OFFICE OF THE ASSISTANT SECRETARY FOR HEALTH Research














- Slides: 29

OFFICE OF THE ASSISTANT SECRETARY FOR HEALTH Research Misconduct: Are You Ready To Respond? Ranjini Ambalavanar, Ph. D. Division of Investigative Oversight (DIO) Office of Research Integrity (ORI) November 2019

O F F I C E O F T H E ASSISTANT SECRETARY FOR HEALTH 2

Applying for a Grant? You submit an NIH grant application which was written by you with the help of the laboratory members. You were not aware of the falsified/fabricated data and/or plagiarized text. Are you liable for research misconduct? Decisions by an ALJ on two of the recent ORI cases established that a PI and/or corresponding author, can be liable for research misconduct even if he/she was completely unaware of any falsification or plagiarism. O F F I C E O F T H E ASSISTANT SECRETARY FOR HEALTH 3

Research misconduct - 42 CFR § 93. 103 Fabrication, falsification, or plagiarism in proposing, performing, or reviewing research, or in reporting research results. PHS Policies on Research Misconduct (a)Fabrication is making up data or results and recording or reporting them. (b) Falsification is manipulating research materials, equipment, or processes, or changing or omitting data or results such that the research is not accurately represented in the research record. (c) Plagiarism is the appropriation of another person's ideas, processes, results, or words without giving appropriate credit. O F F I C E O F T H E ASSISTANT SECRETARY FOR HEALTH 4

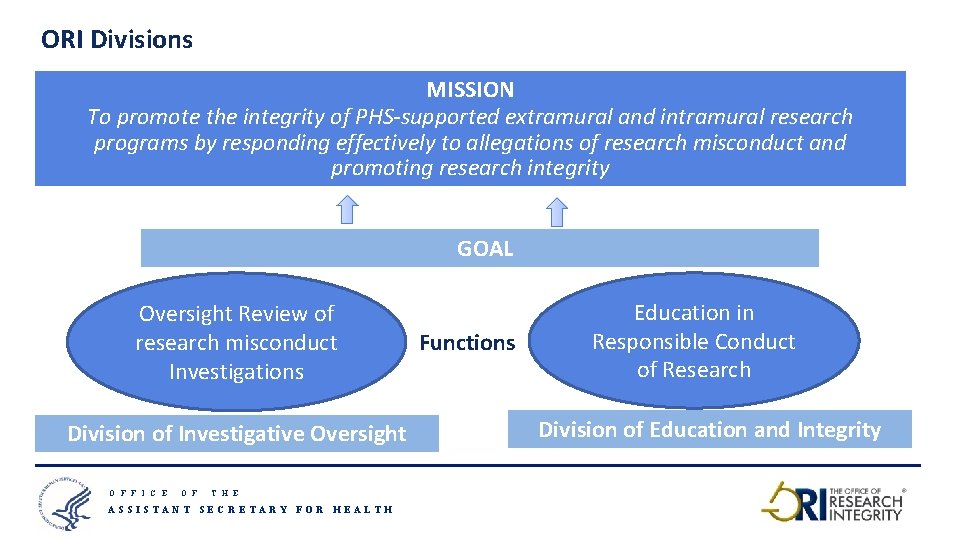
ORI Divisions MISSION To promote the integrity of PHS-supported extramural and intramural research programs by responding effectively to allegations of research misconduct and promoting research integrity GOAL Oversight Review of research misconduct Investigations Division of Investigative Oversight O F F I C E O F Functions Education in Responsible Conduct of Research Division of Education and Integrity T H E ASSISTANT SECRETARY FOR HEALTH 5

