Offlabel and unlicensed medicines in children a population



Off-label and unlicensed medicines in children – a population approach to evidence based treatments James C. Mc. Elnay School of Pharmacy Queen’s University Belfast

Unlicensed and off-label drug use in paediatrics • • 50% in general hospitals 60% in specialist centres 90% in seriously ill neonates Approx. 10% in primary care?

Press coverage

Treatment dilemma • Not desirable to treat children with medicines which have only been tested and licensed for adult use • Not appropriate to withhold potentially effective treatments from children because the manufacturer has not obtained a license for their use in children

Adults compared with children • Testing of drugs in adults involves a series of blood samples taken to measure plasma drug concentrations (large number of samples from a small number of volunteers or patients) • Reluctance to subject children to repeated blood sampling (particularly in neonates) • At present children’s doses often based on adult information • Children are not simply small adults – differences in kinetics (ADME) and dynamics

The problem solved? • Opportunistic drug levels measured in routine blood samples in hospitalised children who have been prescribed off-label medicines • Small number of samples from large number of children – not large number of samples from small number of children • Close monitoring of children to record beneficial outcomes and side-effects • Specialist statistical analysis (sparse data analysis) performed to provide required information for safe and effective drug use
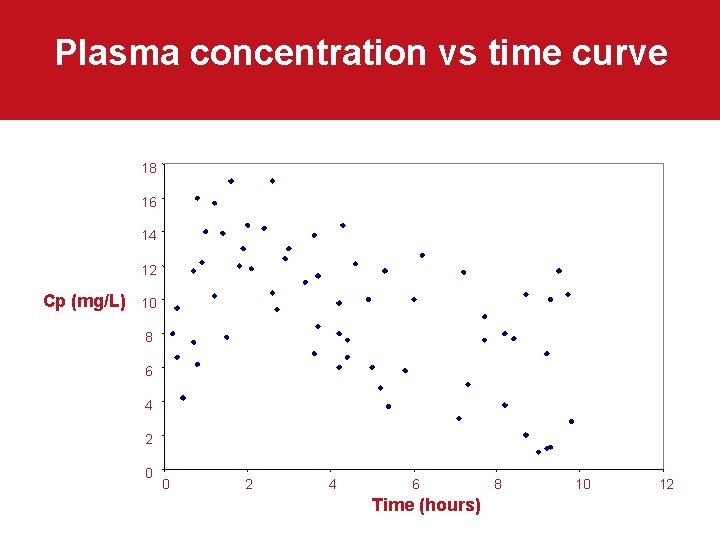
Plasma concentration vs time curve 18 16 14 12 Cp (mg/L) 10 8 6 4 2 0 0 2 4 6 Time (hours) 8 10 12
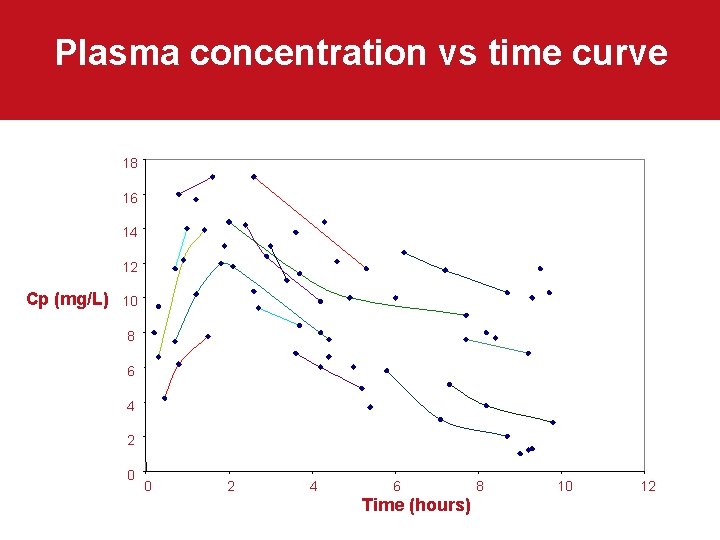
Plasma concentration vs time curve 18 16 14 12 Cp (mg/L) 10 8 6 4 2 0 0 2 4 6 Time (hours) 8 10 12
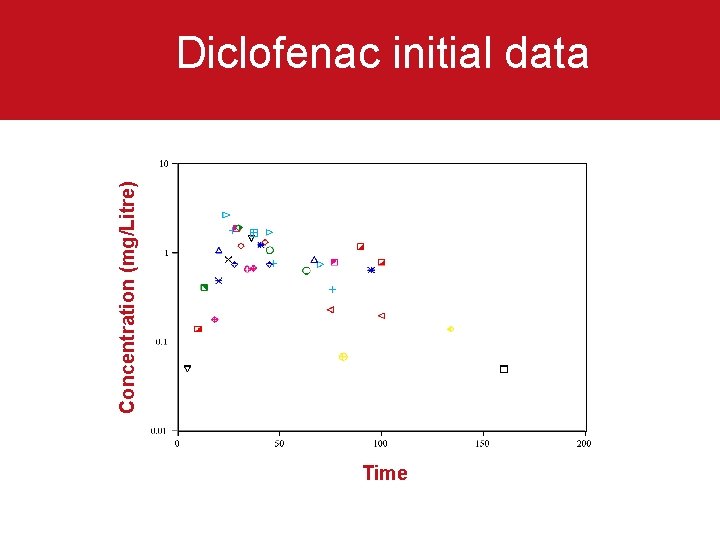
Concentration (mg/Litre) Diclofenac initial data Time

