ON THE ADVANTAGES OF MATRIXISOLATION VIBRATIONAL CIRCULAR DICHROISM

ON THE ADVANTAGES OF MATRIX-ISOLATION VIBRATIONAL CIRCULAR DICHROISM SPECTROSCOPY György Tarczay, Gábor Magyarfalvi Laboratory of Molecular Spectroscopy Elemér Vass Laboratory for Chiroptical Structure Analysis Institute of Chemistry, Eötvös University, Budapest, Hungary 61 st International Symposium on Molecular Spectroscopy, Columbus, 2006
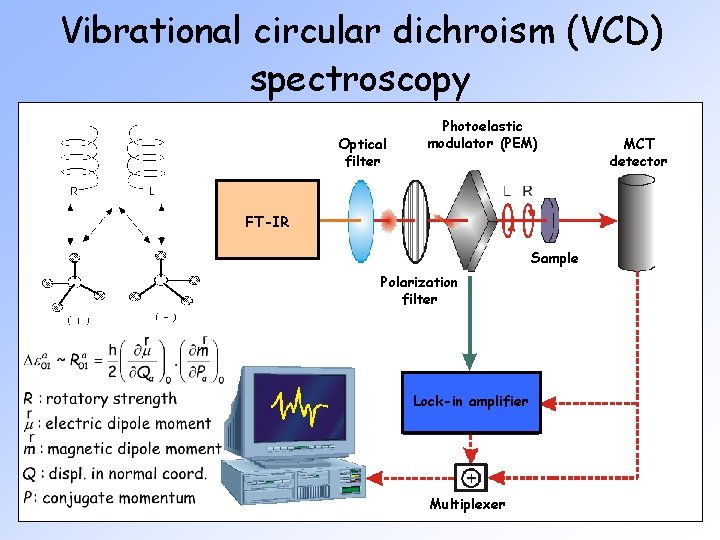
Vibrational circular dichroism (VCD) spectroscopy Optical filter Photoelastic modulator (PEM) FT-IR Sample Polarization filter Lock-in amplifier Multiplexer MCT detector

Advantages of VCD spectroscopy • Unlike in electronic CD spectroscopy (ECD) the investigated chiral molecule does not need to have any chromophoric groups • VCD spectra have richer structural content than ECD spectra the absolute configuration determination can be based on more assigned bands • ab initio calculation of VCD