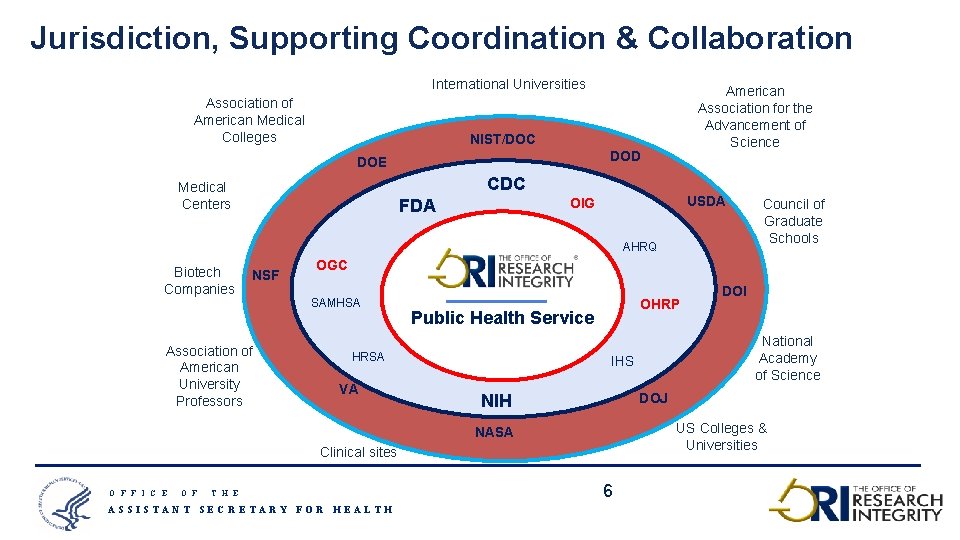
Jurisdiction, Supporting Coordination & Collaboration International Universities Association of American Medical Colleges American Association for the Advancement of Science NIST/DOC DOD DOE CDC Medical Centers USDA OIG FDA AHRQ Biotech Companies NSF Association of American University Professors OGC SAMHSA Public Health Service HRSA VA Clinical sites O F T H E ASSISTANT SECRETARY FOR HEALTH OHRP DOI National Academy of Science IHS DOJ NIH US Colleges & Universities NASA O F F I C E Council of Graduate Schools 6 6

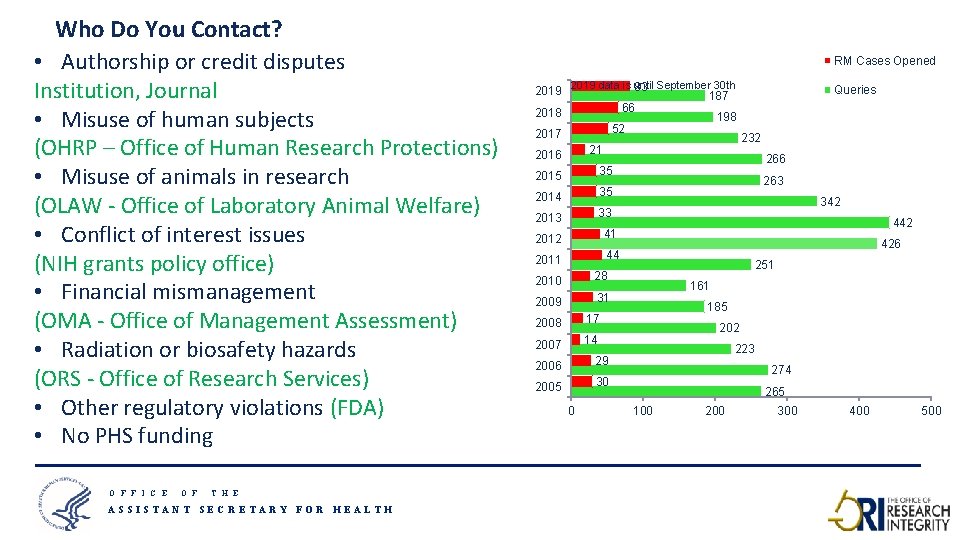
Who Do You Contact? • Authorship or credit disputes Institution, Journal • Misuse of human subjects (OHRP – Office of Human Research Protections) • Misuse of animals in research (OLAW - Office of Laboratory Animal Welfare) • Conflict of interest issues (NIH grants policy office) • Financial mismanagement (OMA - Office of Management Assessment) • Radiation or biosafety hazards (ORS - Office of Research Services) • Other regulatory violations (FDA) • No PHS funding O F F I C E O F RM Cases Opened until September 30 th 2019 data is 83 187 66 2018 198 52 2017 232 21 2016 2015 35 2014 35 2013 33 2012 41 2011 44 2010 28 2009 31 2008 17 2007 14 2006 29 2005 30 0 Queries 266 263 342 426 251 161 185 202 223 274 265 100 200 300 400 500 T H E ASSISTANT SECRETARY FOR HEALTH 7