The team • School of Pharmacy, Queen’s u u u James Mc. Elnay Paul Collier Jeff Millership Shirish Yakkundi Shu Chaw Joanne Smyth Godwill Iheagwaram Ahmed Hawwa Maysa Suyah Paul Westwood Aileen Hamilton

The team • Royal Victoria Hospital Belfast u u u Mike Shields Dennis Carson Henry Halliday Rhona Fair Muriel Millar Gillian Thurley • Research links u u Alder Hey Hospital, Liverpool (Tony Nunn) University of Missouri (Greg Kearns) Keio University, Tokyo (Ken Kosaki) Royal Hospital for Sick Children, Glasgow (Neil Morton)

Research process (1) • Select drugs to be studied • Obtain ethical approval for study • Develop range of pharmacodynamic outcome measurement tools and proformas • Develop drug assays for small volume samples • Having obtained parental consent (+ assent of child if 10 years or over) commence collection of blood samples (outpatient clinics, day hospital, inpatients) • Each time a blood sample is collected, pharmacodynamic measurements are undertaken by research nurse

Research process (2) • Blood sample centrifuged and separated plasma frozen (transported to lab on dry ice) • Plasma sample analysed (HPLC) • Patient data entered into study database: - Demographic and clinical data (patient chart) - Dosing regimen and drug formulation - Time of sample from last dose - Measured drug concentration - Pharmacodynamic data (desired effects and side-effects) • Data analysed using NONMEM

Pharmacodynamic monitoring in children Diclofenac: • Pain relief scale administered at blood sampling times - CRIES (neonatal pain score) - CHEOPS (1 -3 year old) - FACES (3 - 7 years) - Visual analogue scale (7 years +) • Antiplatelet activity • Increased bleeding post surgery

