Olaparib nel trattamento della recidiva BRCAmutata di carcinoma


Olaparib nel trattamento della recidiva BRCAmutata di carcinoma ovarico Vanda Salutari Ginecologia oncologica Policlinico A. Gemelli Roma Sabrina Chiara Cecere Oncologia Medica Uro-Ginecologica IRCCS - Fondazione “G. Pascale” Napoli

Ovarian cancer: not unique disease
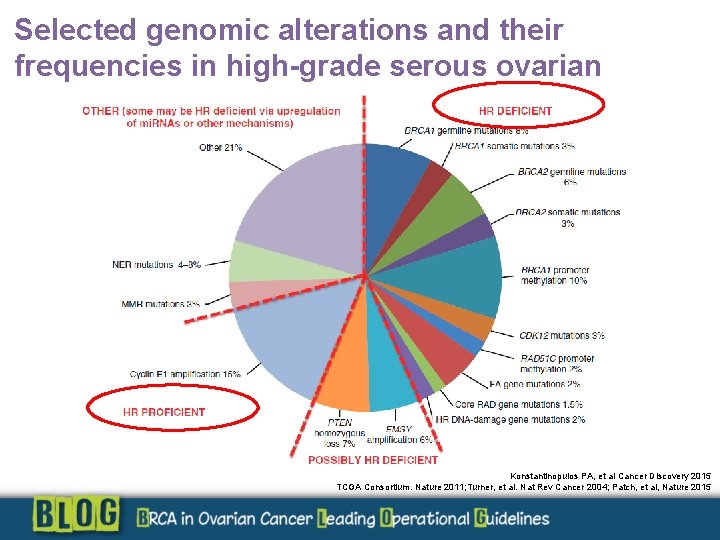
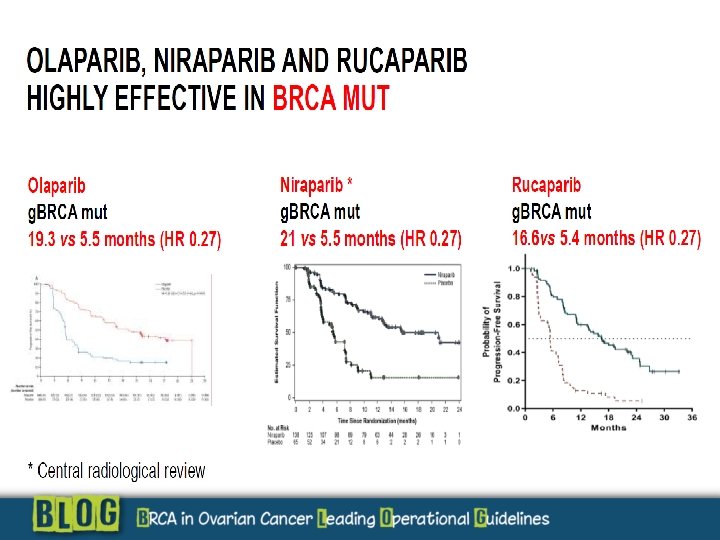
Selected genomic alterations and their frequencies in high-grade serous ovarian Konstantinopulos PA, et al Cancer Discovery 2015 TCGA Consortium. Nature 2011; Turner, et al. Nat Rev Cancer 2004; Patch, et al, Nature 2015

Prevalence of BRCA mutations in ovarian cancer § Approximately 50% of HGSOCs exhibit genetic or epigenetic alterations of HR pathway genes. § Germline BRCA 1 and BRCA 2 mutations are the most common alterations, and are present in: 14– 15% of all EOCs 22. 6% of HGSOCs § Somatic BRCA 1 and BRCA 2 mutations have been identified in 6 -7% of HGSOCs § Up to 44% have no family history Konstantinopulos PA, et al Cancer Discovery 2015
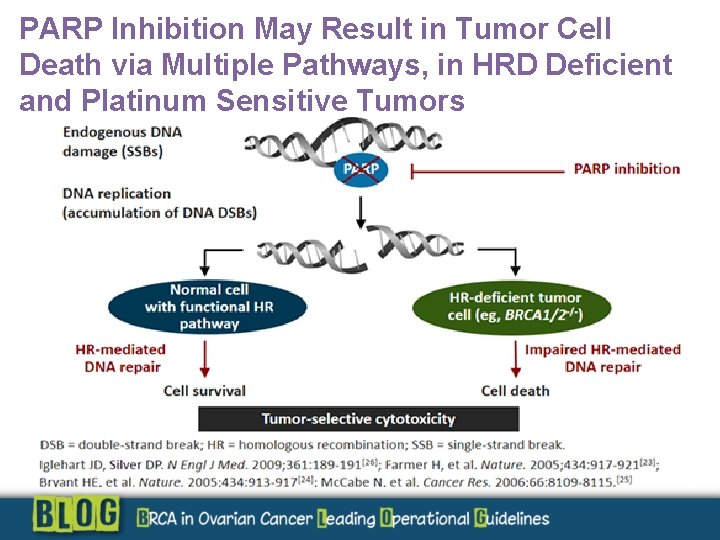
PARP Inhibition May Result in Tumor Cell Death via Multiple Pathways, in HRD Deficient and Platinum Sensitive Tumors
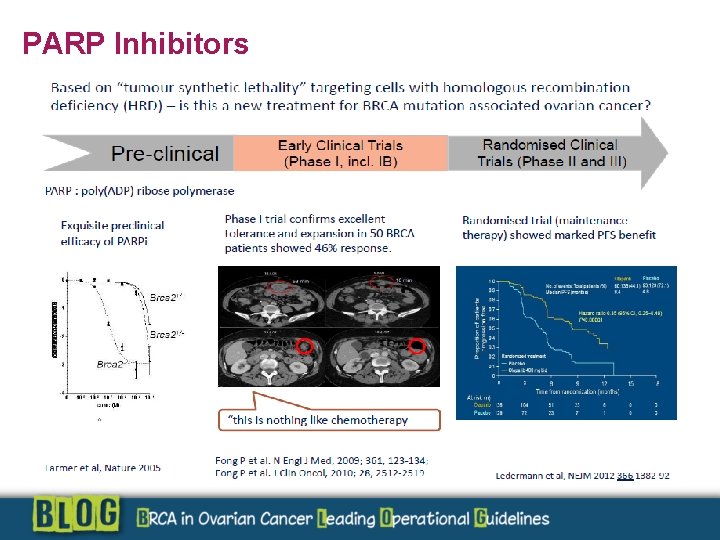
PARP Inhibitors

Olaparib indications: US vs Europe US approval: PAST: Olaparib monotherapy in patient with germline mutated BRCA OC who have received > 3 or more chemotherapy treatments together with a companion diagnsostic test (BRCA analysis CDx). 1 PRESENT: maintenance treatment of adult patients with recurrent epithelial ovarian, fallopian tube or primary peritoneal cancer, who are in a complete or partial response to platinum-based chemotherapy. European approval: Olaparib monotherapy for the maintenance treatment of adult patient with platinum-sensitive relapsed BRCA-mutated (Germline/somatic) OC, high grade sereous, fallopian tube, or primary peritoneal cancer who are in response (compleate or partial) to platinum-based chemotherapy. 