ODAC Temozolomide Oncology Drug Advisory Committee March 13




















- Slides: 21

ODAC Temozolomide Oncology Drug Advisory Committee March 13, 2003 Craig L. Tendler, M. D. Vice President, Oncology Clinical Research 1 SCHERING-PLOUGH RESEARCH INSTITUTE

ODAC Agenda • Basis for the accelerated approval of Temozolomide in refractory Anaplastic Astrocytoma (94 -123) • Post-approval commitment study (98 -13) – Design – Key study milestones – Current status • Ongoing challenges associated with post-approval commitment study • Initiatives to expedite completion of post approval commitment • Schering-Plough development programs with temozolomide in primary brain tumors 2 SCHERING-PLOUGH RESEARCH INSTITUTE

ODAC Temozolomide NDA Submission for Recurrent Glioma (at first relapse) • Recurrent Glioblastoma Multiforme – Phase 2 randomized study temozolomide vs procarbazine (94 -091) – Phase 2 single arm study (94 -122) • Recurrent Anaplastic Astrocytoma – Phase 2 single arm study Anaplastic Astrocytoma 123) 3 (94 - SCHERING-PLOUGH RESEARCH INSTITUTE

ODAC Temozolomide Indication (August 1999) Adult patients with refractory anaplastic astrocytoma, i. e. patients at first relapse who have experienced disease progression on a regimen containing a nitrosourea and procarbazine 4 SCHERING-PLOUGH RESEARCH INSTITUTE

ODAC Basis for Temozolomide Accelerated Approval for Refractory Anaplastic Astrocytoma • Study Design (94 -123) – single arm study (n=162) – adult anaplastic astrocytoma patients at first relapse • ≤ 1 prior chemotherapy regimen • Study conduct – 32 institutions – February 1995 – April 1998 5 SCHERING-PLOUGH RESEARCH INSTITUTE

ODAC Basis for Temozolomide Accelerated Approval for Refractory Anaplastic Astrocytoma • Primary endpoint – PFS at 6 months assessed by Gd MRI (confirmed by central review) • Secondary endpoints – response rate – overall survival • Objective – to show 6 month PFS rate lower boundary of 95% CI of at least 10% 6 SCHERING-PLOUGH RESEARCH INSTITUTE

ODAC Basis for Temozolomide Accelerated Approval for Refractory Anaplastic Astrocytoma • Overall results: ITT Population (94 -123) 7 – PFS 6 Months: 51% (95% CI: 43 -59%) – Median Survival: 13. 6 months – Response Rate: 33% (CR + PR: 5%+28%) SCHERING-PLOUGH RESEARCH INSTITUTE

ODAC Basis for Temozolomide Accelerated Approval for Refractory Anaplastic Astrocytoma • Results: Patients Refractory to Procarbazine Plus a Nitrosourea (n = 54) 8 – Objective Response Rate: 22% (CR = 9%) – Median Duration of Response: 50 weeks (16 -114 weeks) – Duration of Response in Complete Responders: 52 -114 weeks – Median Survival: 15. 9 months SCHERING-PLOUGH RESEARCH INSTITUTE

ODAC Temozolomide Safety in Malignant Glioma • Safety database: 1, 017 temozolomide treated patients (400 GBM & AA) • Temozolomide was administered with few dose reductions or dose delays • Most adverse events were mild to moderate in severity • Study treatment discontinuation due to adverse events was infrequent • Grade 3/4 myelosuppression was also infrequent and non-cumulative 9 SCHERING-PLOUGH RESEARCH INSTITUTE

ODAC Unanimous ODAC Opinion • Patients with relapsed anaplastic astrocytoma after procarbazine and a nitrosourea are considered unresponsive to otherapies – Unmet medical need • Objective response in this setting could be an adequate surrogate for clinical benefit if well defined and of sufficient magnitude 10 SCHERING-PLOUGH RESEARCH INSTITUTE

ODAC Unanimous ODAC Opinion • Efficacy: – Temozolomide is effective for the treatment of anaplastic astrocytoma in patients previously treated with a nitrosourea and procarbazine • Safety: – Safety of Temozolomide is acceptable for this indication 11 SCHERING-PLOUGH RESEARCH INSTITUTE

ODAC Temozolomide Anaplastic Astrocytoma Post Approval Commitment • Planning for first-line study with RTOG already underway at time of the ODAC accelerated approval recommendation • Design (98 -13) – Population: First-line anaplastic astrocytoma – Primary endpoint: Overall Survival RT + TMZ Randomization RT + BCNU RT + TMZ/BCNU • Rationale for TMZ/BCNU Combination – AGAT depletion 12 SCHERING-PLOUGH RESEARCH INSTITUTE

ODAC FDA Agreed Commitments Commitment Date Status • Randomized phase III (98 -13) to be preceded by additional safety assessment of TMZ/BCNU in the study population June ’ 01 Submitted July ‘ 01 • Submission of pediatric study reports -I 93 -125 -H 97 -017 Dec. ‘ 02 Submitted Sept. ‘ 02 • Submission of final study report (Anaplastic Astrocytoma first-line study 98 -13) June ‘ 07 Project ongoing 13 SCHERING-PLOUGH RESEARCH INSTITUTE