Faces pain rating scale Explain to the person that each face is for a person who feels happy because he has no pain or sad because he has a lot of pain. Face 0 is very happy because he doesn’t hurt at all…. Face 5 hurts as much as you can imagine, although you don’t have to be crying to feel this bad. Wong and Baker (1988)
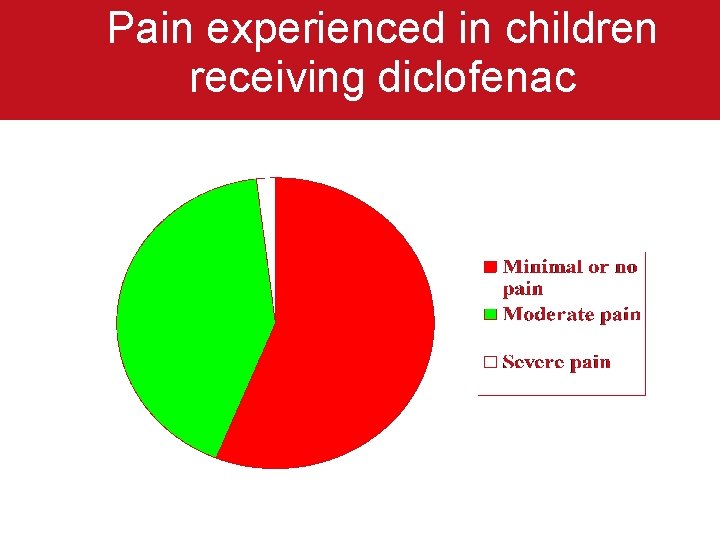
Pain experienced in children receiving diclofenac

Pharmacodynamic monitoring in children Enalapril: • Heart rate / BP • Respiratory rate • Liver size • Side-effects - cough - raised K+ - creatinine - urea

Drugs of interest to the group • • • Indometacin (patent ductus arteriosis)* Diclofenac (0 -12 y): post-op analgesia Enalapril (0 -12 y): hypertension / heart failure Cisapride (0 -12 y): GORD; stress ulceration Ranitidine (0 -7 y): stress ulceration* Omeprazole (0 -12 years): ICU stress ulceration Codeine (0 -1 y; rectal - all ages): post-op pain Spironolactone (0 -12 y): heart failure Midazolam (oral 0 -12 y; all routes 0 -7 y): day clinic procedures Spironolactone (neonates): chronic pulmonary dysplasia Metronidazole (neonates): necrotising enterocolitis*

Exemplars of kinetic analysis • Indometacin • Ranitidine • Metronidazole

Indometacin

Indometacin • • 35 neonates: 1 -6 doses of indometacin for patent ductus arteriosus Age: 25 -34 weeks gestational age (GA) 1 -77 days post natal age (PNA) • Weight: 0. 57 – 2. 19 kg • Dose: 0. 1 – 0. 2 mg / kg i. v. • 185 plasma concentrations of indometacin • Cp: 0. 01 – 2. 37 mg / litre • Closure of ductus in 75% of infants with Cp of >0. 4 mg / L 24 hours post last dose
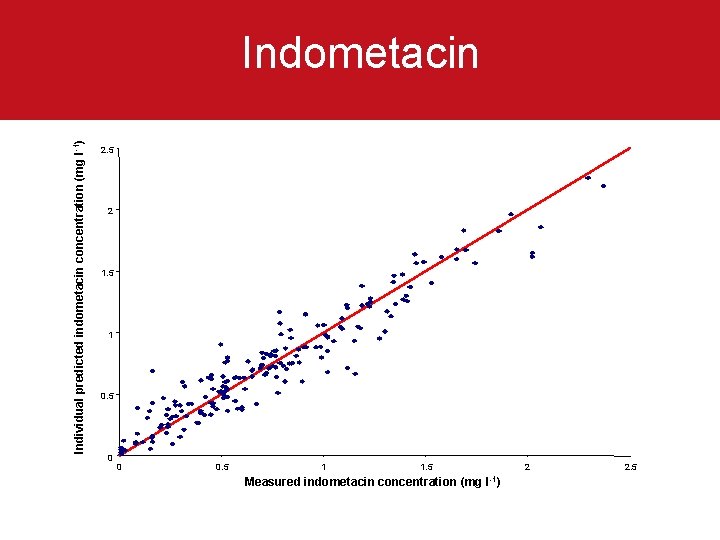
Individual predicted indometacin concentration (mg l -1) Indometacin 2. 5 2 1. 5 1 0. 5 0 0 0. 5 1 1. 5 Measured indometacin concentration (mg 2 l -1) 2. 5
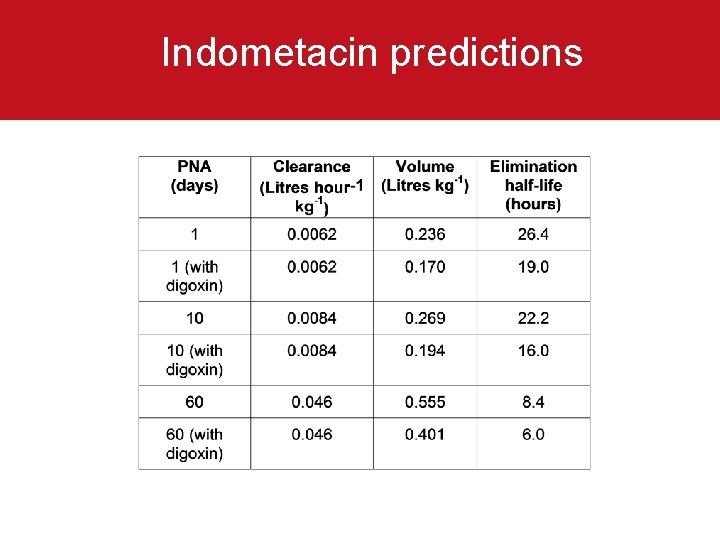
Indometacin predictions

