
Overall trial power: Simulation results for different prevalence rates Hazard ratio B+ Unconditional Power B- Overall power Prob. success in All patients population Prob. Success in B+ subpopulation 30% 40% 50% 0. 5 100. 0 99. 6 99. 8 99. 7 99. 4 99. 2 95. 4 98. 0 99. 6 0. 5 0. 8 94. 7 97. 5 98. 0 68. 7 72. 4 74. 8 92. 3 96. 7 97. 9 0. 5 1. 0 91. 2 95 97. 6 21. 2 32. 0 39. 4 91. 1 94. 9 97. 5 0. 67 89. 2 89. 5 89. 8 84. 0 83. 8 82. 4 57. 1 67. 4 76. 1 0. 67 0. 8 72. 3 76. 9 79. 4 51. 5 57. 3 59. 4 57. 5 67. 2 73. 4 0. 67 1. 0 62. 0 66. 4 72. 2 10. 3 15. 0 23. 2 60. 9 65. 2 71. 4 20 Oncology Analytics - Biostatistics
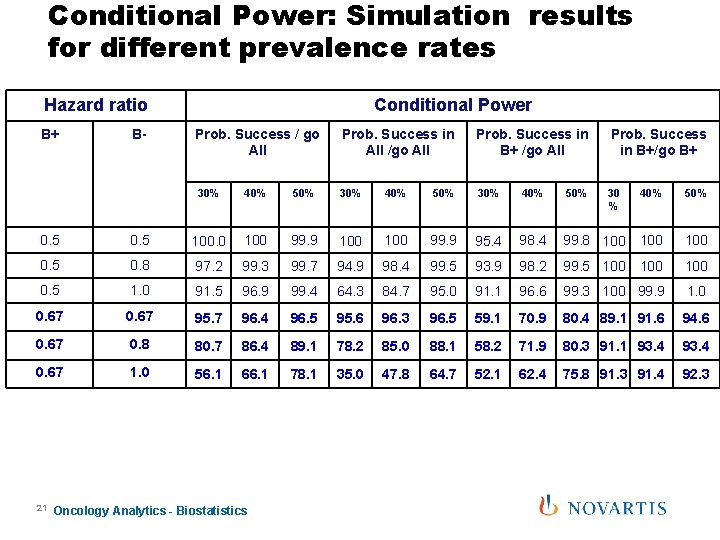
Conditional Power: Simulation results for different prevalence rates Hazard ratio B+ B- Conditional Power Prob. Success / go All Prob. Success in All /go All Prob. Success in B+/go B+ 30% 40% 50% 30 % 40% 50% 0. 5 100. 0 100 99. 9 95. 4 98. 4 99. 8 100 100 0. 5 0. 8 97. 2 99. 3 99. 7 94. 9 98. 4 99. 5 93. 9 98. 2 99. 5 100 100 0. 5 1. 0 91. 5 96. 9 99. 4 64. 3 84. 7 95. 0 91. 1 96. 6 99. 3 100 99. 9 1. 0 0. 67 95. 7 96. 4 96. 5 95. 6 96. 3 96. 5 59. 1 70. 9 80. 4 89. 1 91. 6 94. 6 0. 67 0. 8 80. 7 86. 4 89. 1 78. 2 85. 0 88. 1 58. 2 71. 9 80. 3 91. 1 93. 4 0. 67 1. 0 56. 1 66. 1 78. 1 35. 0 47. 8 64. 7 52. 1 62. 4 75. 8 91. 3 91. 4 92. 3 21 Oncology Analytics - Biostatistics
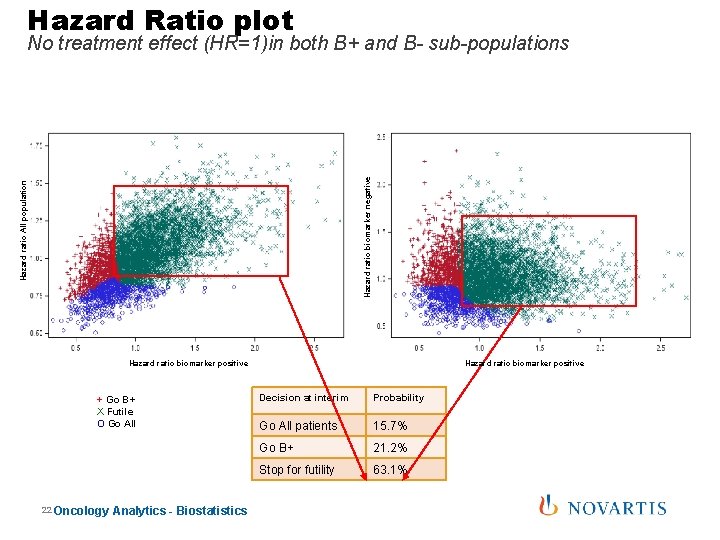
Hazard Ratio plot Hazard ratio All population Hazard ratio biomarker negative No treatment effect (HR=1)in both B+ and B- sub-populations Hazard ratio biomarker positive + Go B+ X Futile O Go All 22 Oncology Analytics - Biostatistics Decision at interim Probability Go All patients 15. 7% Go B+ 21. 2% Stop for futility 63. 1%
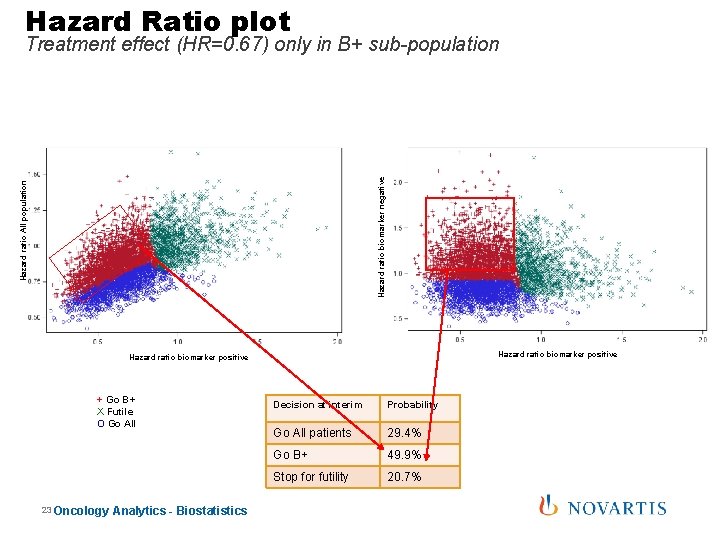
Hazard Ratio plot Hazard ratio All population Hazard ratio biomarker negative Treatment effect (HR=0. 67) only in B+ sub-population Hazard ratio biomarker positive + Go B+ X Futile O Go All 23 Oncology Analytics - Biostatistics Decision at interim Probability Go All patients 29. 4% Go B+ 49. 9% Stop for futility 20. 7%
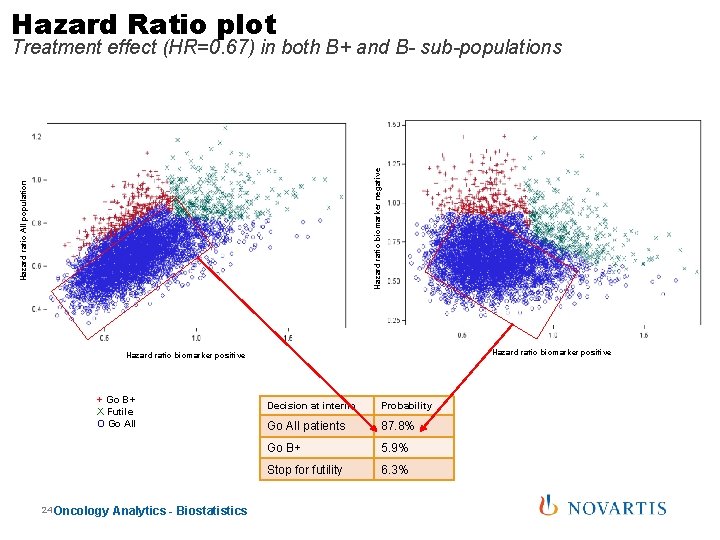
Hazard Ratio plot Hazard ratio All population Hazard ratio biomarker negative Treatment effect (HR=0. 67) in both B+ and B- sub-populations Hazard ratio biomarker positive + Go B+ X Futile O Go All 24 Oncology Analytics - Biostatistics Decision at interim Probability Go All patients 87. 8% Go B+ 5. 9% Stop for futility 6. 3%