spectra can be carried out much more accurately the probability of an incorrect assignment is much smaller than in the case of ECD spectroscopy • Commercially available spectrometers from the late 90 s (Bio. Tools, Bruker, Jasco)
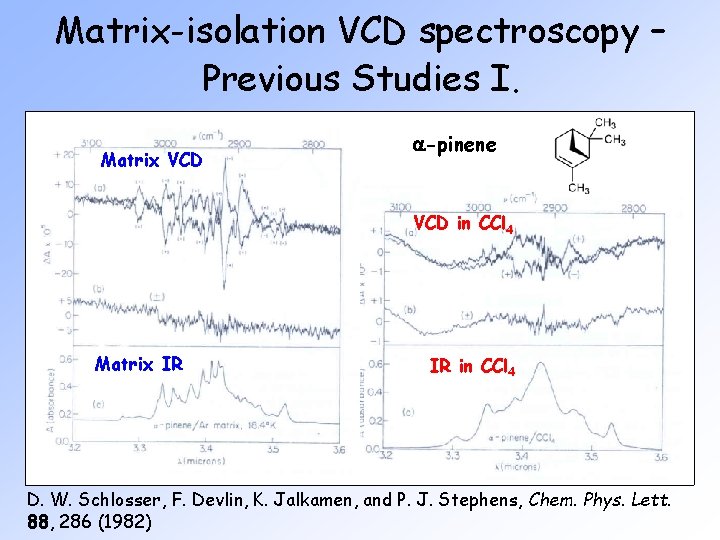
Matrix-isolation VCD spectroscopy – Previous Studies I. Matrix VCD a-pinene VCD in CCl 4 Matrix IR IR in CCl 4 D. W. Schlosser, F. Devlin, K. Jalkamen, and P. J. Stephens, Chem. Phys. Lett. 88, 286 (1982)
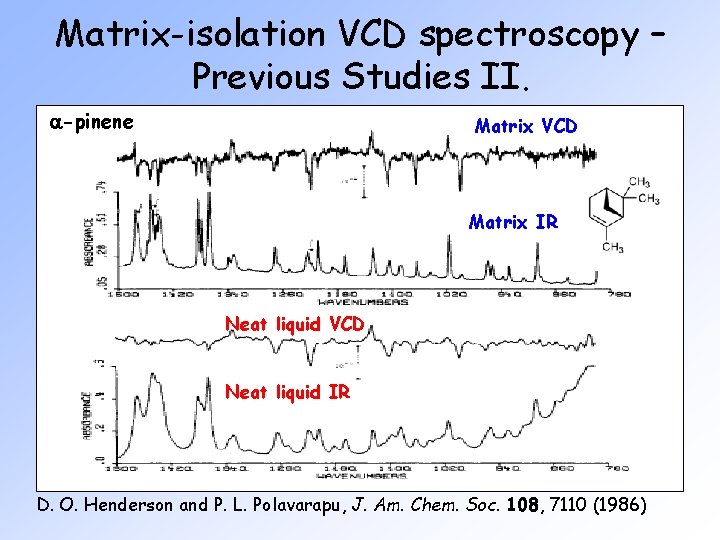
Matrix-isolation VCD spectroscopy – Previous Studies II. a-pinene Matrix VCD Matrix IR Neat liquid VCD Neat liquid IR D. O. Henderson and P. L. Polavarapu, J. Am. Chem. Soc. 108, 7110 (1986)

Conclusions of previous matrix-isolation VCD studies • Well resolved features • Intensity gain compared to spectra of neat liquids or solutions • Better correspondence between experiment and computations BUT • No work has been published