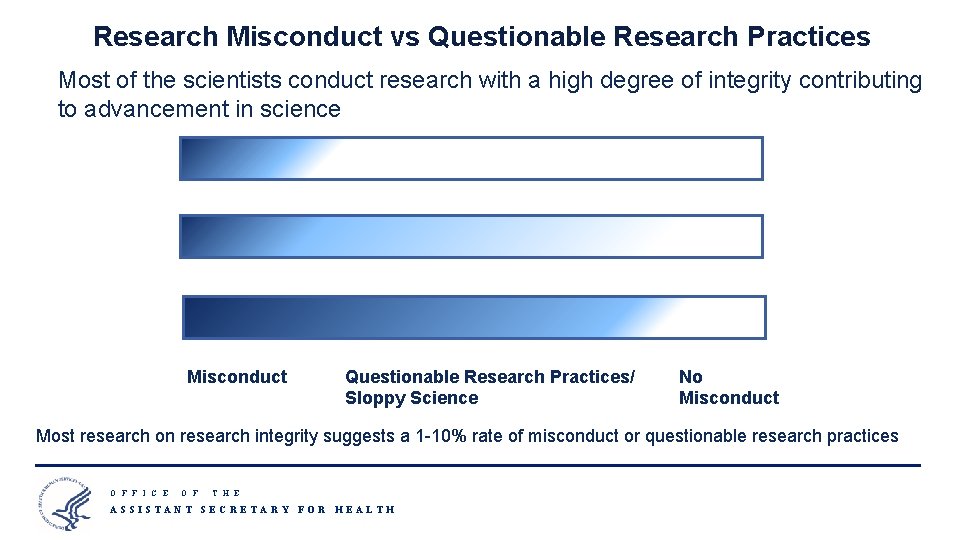
Research Misconduct vs Questionable Research Practices Most of the scientists conduct research with a high degree of integrity contributing to advancement in science Misconduct Questionable Research Practices/ Sloppy Science No Misconduct Most research on research integrity suggests a 1 -10% rate of misconduct or questionable research practices O F F I C E O F T H E ASSISTANT SECRETARY FOR HEALTH 8

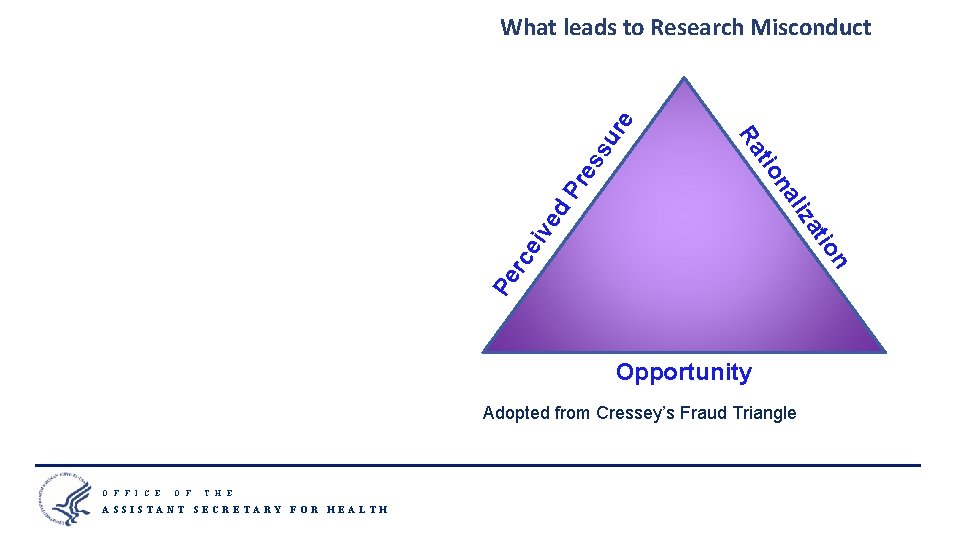
P re Pe n rc eiv io at ed liz na tio Ra ss ur e What leads to Research Misconduct Opportunity Adopted from Cressey’s Fraud Triangle O F F I C E O F T H E ASSISTANT SECRETARY FOR HEALTH 9