Adaptive enrichment design: Operational elements Biomarker prevalence, DMC and enrollment hold • Biomarker prevalence can impact the decisions • Data Monitoring Committee (DMC) should be involved in the decision making for adaptation • Following DMC decision, randomization scheme will need to take into account the impact on randomization and stratification • Investigators should be informed of the possible actions and events in the course of the study which could result from the decision making in this trial • Follow-up of ongoing biomarker negative patients in case the study adapts to biomarker positive sub-population at the time of interim analysis • Enrollment hold during DMC review for adaptation 25 Oncology Analytics - Biostatistics

Summary • Prevalence of biomarker groups plays a crucial role in the study design and statistical testing sequence – Should be continuously monitored during the study • Inclusion/exclusion of ‘unknowns’ should be carefully considered at the study design stage • Designs such as gate-keeping procedures, adaptive designs or stratified designs can be used depending on the strategy 26 Oncology Analytics - Biostatistics

Acknowledgments • Frank Bretz • Nathalie Fretault • Sasikiran Goteti • Samit Hirawat • Cristian Massacesi • Willi Maurer • Sylvie Le Mouhaer • Kannan Natarajan • Yuanbo Song 27 Oncology Analytics - Biostatistics • Patrick Urban • Emmanuel Zuber

References • Bretz F, Maurer W, Brannath W, et al: A graphical approach to sequentially rejective multiple test procedures. Stat Med 28: 586 -604, 2009 • Brannath W, Zuber E et al: Confirmatory adaptive designs with Bayesian decision tools for a targeted therapy in oncology, Stats. Med; 28: 1445 -1463, 2009 • Freidlin B, Sun Z, Gray R, et al: Phase III clinical trials that integrate treatment and biomarker evaluation. JCO 31: 3158 -3161, 2013 • Goteti S, Hirawat S, Massacesi C, Fretault N, Bretz F and Dharan B: Some practical considerations for phase III studies with biomarker evaluations. JCO 32: 854– 855, 2014 • Rothmann et al, Testing in a Pre-specified Subgroup and the Intent-to-Treat Population: DIA, 175 -179, 46(2), 2012 28 Oncology Analytics - Biostatistics
- Slides: 28