ODAC Temozolomide Anaplastic Astrocytoma Post Approval Commitment: Key Milestones • 6/99 Protocol first submitted to FDA • 8/99 Accelerated approval granted • 10/99 Revised protocol incorporating FDA comments submitted to FDA • 12/99 FDA indicated that the protocol should not proceed until additional safety data on the TMZ + BCNU in the study population was submitted • 2/00 Agreement on design of phase I safety assessment • 4/00 IND filed by RTOG 14 SCHERING-PLOUGH RESEARCH INSTITUTE

ODAC Key Milestones - Continued • 6/00 Initiation of Phase I safety assessment with TMZ/BCNU • 3/01 Completion of enrollment for safety assessment • 7/01 Submission of safety data to FDA • 9/01 Enrollment initiated for second safety cohort • 1/02 Completion of enrollment for 2 nd safety cohort • 6/02 Combination arm discontinued due to safety profile • 1/03 Randomized Phase 3 opened 15 SCHERING-PLOUGH RESEARCH INSTITUTE

ODAC RTOG 98 -13: Status March ‘ 03 RT + TMZ (HR target 1. 5; median OS 36 vs. 54 months) • Study design RT + BCNU • Phase 3 opened to enrollment Jan. ‘ 03 • Projected enrollment 24 patients/month for total of 454 patients • Inter-group study infrastructure provides broadest access to U. S. and Canadian investigators – RTOG, ECOG, SWOG, NCCTG (>300 sites) 16 SCHERING-PLOUGH RESEARCH INSTITUTE

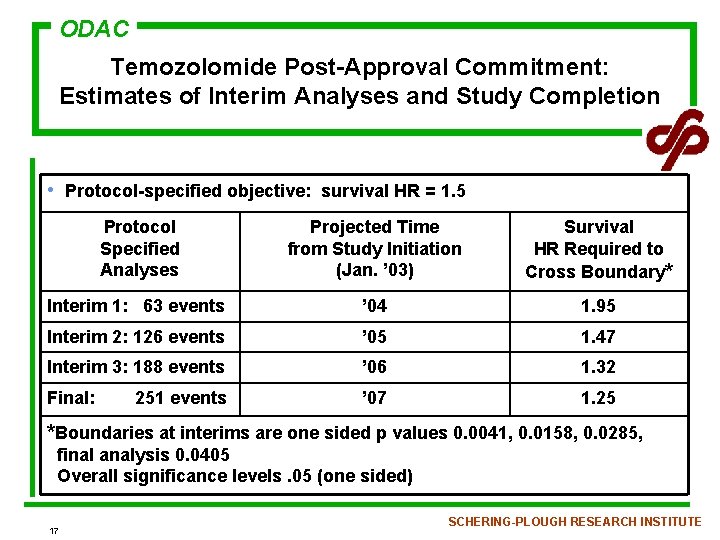
ODAC Temozolomide Post-Approval Commitment: Estimates of Interim Analyses and Study Completion • Protocol-specified objective: survival HR = 1. 5 Protocol Specified Analyses Projected Time from Study Initiation (Jan. ’ 03) Survival HR Required to Cross Boundary* Interim 1: 63 events ’ 04 1. 95 Interim 2: 126 events ’ 05 1. 47 Interim 3: 188 events ’ 06 1. 32 Final: ’ 07 1. 25 251 events *Boundaries at interims are one sided p values 0. 0041, 0. 0158, 0. 0285, final analysis 0. 0405 Overall significance levels. 05 (one sided) 17 SCHERING-PLOUGH RESEARCH INSTITUTE

ODAC Challenges of Survival Trial in First. Line Anaplastic Astrocytoma • Low and declining annual incidence (~3, 000 newly diagnosed U. S. patients/year) • Long median survival (3 -4 years) 18 SCHERING-PLOUGH RESEARCH INSTITUTE

ODAC Schering-Plough and RTOG Initiatives to Expedite Study Completion • Communications: – Investigators: investigator meetings, target neuro-surgeons, monthly teleconference with the lead investigators at each cooperative group, cooperative group newsletters – Patients: internet listing, patient brochures, patient brain tumor support groups (National Brain Tumor Foundation and American Brain Tumor Foundation) • Project Management: – RTOG HQ staff – Monthly progress reviews (SPRI / Inter-group PIs) – Institutional data management support • International Sites 19 SCHERING-PLOUGH RESEARCH INSTITUTE

ODAC Temozolomide Additional Initiatives in Malignant Gliomas • SPRI-supported EORTC/NCIC phase 3 study of Temozolomide plus radiation versus radiation in newly diagnosed Glioblastoma Multiforme • Enrollment completed March ‘ 02 (573 patients) • Primary endpoint is overall survival 20 SCHERING-PLOUGH RESEARCH INSTITUTE

ODAC Temozolomide Development Programs in Primary Brain Cancer • Phase 3 Newly diagnosed Anaplastic Astrocytoma (98 -13) • Phase 3 Newly diagnosed Glioblastoma Multiforme • Phase 2 Anaplastic Oligodendroglioma-RTOG • Phase 3 Low Grade Glioma-EORTC • Phase 1/2 studies in recurrent pediatric CNS tumors 21 SCHERING-PLOUGH RESEARCH INSTITUTE