Steps in Research Misconduct Proceedings Allegation Assessment Inquiry 60 days From: • Within institution • Colleagues • Peer reviewers • Coauthors • Journal editors 120 days Research Integrity Officer (RIO) • Initial assessment • Organizes the remaining institutional processes To: • Institution: –Supervisor, Chair –RIO • Funding agency • Journal • ORI O F F I C E Investigation All ORI Findings are published in The Federal Register, the ORI website and newsletter, and the NIH website O F ORI oversight Additional evidence: • Interviews • Expert opinion • Forensic analysis • Additional Issues • Agree, or • Insufficient evidence for ORI finding Recommend Administrative Actions based on: • Scope of the misconduct • Aggravating /mitigating factors Actions Include: • Fix research record • Require special certification(s) • Suspend/terminate PHS-funding • Supervise offender(s) • Prohibit PHS-advisory role • Debar from future funding T H E ASSISTANT SECRETARY FOR HEALTH 10

Respondents’ statements…… O F F I C E O F T H E ASSISTANT SECRETARY FOR HEALTH 11

Some Red Flags O F F I C E O F • Inadequate supervision, guidance or training • PI not present in the laboratory • Excessive work-load • Demanding desired results to meet a deadline • Use of threats and intimidation as tactics to obtain desired results • No review of raw data • Sloppy research records • No guidance or standards for keeping data • Not protected over time (conveniently lost) • Accessible to multiple staff • Not backed up and stored securely • Researchers take data from the institution T H E ASSISTANT SECRETARY FOR HEALTH 12

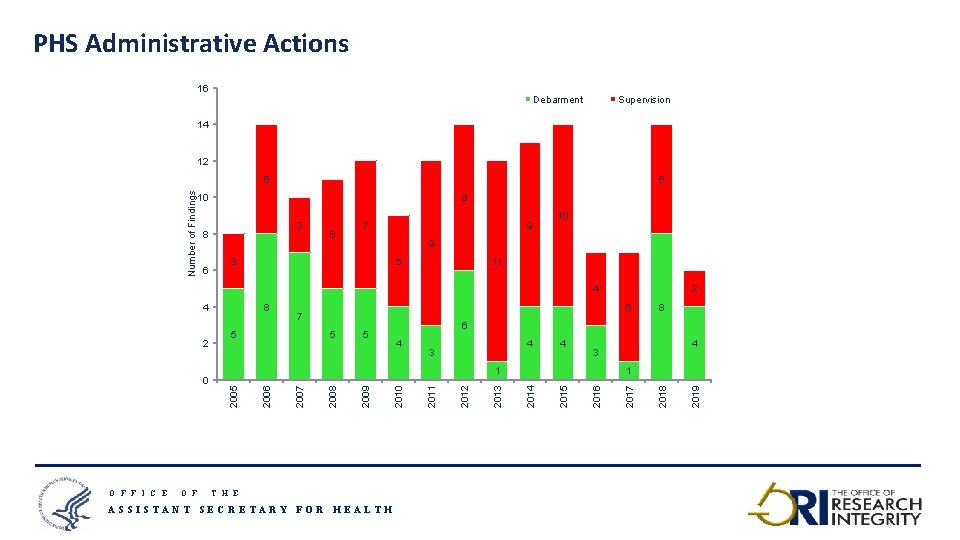
PHS Administrative Actions 16 Debarment Supervision 14 12 6 10 8 3 8 6 6 7 9 10 9 3 5 11 4 O F F I C E O F 5 6 4 4 3 4 2016 2015 1 2014 2013 2012 2011 2010 2009 2008 4 3 1 2007 8 2018 5 2006 0 6 7 5 2005 2 8 2017 4 2 2019 Number of Findings 6 T H E ASSISTANT SECRETARY FOR HEALTH 13