2 From February 2018 Lynparza is indicated as monotherapy for the maintenance treatment of adult patients with platinum-sensitive relapsed high-grade epithelial ovarian, fallopian tube, or primary peritoneal cancer who are in response (complete or partial) to platinum-based chemotherapy 1. Kaufman B, et al. J Clin Oncol 2015. 2. Ledermann J et al. New Engl J Med 2012
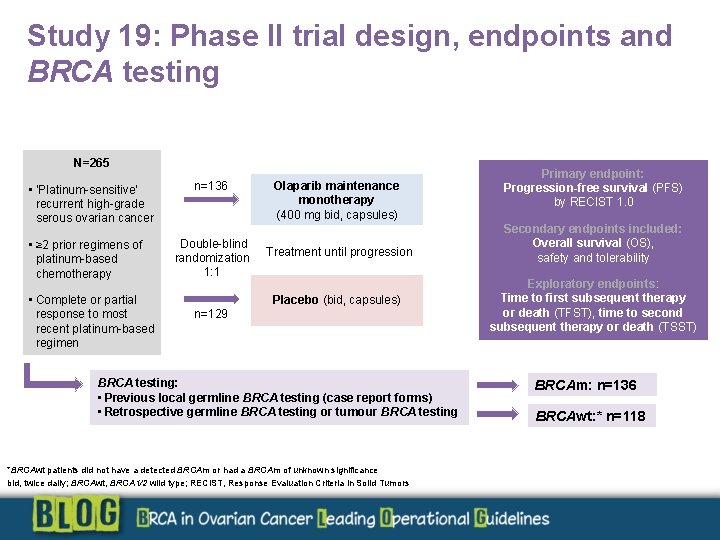
Study 19: Phase II trial design, endpoints and BRCA testing N=265 • ‘Platinum-sensitive’ recurrent high-grade serous ovarian cancer • ≥ 2 prior regimens of platinum-based chemotherapy • Complete or partial response to most recent platinum-based regimen n=136 Double-blind randomization 1: 1 Olaparib maintenance monotherapy (400 mg bid, capsules) Treatment until progression Placebo (bid, capsules) n=129 BRCA testing: • Previous local germline BRCA testing (case report forms) • Retrospective germline BRCA testing or tumour BRCA testing *BRCAwt patients did not have a detected BRCAm or had a BRCAm of unknown significance bid, twice daily; BRCAwt, BRCA 1/2 wild type; RECIST, Response Evaluation Criteria in Solid Tumors Primary endpoint: Progression-free survival (PFS) by RECIST 1. 0 Secondary endpoints included: Overall survival (OS), safety and tolerability Exploratory endpoints: Time to first subsequent therapy or death (TFST), time to second subsequent therapy or death (TSST) BRCAm: n=136 BRCAwt: * n=118
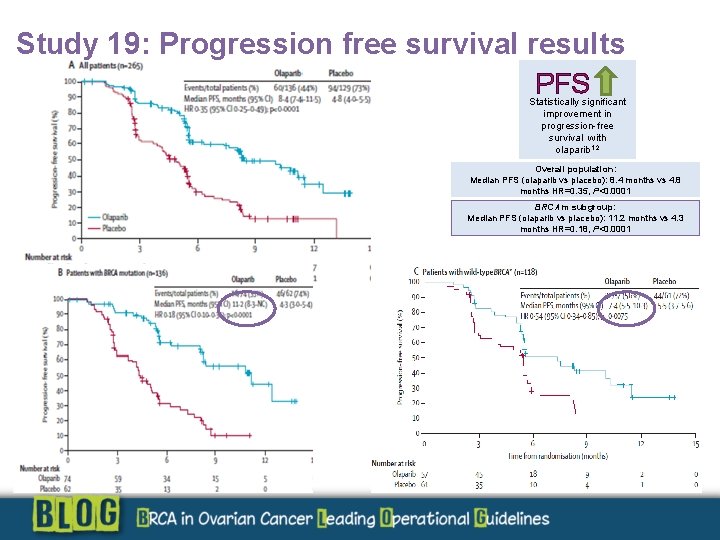
Study 19: Progression free survival results PFS Statistically significant improvement in progression-free survival with olaparib 1, 2 Overall population: Median PFS (olaparib vs placebo): 8. 4 months vs 4. 8 months HR=0. 35, P<0. 0001 BRCAm subgroup: Median PFS (olaparib vs placebo): 11. 2 months vs 4. 3 months HR=0. 18, P<0. 0001