after these two studies • It seems that the more labor-intensive matrix VCD measurements are not worth the candle in the case of single-conformer and/or weakly interacting molecules WHAT IS THE CASE WITH MORE COMPLEX SYSTEMS? • molecules with multiple conformers and/or H-bonding capability • chiral٠ chiral and chiral٠ achiral molecular complexes • chiral radicals
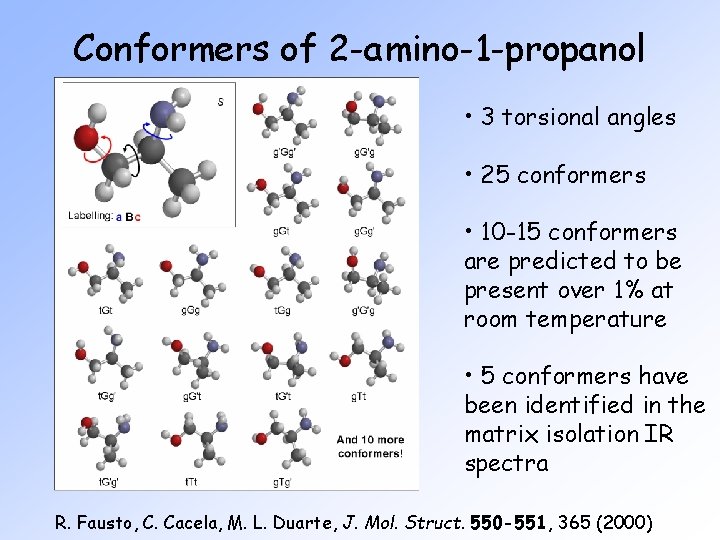
Conformers of 2 -amino-1 -propanol • 3 torsional angles • 25 conformers • 10 -15 conformers are predicted to be present over 1% at room temperature • 5 conformers have been identified in the matrix isolation IR spectra R. Fausto, C. Cacela, M. L. Duarte, J. Mol. Struct. 550 -551, 365 (2000)
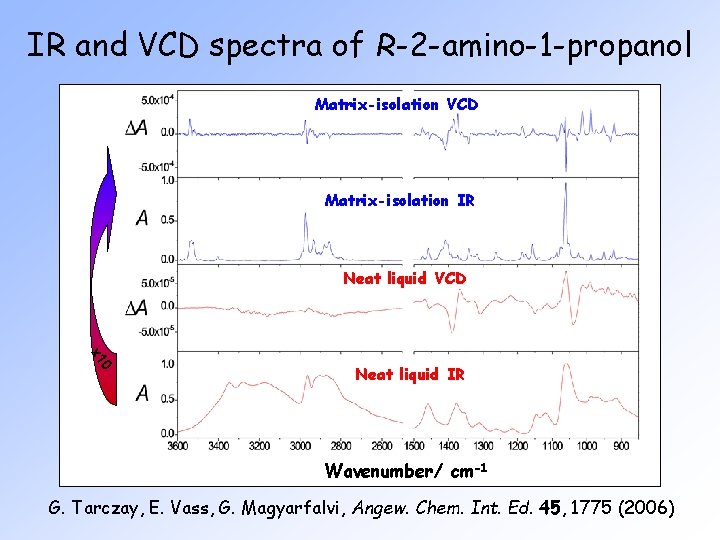
IR and VCD spectra of R-2 -amino-1 -propanol Matrix-isolation VCD Matrix-isolation IR Neat liquid VCD 0 1 Neat liquid IR Wavenumber/ cm-1 G. Tarczay, E. Vass, G. Magyarfalvi, Angew. Chem. Int. Ed. 45, 1775 (2006)