Ranitidine

Ranitidine • • 60 children Age: 1 month – 12 years Weight: 2. 5 – 45 kg Dose: 0. 77 – 2 mg / kg (iv) & oral /NG (0. 7 – 7. 6 mg / kg) • 152 plasma samples • Cp: 9 – 1250 ng / ml

Preliminary results for ranitidine • Mean pharmacokinetic parameter estimates - CL -V - ka 0. 68 Litres / hour / kg 3. 31 Litres / kg 0. 33 / hour • CL and V linearly related to weight • Ka and F lower than reported adult values; CL within normal adult range, V and t 1/2 are larger

Metronidazole

Dried Blood Spots • Dried Blood Spots (DBS) - Guthrie Cards (quality controlled absorbent paper) – Inborn errors of metabolism – Other applications – e. g. antimalarials at bush clinics; drugs in sport – Applications in drug level determinations in paediatric patients – e. g. Oliveira et al. (2002); Millership et al. (2003)


DBS methodology • Blood spots from a heel prick or indwelling catheter are spotted onto a “Guthrie Card” and the spots are allowed to dry • Once dried the cards are placed in a wallet and transported to the lab for analysis • Drug concentrations in DBS determined by HPLC (with range of detection systems e. g. UV, fluorescence and MS/MS)

Advantages of the DBS approach • Consent may be easier since the volume of blood required is much smaller (particularly in neonates) • Purposive sampling throughout dosing interval will be facilitated if patient is not catheterised • Can use technique to collect samples at outpatient clinics and in patients’ homes (since venipuncture not required) • Transportation facilitated since drugs and metabolites more stable in dried form
Diclofenac (HPLC in DBS using UV detection)
Necrotising enterocolitis (NEC) and metronidazole • Analysis of metronidazole in DBS