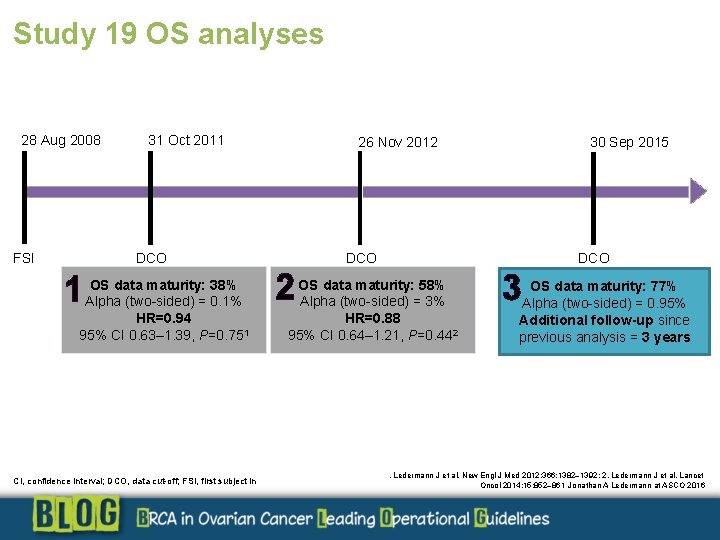
Study 19 OS analyses 28 Aug 2008 31 Oct 2011 DCO FSI 1 OS data maturity: 38% Alpha (two-sided) = 0. 1% HR=0. 94 95% CI 0. 63– 1. 39, P=0. 751 CI, confidence interval; DCO, data cut-off; FSI, first subject in 30 Sep 2015 26 Nov 2012 2 DCO OS data maturity: 58% Alpha (two-sided) = 3% HR=0. 88 95% CI 0. 64– 1. 21, P=0. 442 3 OS data maturity: 77% Alpha (two-sided) = 0. 95% Additional follow-up since previous analysis = 3 years . Ledermann J et al. New Engl J Med 2012; 366: 1382– 1392; 2. Ledermann J et al. Lancet Oncol 2014; 15: 852– 861 Jonathan A Ledermann at ASCO 2016
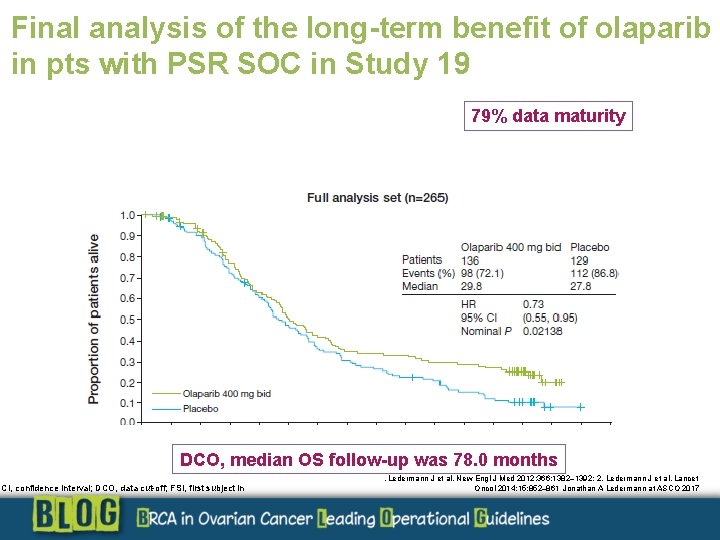
Final analysis of the long-term benefit of olaparib in pts with PSR SOC in Study 19 79% data maturity DCO, median OS follow-up was 78. 0 months CI, confidence interval; DCO, data cut-off; FSI, first subject in . Ledermann J et al. New Engl J Med 2012; 366: 1382– 1392; 2. Ledermann J et al. Lancet Oncol 2014; 15: 852– 861 Jonathan A Ledermann at ASCO 2017
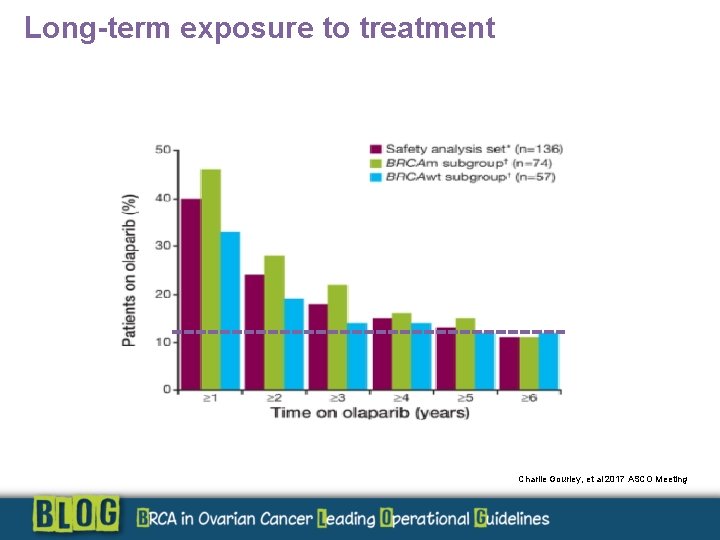
Long-term exposure to treatment Charlie Gourley, et al 2017 ASCO Meeting
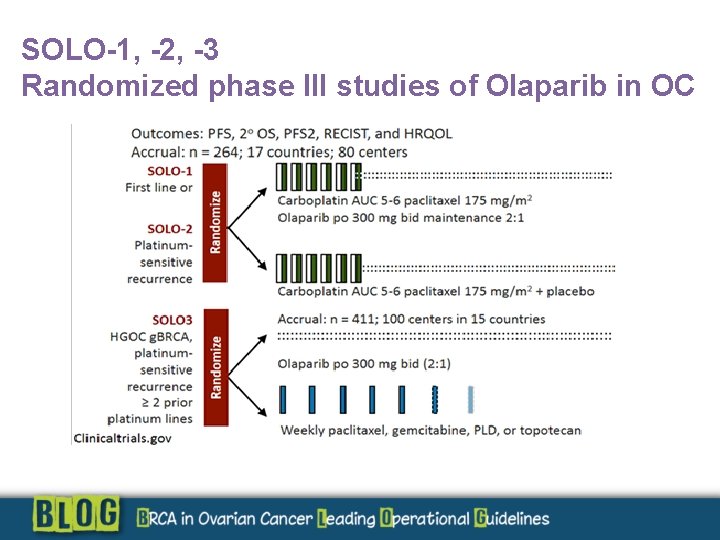
SOLO-1, -2, -3 Randomized phase III studies of Olaparib in OC
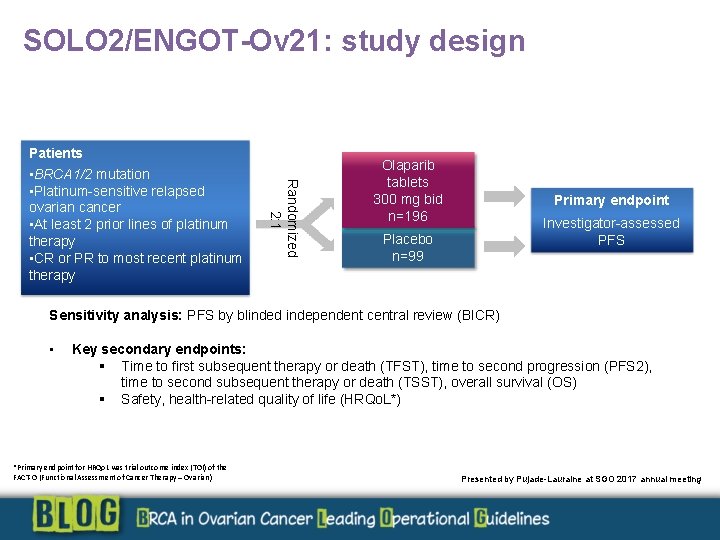
SOLO 2/ENGOT-Ov 21: study design Randomized 2: 1 Patients • BRCA 1/2 mutation • Platinum-sensitive relapsed ovarian cancer • At least 2 prior lines of platinum therapy • CR or PR to most recent platinum therapy Olaparib tablets 300 mg bid n=196 Primary endpoint Investigator-assessed PFS Placebo n=99 Sensitivity analysis: PFS by blinded independent central review (BICR) • Key secondary endpoints: § Time to first subsequent therapy or death (TFST), time to second progression (PFS 2), time to second subsequent therapy or death (TSST), overall survival (OS) § Safety, health-related quality of life (HRQo. L*) *Primary endpoint for HRQo. L was trial outcome index (TOI) of the FACT-O (Functional Assessment of Cancer Therapy – Ovarian) Presented by Pujade-Lauraine at SGO 2017 annual meeting