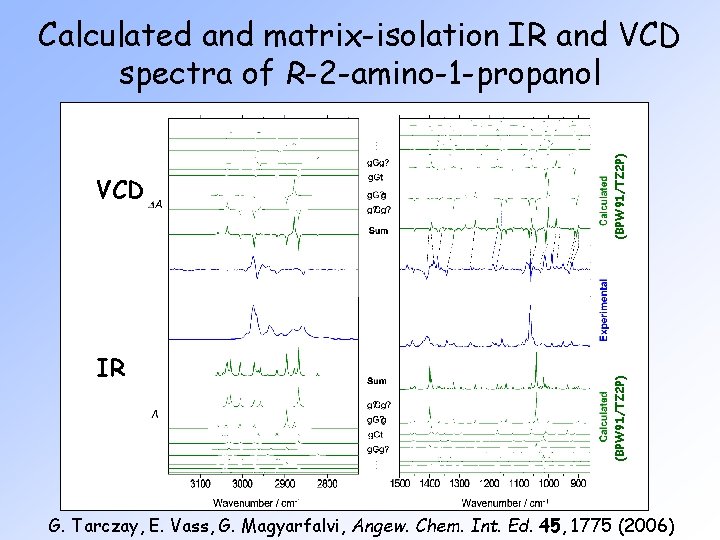
IR (BPW 91/TZ 2 P) VCD (BPW 91/TZ 2 P) Calculated and matrix-isolation IR and VCD spectra of R-2 -amino-1 -propanol G. Tarczay, E. Vass, G. Magyarfalvi, Angew. Chem. Int. Ed. 45, 1775 (2006)
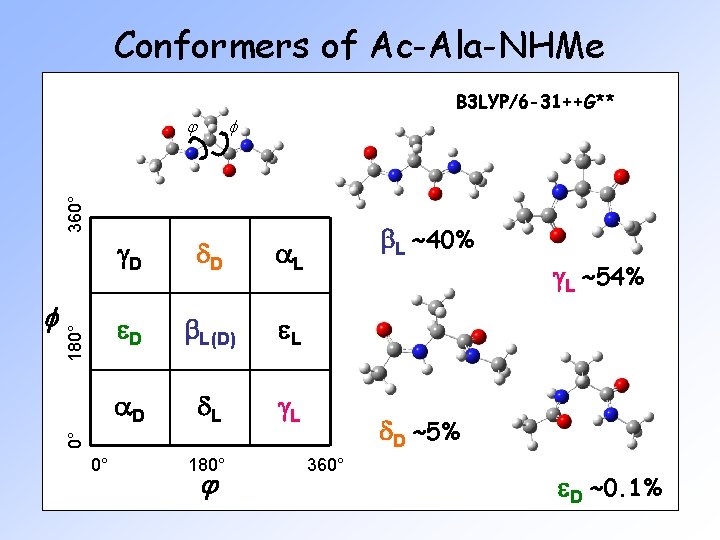
Conformers of Ac-Ala-NHMe f g. D d. D a. L e. D b. L(D) e. L a. D d. L g. L b. L ~40% 0° 180° j g. L ~54% d. D ~5% 0° f 180° 360° j B 3 LYP/6 -31++G** 360° e. D ~0. 1%
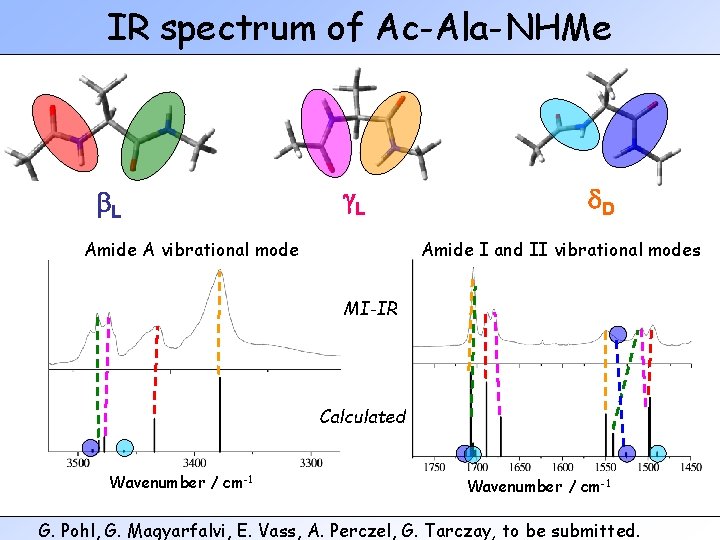
IR spectrum of Ac-Ala-NHMe b. L g. L Amide A vibrational mode d. D Amide I and II vibrational modes MI-IR Calculated Wavenumber / cm-1 G. Pohl, G. Magyarfalvi, E. Vass, A. Perczel, G. Tarczay, to be submitted.
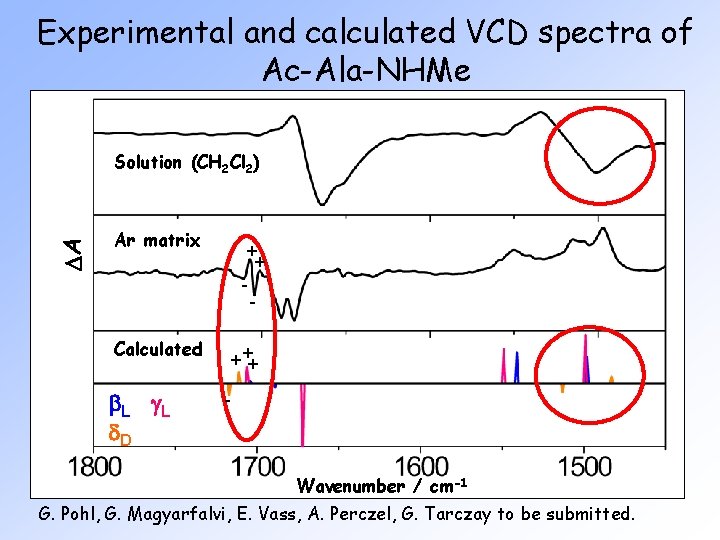
Experimental and calculated VCD spectra of Ac-Ala-NHMe DA Solution (CH 2 Cl 2) Ar matrix Calculated b L g. L d. D + ++ - Wavenumber / cm-1 G. Pohl, G. Magyarfalvi, E. Vass, A. Perczel, G. Tarczay to be submitted.