Case Summaries and NIH e. RA Links to PHS Administrative Action Bulletin Board O F F I C E O F T H E ASSISTANT SECRETARY FOR HEALTH 14

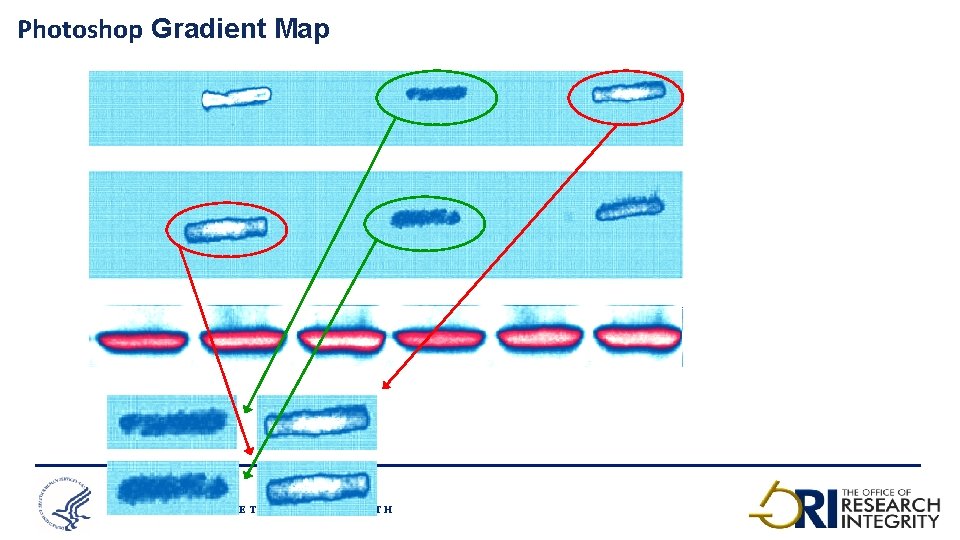
Photoshop Gradient Map O F F I C E O F T H E ASSISTANT SECRETARY FOR HEALTH 15

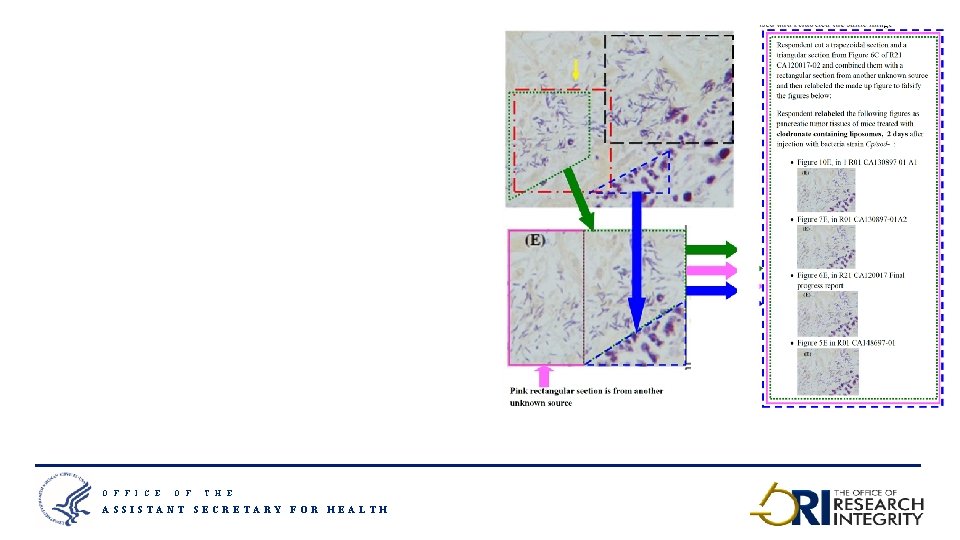
O F F I C E O F T H E ASSISTANT SECRETARY FOR HEALTH 16