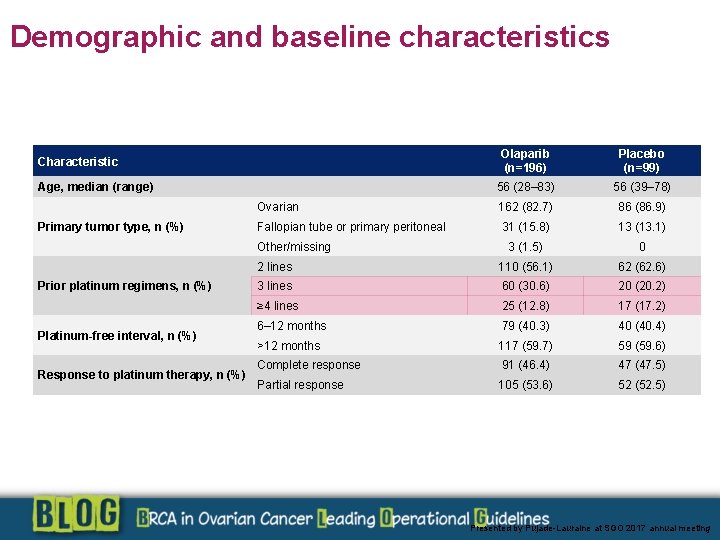
Demographic and baseline characteristics Olaparib (n=196) Placebo (n=99) 56 (28– 83) 56 (39– 78) Ovarian 162 (82. 7) 86 (86. 9) Fallopian tube or primary peritoneal 31 (15. 8) 13 (13. 1) 3 (1. 5) 0 2 lines 110 (56. 1) 62 (62. 6) 3 lines 60 (30. 6) 20 (20. 2) ≥ 4 lines 25 (12. 8) 17 (17. 2) 6– 12 months 79 (40. 3) 40 (40. 4) >12 months 117 (59. 7) 59 (59. 6) Complete response 91 (46. 4) 47 (47. 5) Partial response 105 (53. 6) 52 (52. 5) Characteristic Age, median (range) Primary tumor type, n (%) Other/missing Prior platinum regimens, n (%) Platinum-free interval, n (%) Response to platinum therapy, n (%) Presented by Pujade-Lauraine at SGO 2017 annual meeting
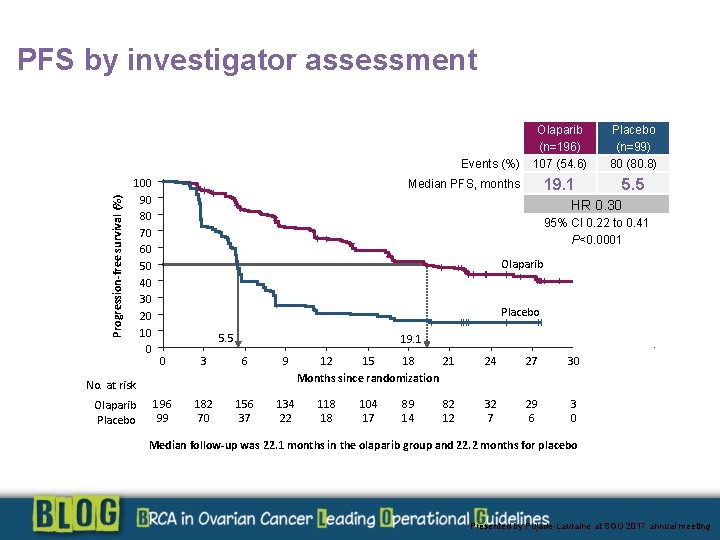
PFS by investigator assessment Progression-free survival (%) Events (%) 100 90 80 70 60 50 40 30 20 10 0 Placebo (n=99) 80 (80. 8) 19. 1 5. 5 Median PFS, months HR 0. 30 95% CI 0. 22 to 0. 41 P<0. 0001 Olaparib Placebo 5. 5 19. 1 0 3 6 9 196 99 182 70 156 37 134 22 No. at risk Olaparib Placebo Olaparib (n=196) 107 (54. 6) 12 15 18 21 Months since randomization 118 18 104 17 89 14 82 12 24 27 30 32 7 29 6 3 0 33 2 0 36 0 0 Median follow-up was 22. 1 months in the olaparib group and 22. 2 months for placebo Presented by Pujade-Lauraine at SGO 2017 annual meeting
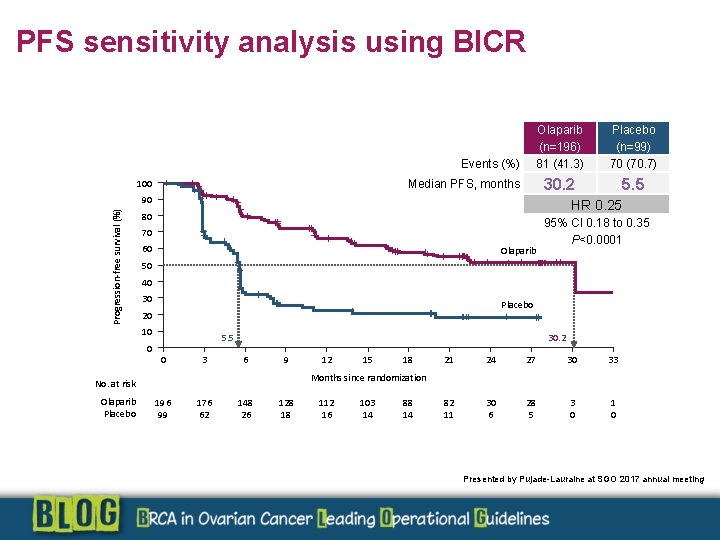
