OHRE Update Whats new with the IRBs Daniel

OHRE Update: What’s new with the IRBs? Daniel Nelson Director, Office of Human Research Ethics Associate Professor of Social Medicine and Pediatrics University of North Carolina- Chapel Hill

Office of Human Research Ethics Responsible for ethical and regulatory oversight of research at UNC-Chapel Hill that involves human subjects l OHRE administers, supports, guides and oversees the work of the campus Institutional Review Boards (IRBs) and all related activities l
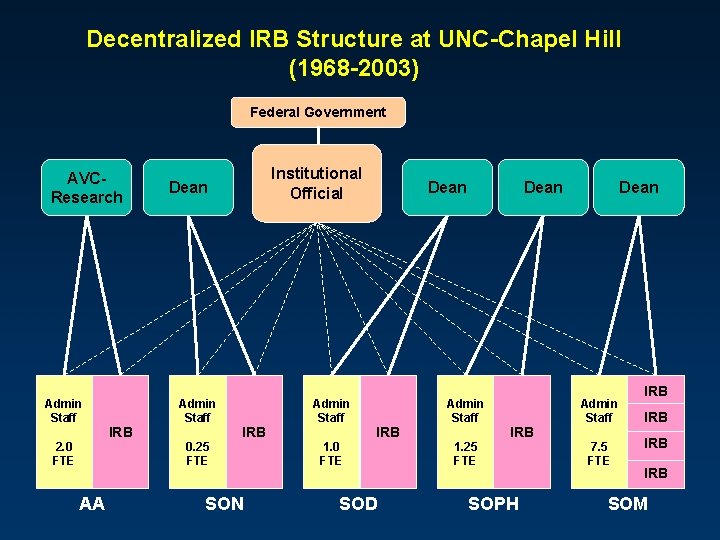
Decentralized IRB Structure at UNC-Chapel Hill (1968 -2003) Federal Government AVCResearch Admin Staff 2. 0 FTE AA IRB Institutional Official Dean Admin Staff 0. 25 FTE IRB SON Admin Staff 1. 0 FTE Dean IRB SOD Dean Admin Staff 1. 25 FTE IRB SOPH Dean Admin Staff 7. 5 FTE IRB IRB SOM

Problems with Decentralized Structure l Inefficiency 5 of everything! l l l Variability l l l 5 sets of operating procedures 5 websites and databases 5 application forms 5 documentation standards 5 channels for communication and policy Inconsistency in applying common regulations Little sharing of best practices Staffing and budgetary support Workload, experience, expertise, compliance Fragmented reporting lines l l School-based operations University-based obligations
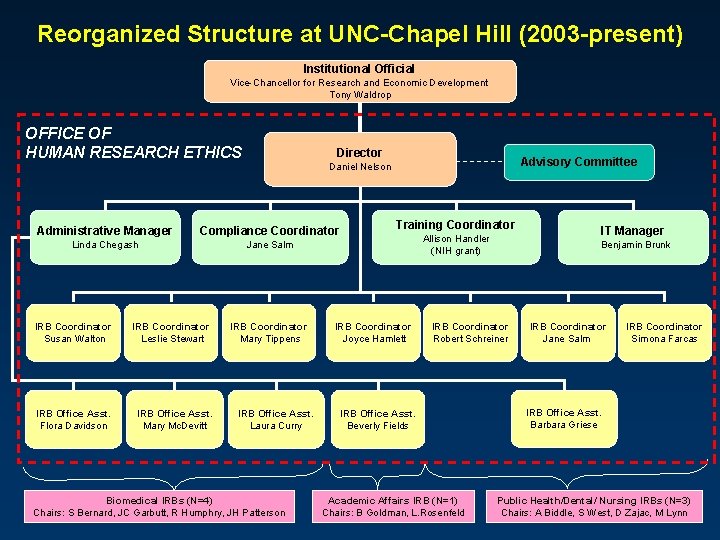
Reorganized Structure at UNC-Chapel Hill (2003 -present) Institutional Official Vice-Chancellor for Research and Economic Development Tony Waldrop OFFICE OF HUMAN RESEARCH ETHICS Director Administrative Manager Compliance Coordinator Linda Chegash Jane Salm IRB Coordinator Susan Walton IRB Office Asst. Flora Davidson IRB Coordinator Leslie Stewart IRB Office Asst. Mary Mc. Devitt Advisory Committee Daniel Nelson IRB Coordinator Mary Tippens IRB Office Asst. Laura Curry Biomedical IRBs (N=4) Chairs: S Bernard, JC Garbutt, R Humphry, JH Patterson Training Coordinator Allison Handler (NIH grant) IRB Coordinator Joyce Hamlett IRB Coordinator Robert Schreiner IRB Office Asst. Beverly Fields Academic Affairs IRB (N=1) Chairs: B Goldman, L. Rosenfeld IT Manager Benjamin Brunk IRB Coordinator Jane Salm IRB Coordinator Simona Farcas IRB Office Asst. Barbara Griese Public Health/Dental/ Nursing IRBs (N=3) Chairs: A Biddle, S West, D Zajac, M Lynn

“Nice org chart. . . So what have you been doing for the past year? ”
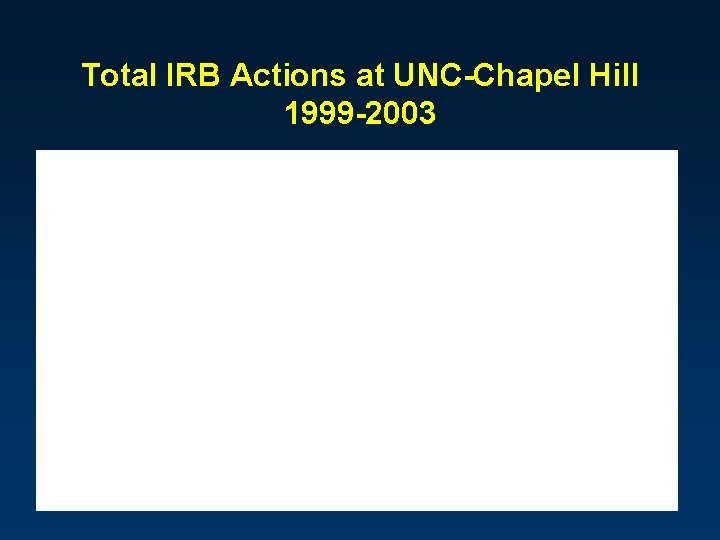
Total IRB Actions at UNC-Chapel Hill 1999 -2003 6, 202 4, 728 4, 079 4, 850 5, 307

What have we been doing for the past year? l l Ongoing oversight of 4000 active research studies OHRE created! l l l Centralized reporting lines New positions established and filled Existing positions standardized and consolidated Advisory Committee Integrated Budget Model for FY’ 05 Working Groups on Common Practices l l l l Standard Operating Procedures (SOPs) IRB application form Consent form templates Website Database Training policy and modules Public outreach and education Accreditation

What is coming for the research community? l SOPs l l l Common Application and Consent Forms l l Upgraded training for research teams and IRBs (CITI) Linked to application process Public Outreach and Education l l Better management of workflow and protocol info Links to other campus data Access by research teams and dept managers Required Education l l l One stop shopping, enhanced guidance Common Database l l l Less confusion, greater consistency, interchangeable across IRBs Electronic submissions!? Common Website l l Better guidance for IRBs and research teams Investigators Manual Brochure for research volunteers Accreditation l l l Ongoing self-assessment (QA/QI) De facto requirement Demonstration of institutional commitment to highest standards
- Slides: 9