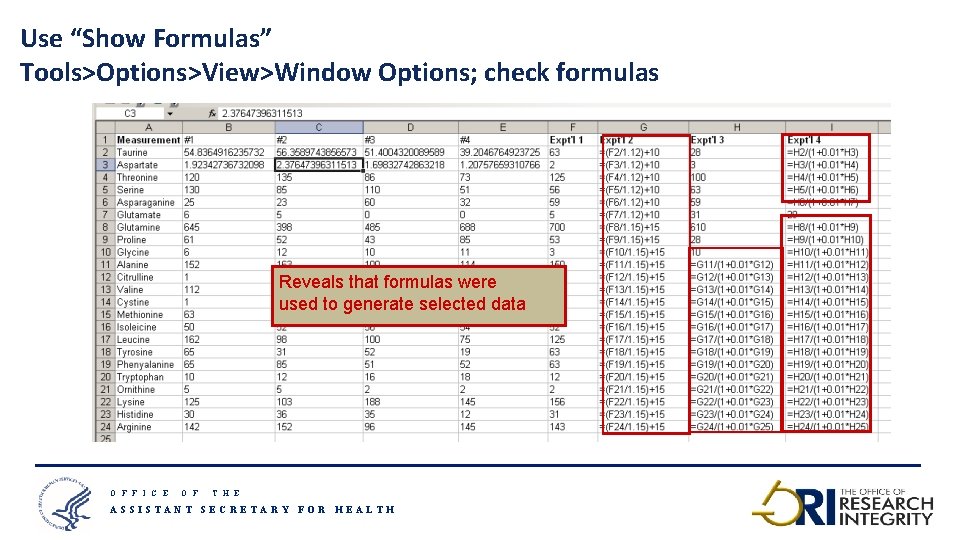
Use “Show Formulas” Tools>Options>View>Window Options; check formulas Reveals that formulas were used to generate selected data O F F I C E O F T H E ASSISTANT SECRETARY FOR HEALTH 17

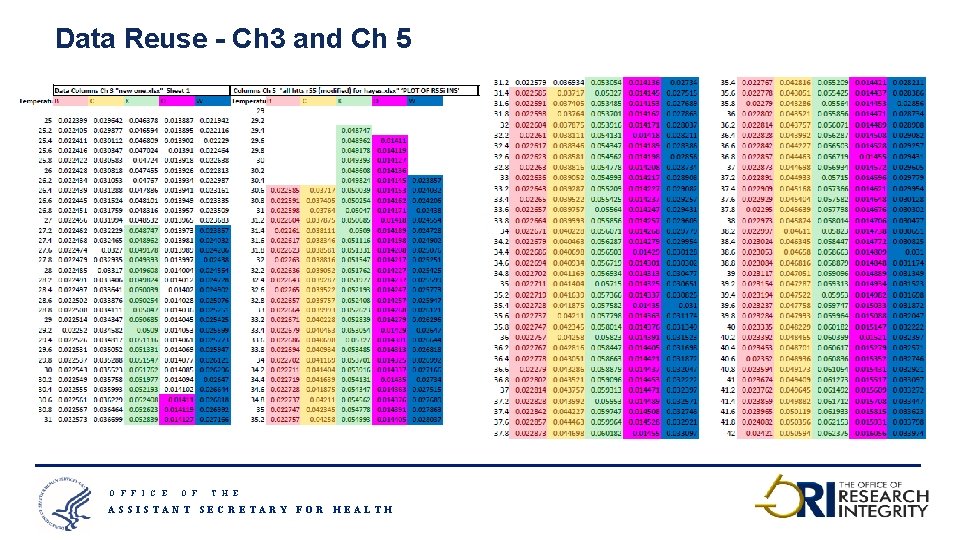
Data Reuse - Ch 3 and Ch 5 O F F I C E O F T H E ASSISTANT SECRETARY FOR HEALTH 18