PFS sensitivity analysis using BICR Events (%) Olaparib (n=196) 81 (41. 3) Placebo (n=99) 70 (70. 7) 30. 2 5. 5 Median PFS, months 100 Progression-free survival (%) 90 HR 0. 25 80 70 60 Olaparib 50 40 30 Placebo 20 10 0 5. 5 0 3 30. 2 6 9 12 15 18 21 24 27 30 33 82 11 30 6 28 5 3 0 1 0 Months since randomization No. at risk Olaparib Placebo 95% CI 0. 18 to 0. 35 P<0. 0001 196 99 176 62 148 26 128 18 112 16 103 14 88 14 Presented by Pujade-Lauraine at SGO 2017 annual meeting
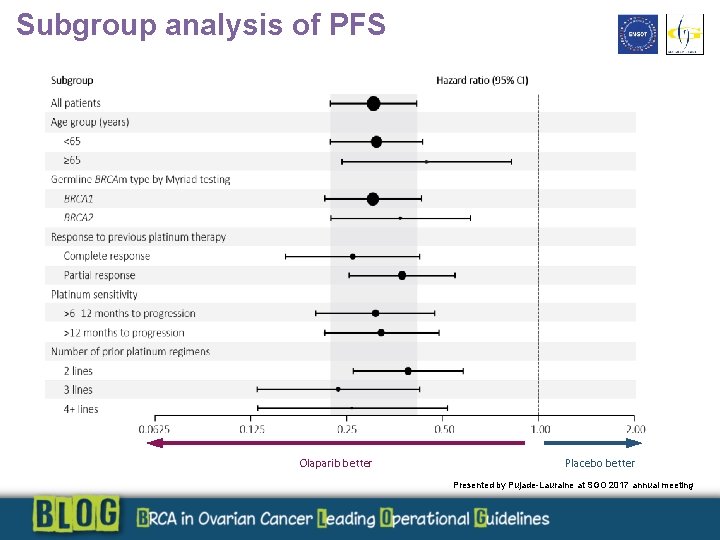
Subgroup analysis of PFS Olaparib better Placebo better Presented by Pujade-Lauraine at SGO 2017 annual meeting
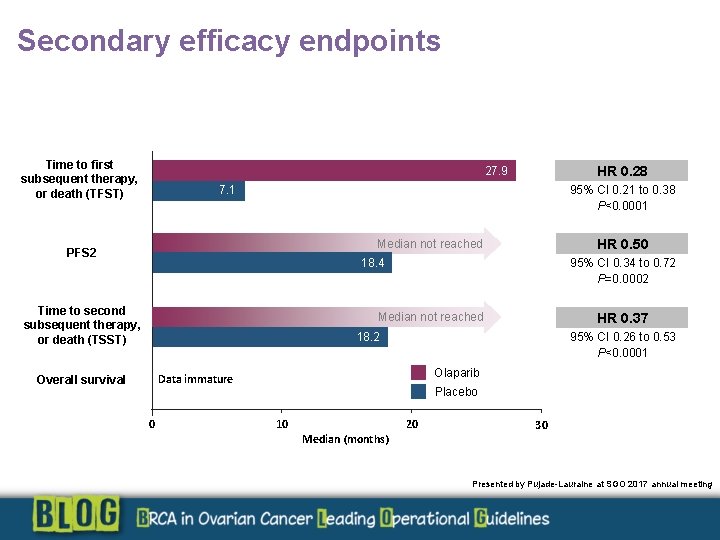
Secondary efficacy endpoints Time to first subsequent therapy, or death (TFST) HR 0. 28 27. 9 95% CI 0. 21 to 0. 38 P<0. 0001 7. 1 HR 0. 50 Median not reached PFS 2 95% CI 0. 34 to 0. 72 P=0. 0002 18. 4 Time to second subsequent therapy, or death (TSST) Not reached Median not reached HR 0. 37 95% CI 0. 26 to 0. 53 P<0. 0001 18. 2 Olaparib Data immature Overall survival 0 Placebo 10 Median (months) 20 30 Presented by Pujade-Lauraine at SGO 2017 annual meeting
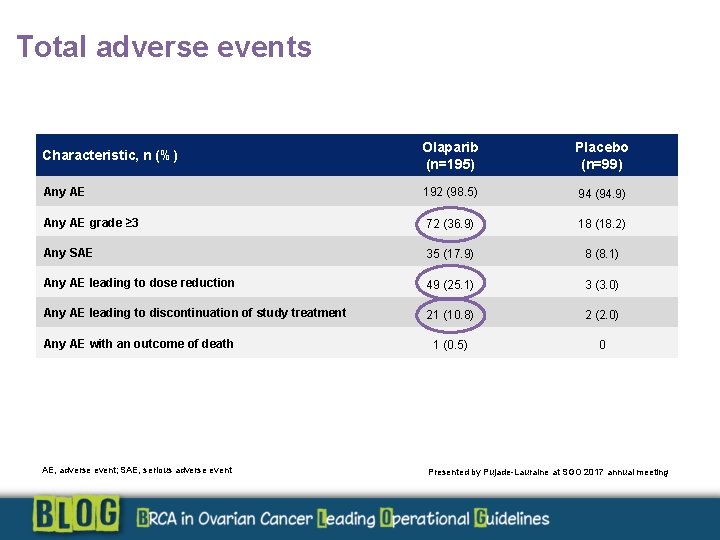
Total adverse events Characteristic, n (%) Olaparib (n=195) Placebo (n=99) Any AE 192 (98. 5) 94 (94. 9) Any AE grade ≥ 3 72 (36. 9) 18 (18. 2) Any SAE 35 (17. 9) 8 (8. 1) Any AE leading to dose reduction 49 (25. 1) 3 (3. 0) Any AE leading to discontinuation of study treatment 21 (10. 8) 2 (2. 0) 1 (0. 5) 0 Any AE with an outcome of death AE, adverse event; SAE, serious adverse event Presented by Pujade-Lauraine at SGO 2017 annual meeting
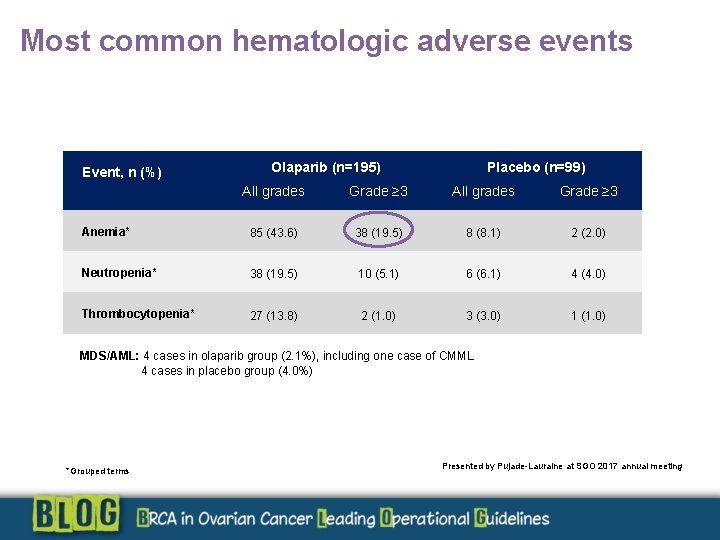
Most common hematologic adverse events Event, n (%) Olaparib (n=195) Placebo (n=99) All grades Grade ≥ 3 Anemia* 85 (43. 6) 38 (19. 5) 8 (8. 1) 2 (2. 0) Neutropenia* 38 (19. 5) 10 (5. 1) 6 (6. 1) 4 (4. 0) Thrombocytopenia* 27 (13. 8) 2 (1. 0) 3 (3. 0) 1 (1. 0) MDS/AML: 4 cases in olaparib group (2. 1%), including one case of CMML 4 cases in placebo group (4. 0%) *Grouped terms Presented by Pujade-Lauraine at SGO 2017 annual meeting