Metronidazole in neonates • Initial pharmacokinetic study of 6 patients • Prescribed 7. 5 mg/kg IV metronidazole tds • Bloodspot samples taken on Guthrie cards at random times- 2 per dose interval • 44 bloodspots collected analysed • NONMEM used to calculate population and individual CL and Vd
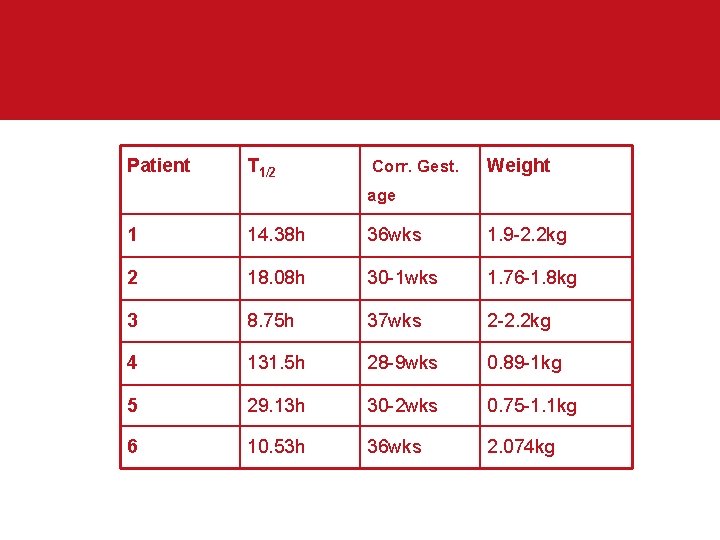
Patient T 1/2 Corr. Gest. Weight age 1 14. 38 h 36 wks 1. 9 -2. 2 kg 2 18. 08 h 30 -1 wks 1. 76 -1. 8 kg 3 8. 75 h 37 wks 2 -2. 2 kg 4 131. 5 h 28 -9 wks 0. 89 -1 kg 5 29. 13 h 30 -2 wks 0. 75 -1. 1 kg 6 10. 53 h 36 wks 2. 074 kg

Metronidazole in neonates In the final preliminary model • Clearance – Related to corrected gestational age, vancomycin therapy and weight 0. 75 • Volume of distribution – Related to frusemide therapy • Need more patients to provide a better assessment of covariate effects

The future

Future developments • Laboratory facilities upgraded to GCP standards (including LCMSMS) • New funding achieved from HPSS R&D Office and Department of Trade and Industry (KTP) (developmental work funded by Action Research) • Extend the number of centres involved in studies and become LRN within UK Medicines for Children Research Network (UKCRN) • Increasingly use of dried blood spots (DBSs) rather than plasma samples (incl. primary care) • Add a pharmacogenetics aspect to ongoing research – Children’s Mercy Hospital and Clinics, Kansas City, USA (Greg Kearns) – Keio University, Tokyo, Japan (Ken Kosaki)

Pharmacogenetic analyses • Diclofenac (USA) • Omeprazole (Japan)

Metabolism of diclofenac • Substantial first pass effect (50%) • CYP 2 C 9 - Major Metabolite 4’-hydroxydiclofenac - Minor Metabolites 3’-hydroxydiclofenac 3’-hydroxy-4’-methoxydiclofenac • CYP 3 A 4 - 5 -hydroxydiclofenac

CYP 2 C 9 polymorphism • CYP 2 C 9*1 wild type, homozygous CYP 2 C 9*2 CYP 2 C 9*3 8 -12% 3 -8% Caucasian 0% 2 -3% Oriental 1 -4% 0. 5 -2% Black Conflicting reports that CYP 2 C 9*3 is associated with decreased metabolic activity • Metabolism of diclofenac dependent on genetic makeup

Omeprazole • Metabolism - CYP 2 C 19 5 -hydroxyomeprazole • Poly morphism - CYP 2 C 19*1 (wild type) Extensive metabolisers - CYP 2 C 19*2 CYP 2 C 19*6 Poor metabolisers (2 -5% Caucasians, 13 -23% Orientals)

Conclusions Much more work required, however, using innovative research methodologies, and via government support to industry and research networks, the next 5 -10 years will see a major increase in evidence available for the safe and effective use of medicines in children.

Off-label and unlicensed medicines in children – a population approach to evidence based treatments James C. Mc. Elnay School of Pharmacy Queen’s University Belfast


- Slides: 48