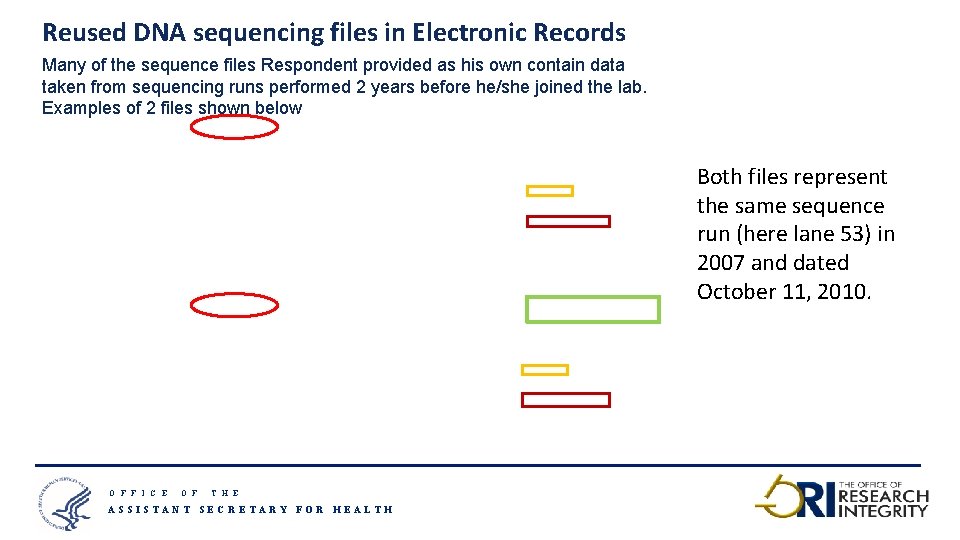
Reused DNA sequencing files in Electronic Records Many of the sequence files Respondent provided as his own contain data taken from sequencing runs performed 2 years before he/she joined the lab. Examples of 2 files shown below Both files represent the same sequence run (here lane 53) in 2007 and dated October 11, 2010. O F F I C E O F T H E ASSISTANT SECRETARY FOR HEALTH 19

Clinical Research Data Interviews Entry criteria Screening logs Approval forms Follow-up exams/data Consent forms Test scores Laboratory results Patient data Number of subjects Dates of procedures Protocol Study results O F F I C E O F Respondents in 68 cases with finding Research Asst. /Assoc. Research Coordinator/Nurse Practitioner Technician Students Fellows All Professors Other T H E ASSISTANT SECRETARY FOR HEALTH 20

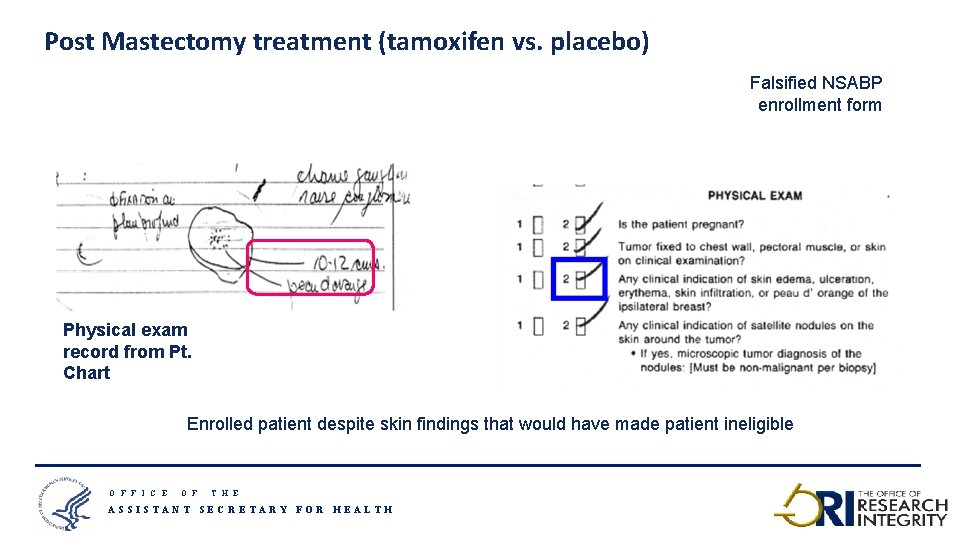
Post Mastectomy treatment (tamoxifen vs. placebo) Falsified NSABP enrollment form Physical exam record from Pt. Chart Enrolled patient despite skin findings that would have made patient ineligible O F F I C E O F T H E ASSISTANT SECRETARY FOR HEALTH 21