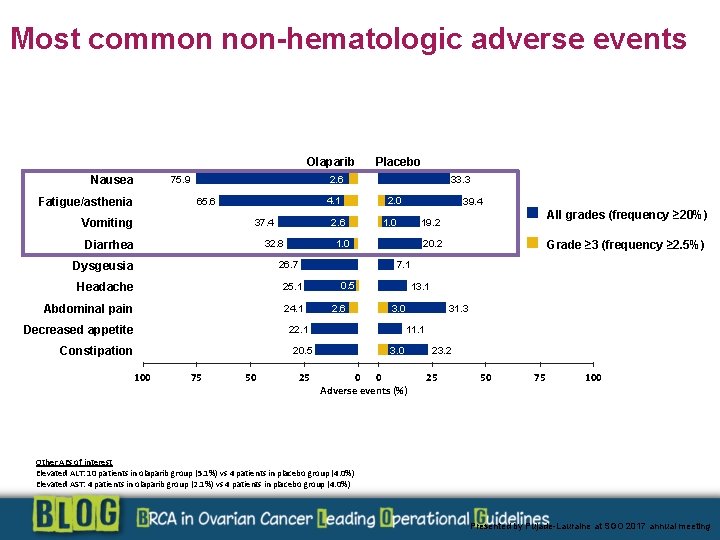
Most common non-hematologic adverse events Olaparib Nausea 2. 6 75. 9 Fatigue/asthenia Placebo 4. 1 65. 6 Vomiting Diarrhea 26. 7 1. 0 25. 1 Abdominal pain 24. 1 0. 5 2. 6 13. 1 3. 0 75 50 25 31. 3 11. 1 3. 0 20. 5 100 Grade ≥ 3 (frequency ≥ 2. 5%) 20. 2 22. 1 Constipation All grades (frequency ≥ 20%) 19. 2 7. 1 Headache Decreased appetite 39. 4 1. 0 32. 8 Dysgeusia 2. 0 2. 6 37. 4 33. 3 0 0 Adverse events (%) 23. 2 25 50 75 100 Other AEs of interest Elevated ALT: 10 patients in olaparib group (5. 1%) vs 4 patients in placebo group (4. 0%) Elevated AST: 4 patients in olaparib group (2. 1%) vs 4 patients in placebo group (4. 0%) Presented by Pujade-Lauraine at SGO 2017 annual meeting
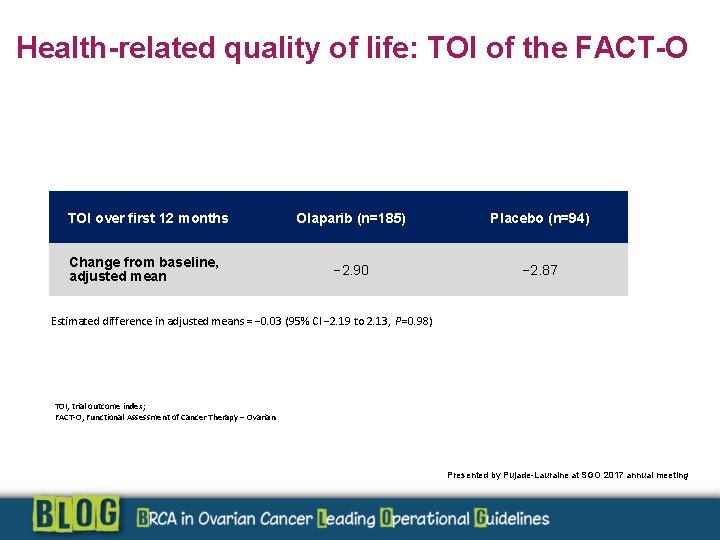
Health-related quality of life: TOI of the FACT-O TOI over first 12 months Change from baseline, adjusted mean Olaparib (n=185) Placebo (n=94) − 2. 90 − 2. 87 Estimated difference in adjusted means = − 0. 03 (95% CI − 2. 19 to 2. 13, P=0. 98) TOI, trial outcome index; FACT-O, Functional Assessment of Cancer Therapy – Ovarian Presented by Pujade-Lauraine at SGO 2017 annual meeting
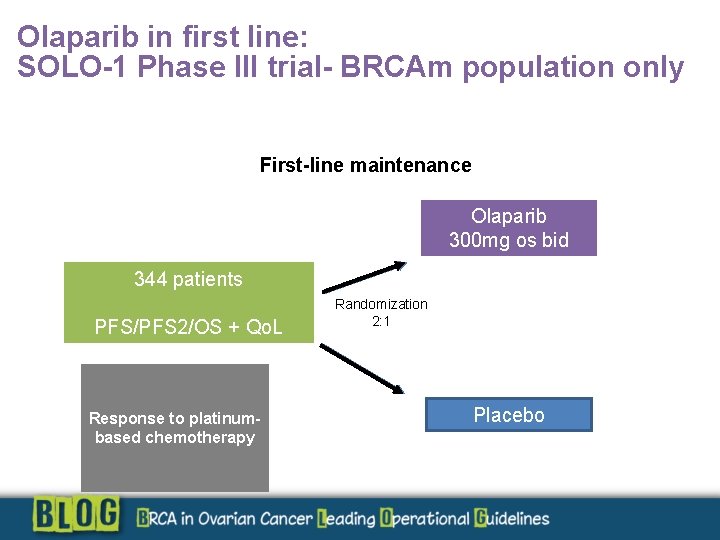
Olaparib in first line: SOLO-1 Phase III trial- BRCAm population only First-line maintenance Olaparib 300 mg os bid 344 patients PFS/PFS 2/OS + Qo. L Response to platinumbased chemotherapy Randomization 2: 1 Placebo
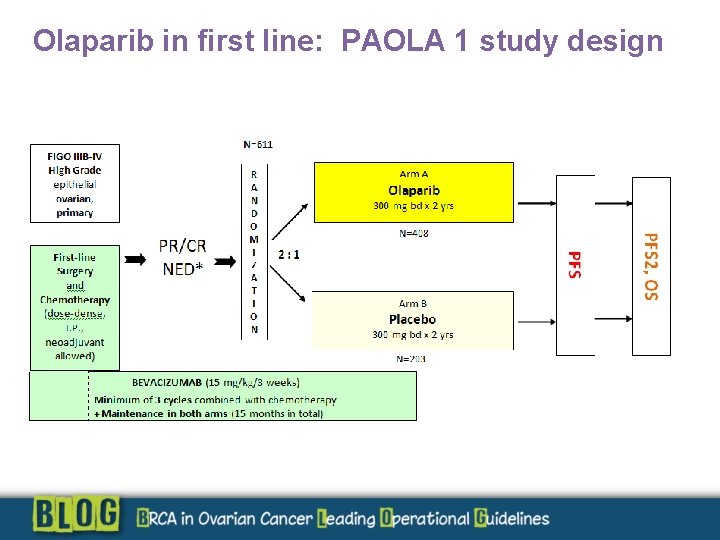
Olaparib in first line: PAOLA 1 study design

Unsolved questions on HRD and PARP inhibitors

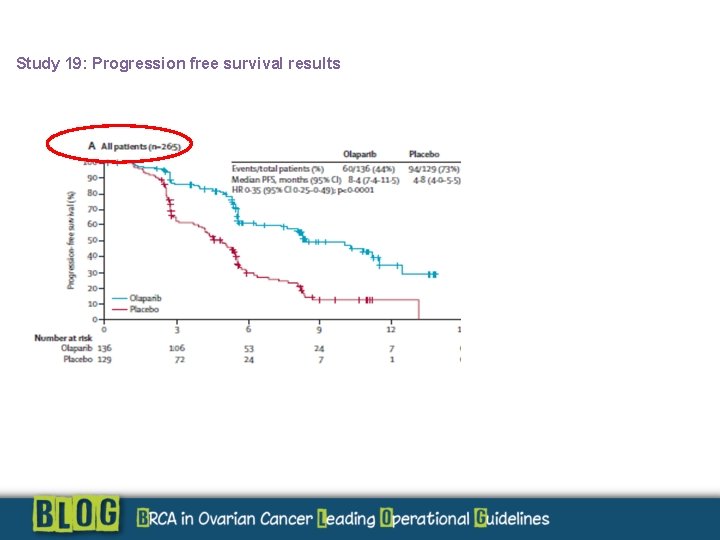
Study 19: Progression free survival results

Why do some HRD negative cases respond to platinum and to PARPi? The test can miss some HRD positive cases? HRD tumours are more prevalent than previously believed in serous high grade? Other non HRD DNA repair impaired mechanisms in these patients?