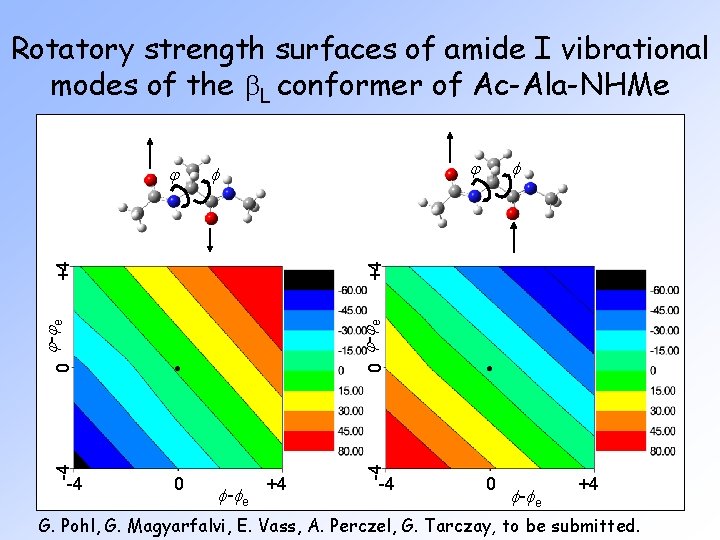
Rotatory strength surfaces of amide I vibrational modes of the b. L conformer of Ac-Ala-NHMe j 0 j -j e 0 f -f e +4 -4 j -j e 0 -4 -4 f +4 j -4 0 f -f e +4 G. Pohl, G. Magyarfalvi, E. Vass, A. Perczel, G. Tarczay, to be submitted.
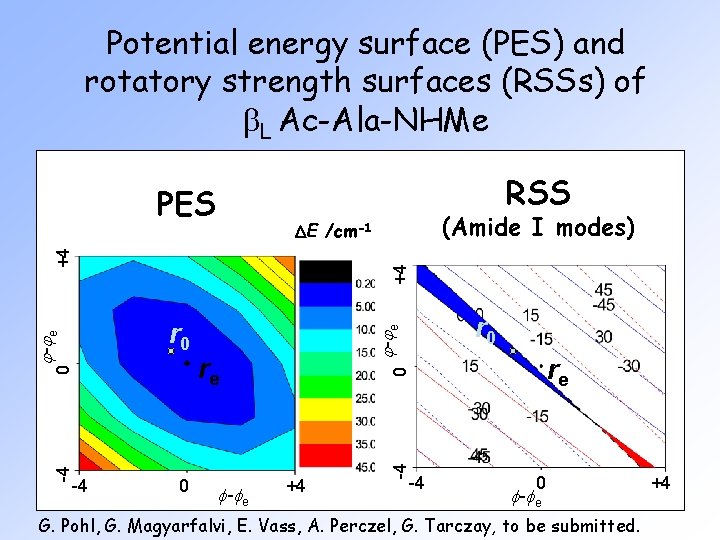
Potential energy surface (PES) and rotatory strength surfaces (RSSs) of b. L Ac-Ala-NHMe RSS PES +4 +4 0 j -j e re 0 re f -f e r 0 +4 -4 -4 0 j -j e r 0 -4 (Amide I modes) DE /cm-1 -4 0 f -f e G. Pohl, G. Magyarfalvi, E. Vass, A. Perczel, G. Tarczay, to be submitted. +4
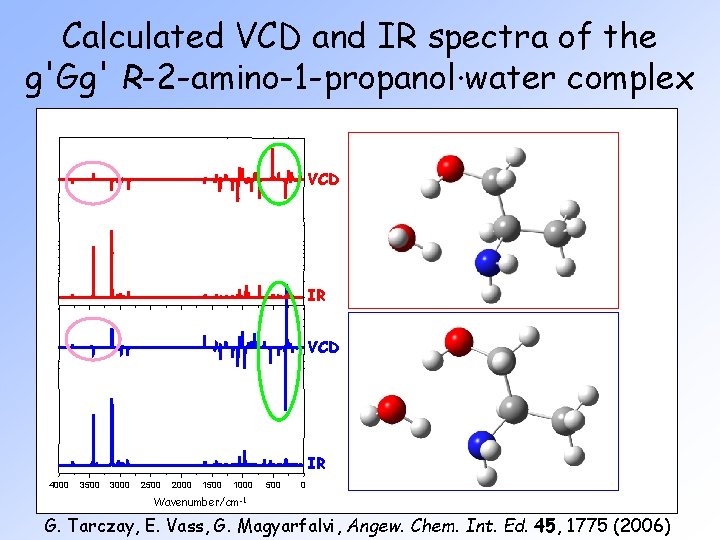
Calculated VCD and IR spectra of the g'Gg' R-2 -amino-1 -propanol∙water complex VCD IR 4000 3500 3000 2500 2000 1500 1000 500 0 Wavenumber/cm-1 G. Tarczay, E. Vass, G. Magyarfalvi, Angew. Chem. Int. Ed. 45, 1775 (2006)
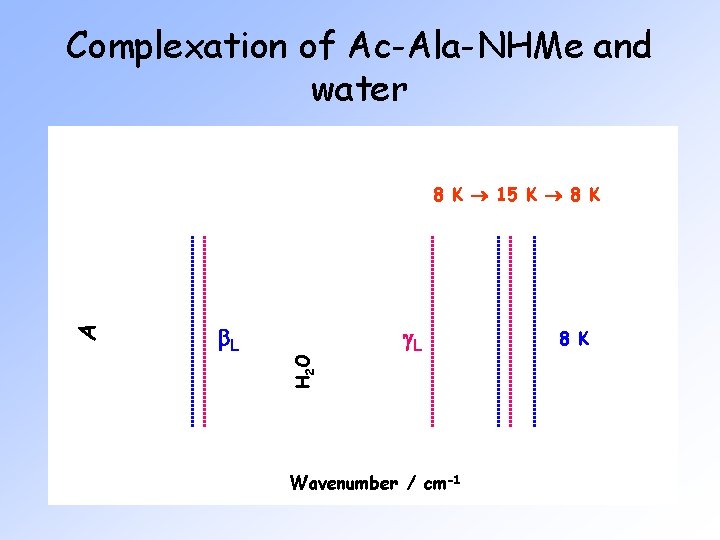
Complexation of Ac-Ala-NHMe and water b. L H 2 O A 8 K 15 K 8 K g. L Wavenumber / cm-1 8 K

Matrix-isolation VCD spectroscopy ADVANTAGES • Investigation of complex systems (multiple conformers, complexes, radicals) • Simultaneous determination of configuration and conformation • It can help the assignment of matrix isolation IR spectra • Neither aggregates nor complexes with solvents have to be considered for theoretical calculations LIMITATIONS • Maximum size of the molecule that can be evaporated (Knudsen cell) • Maximum size of the molecule that can be calculated (parallel codes) DIFFICULTIES • thick and homogenous matrices are required (long deposition times) • light scattering on thick layers (long measurements) • possible artifacts due to non-parallel alignment, strength in window material (Cs. I Ba. F 2), interference, background (dual modulation)

Matrix 2006 – Eötvös University György Tarczay Gábor Magyarfalvi Elemér Vass „Matrix isolation remains an activity involving ‘anti-social’ hours of work. ” Cradock and Hinchliffe: Matrix Isolation, Cambridge University Press Edit Mátyus Gábor Bazsó Gábor Pohl
- Slides: 18