Follow up Visits For Patient 10: dated “ 01 -18 -88” O F F I C E O F T H E ASSISTANT SECRETARY FOR HEALTH 22

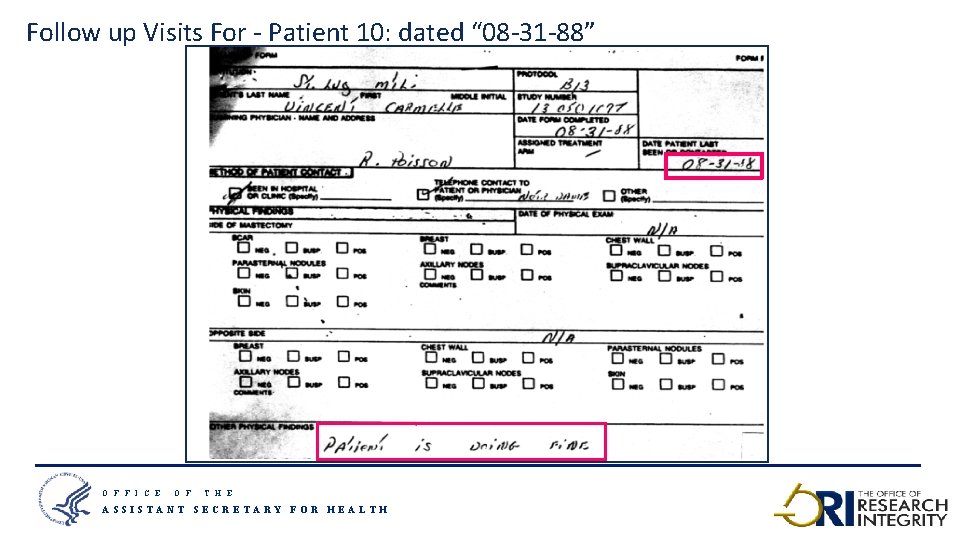
Follow up Visits For - Patient 10: dated “ 08 -31 -88” O F F I C E O F T H E ASSISTANT SECRETARY FOR HEALTH 23

Follow up Visits For - Patient 10: dated “ 11 -29 -88” O F F I C E O F T H E ASSISTANT SECRETARY FOR HEALTH 24

Follow up Visits For - Patient 10: dated “ 03 -21 -89” O F F I C E O F T H E ASSISTANT SECRETARY FOR HEALTH 25

Follow up Visits For - Patient 10: dated “ 02 -02 -90” O F F I C E O F T H E ASSISTANT SECRETARY FOR HEALTH 26

Patient 10: Death Certificate “September 29, 1987” 28 months prior to last reported follow-up (2 -2 -90) 4 months prior to first shown (1 -18 -88) follow up O F F I C E O F T H E ASSISTANT SECRETARY FOR HEALTH 27

Pressures are Perpetuated at Every Level. What can You Do? O F F I C E O F T H E ASSISTANT SECRETARY FOR HEALTH 28

O F F I C E O F T H E ASSISTANT SECRETARY FOR HEALTH 29