Why do some HRD negative cases respond to platinum and to PARPi? The test can miss some HRD positive cases? HRD tumours are more prevalent than previously believed in serous high grade? Other non HRD DNA repair impaired mechanisms in these patients?

Why some HRD negative cases respond to platinum and to PARPi? Does the test miss some HRD positive cases? Specificity is not 100% u Basal heterogeneity u Prospective genetic instability? Chien J et al Front Oncol 2013 ENGOT-OV 16/NOVA Trial discussed by Sandro Pignata Barbara Norquist at 2017 ASCO Annual Meeting
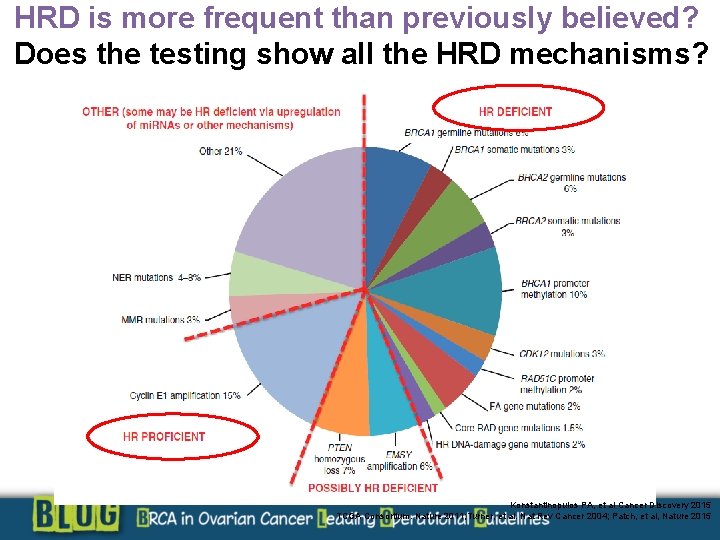
HRD is more frequent than previously believed? Does the testing show all the HRD mechanisms? Konstantinopulos PA, et al Cancer Discovery 2015 TCGA Consortium. Nature 2011; Turner, et al. Nat Rev Cancer 2004; Patch, et al, Nature 2015

Comparing the toxicity of PARP inhibitors
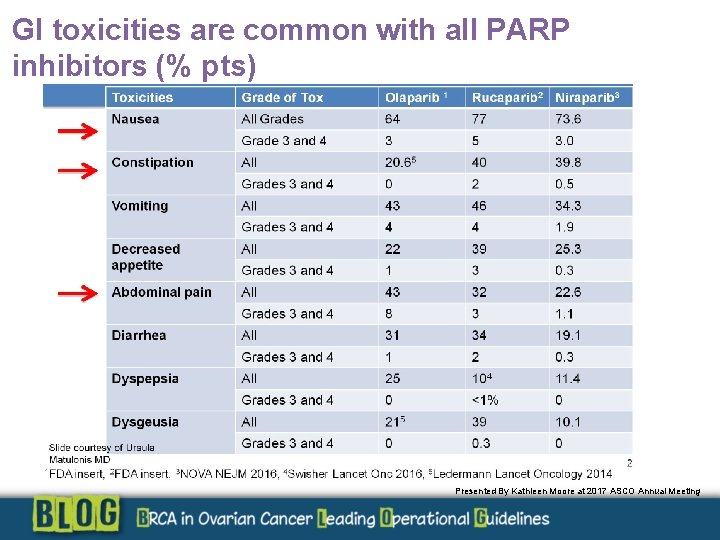
GI toxicities are common with all PARP inhibitors (% pts) GI toxicities are common with all PARP inhibitors (% of pts) Presented By Kathleen Moore at 2017 ASCO Annual Meeting
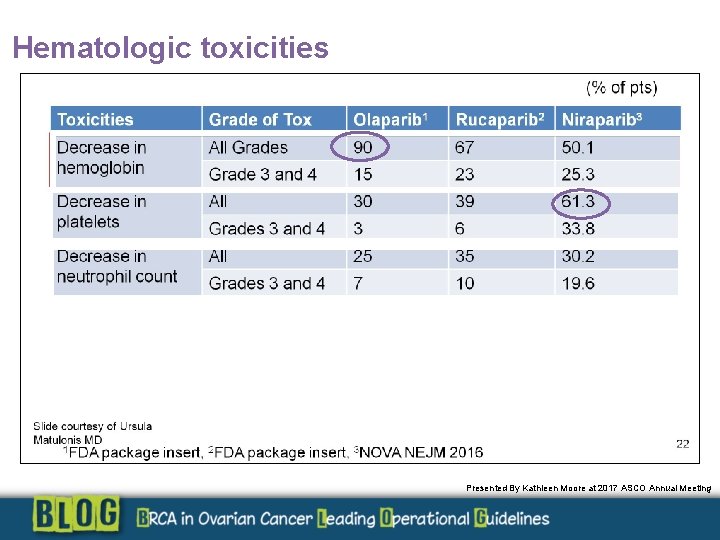
Hematologic toxicities Presented By Kathleen Moore at 2017 ASCO Annual Meeting
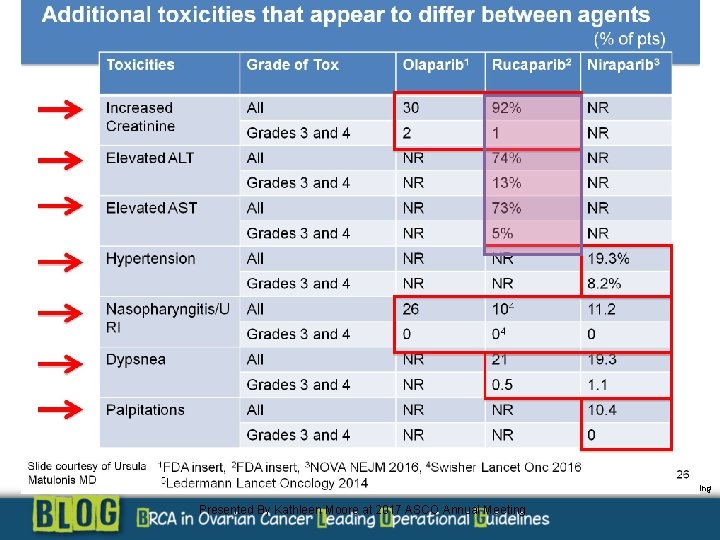
Additional toxicities that appear to differ between agents Presented By Kathleen Moore at 2017 ASCO Annual Meeting

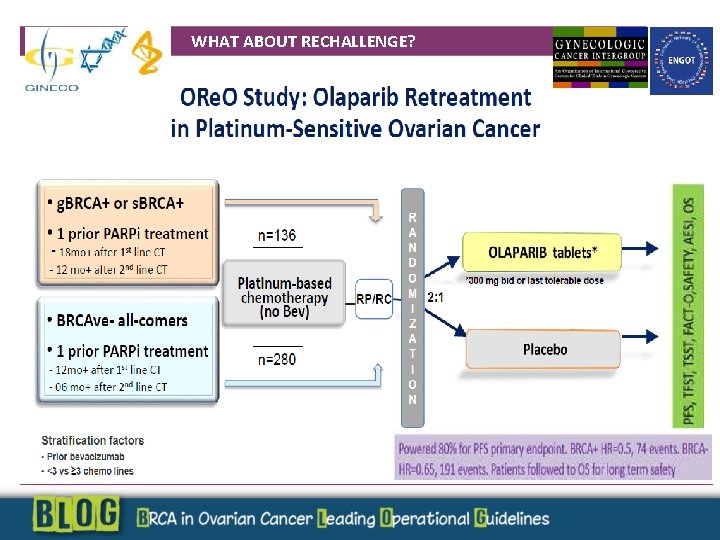
WHAT ABOUT RECHALLENGE?

Perspectives for PARP INHIBITION WHAT ABOUT RECHALLENGE? Combinations : Olaparib + immuno check point inhibitors Resistant disease: Wee-1 inhibitors + Olaparib (phase 1)
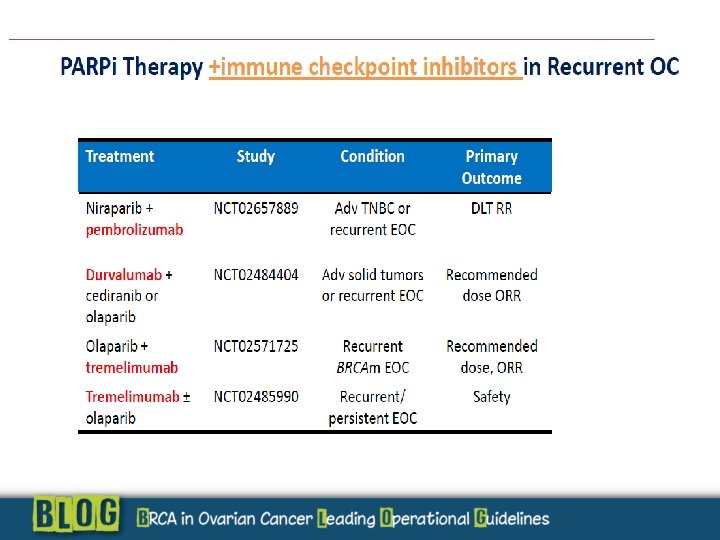

Resistence to PARP inhibitors
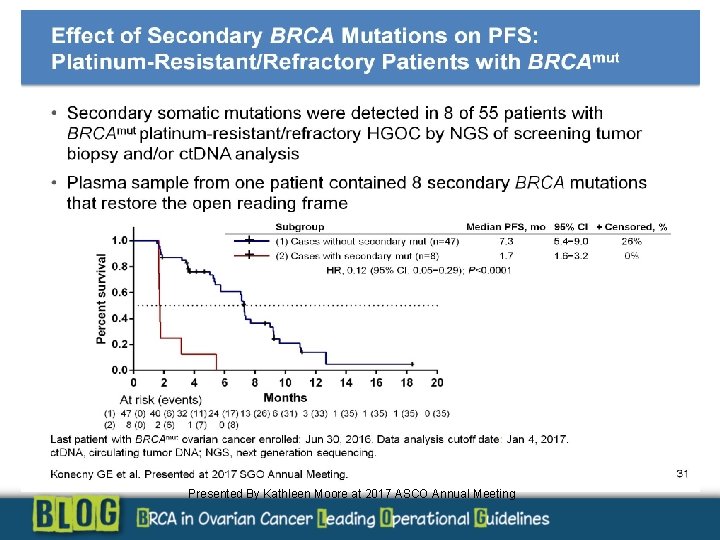
Presented By Kathleen Moore at 2017 ASCO Annual Meeting
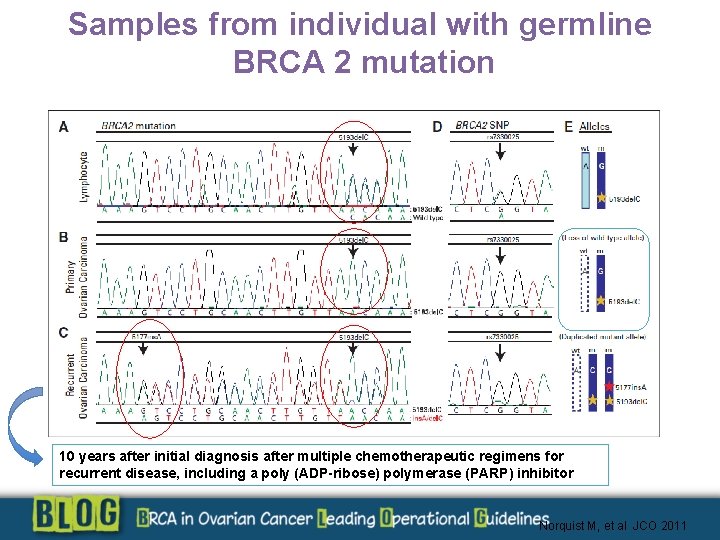
Samples from individual with germline BRCA 2 mutation 10 years after initial diagnosis after multiple chemotherapeutic regimens for recurrent disease, including a poly (ADP-ribose) polymerase (PARP) inhibitor Norquist M, et al JCO 2011
- Slides: 45