OCEANESS 410 Class 7 Mantle Melting and Phase








- Slides: 23

OCEAN/ESS 410 Class 7. Mantle Melting and Phase Diagrams William Wilcock

Lecture/Lab Learning Goals • Understand the basic concept of decompressional melting and know how its distribution in the mantle is related to plate tectonics. • Be able to define the terms liquidus, solidus, mantle adiabat and use them to explain mantle melting beneath mid-ocean ridges with a plot of depth (or pressure) against temperature. • Be familiar with the simple two phase diagrams and the lever rule • Know what a eutectic is and why it explains the relatively uniform composition of basalts in different tectonic settings (and on different planets). • Be able explain oceanic crustal thickness in terms of the degree of partial melting in the crust and be able to identify melting/solidification pathways on binary phase diagrams (LAB)

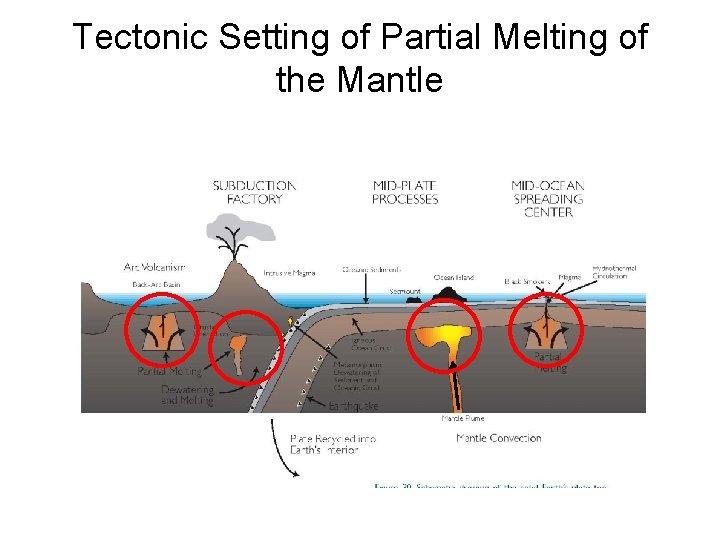
Tectonic Setting of Partial Melting of the Mantle

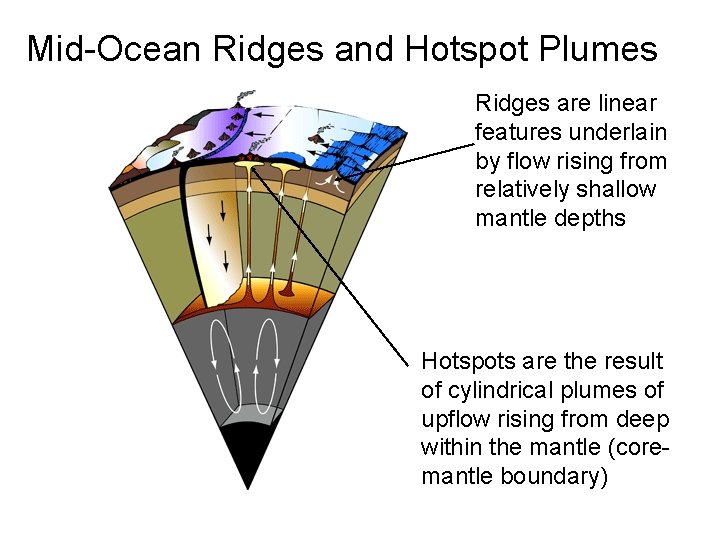
Mid-Ocean Ridges and Hotspot Plumes Ridges are linear features underlain by flow rising from relatively shallow mantle depths Hotspots are the result of cylindrical plumes of upflow rising from deep within the mantle (coremantle boundary)

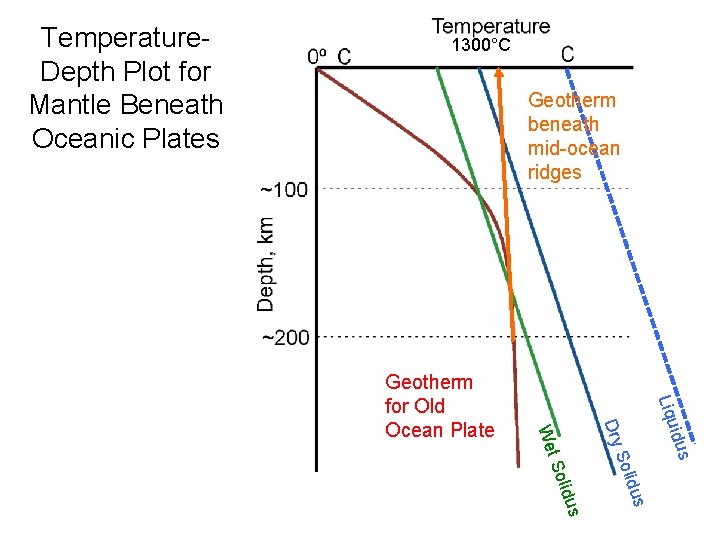
Temperature. Depth Plot for Mantle Beneath Oceanic Plates 1300°C Geotherm beneath mid-ocean ridges idus Liqu s lidu So Dry s lidu t So We Geotherm for Old Ocean Plate

Terminology Geotherm – Vertical temperature profile in the earth Adiabat – Temperature that a packet of the mantle that moves up/down without gaining or loosing heat Solidus – Temperature at which a rock will first start to melt Liquidus – Temperature at which a rock will be fully molten.

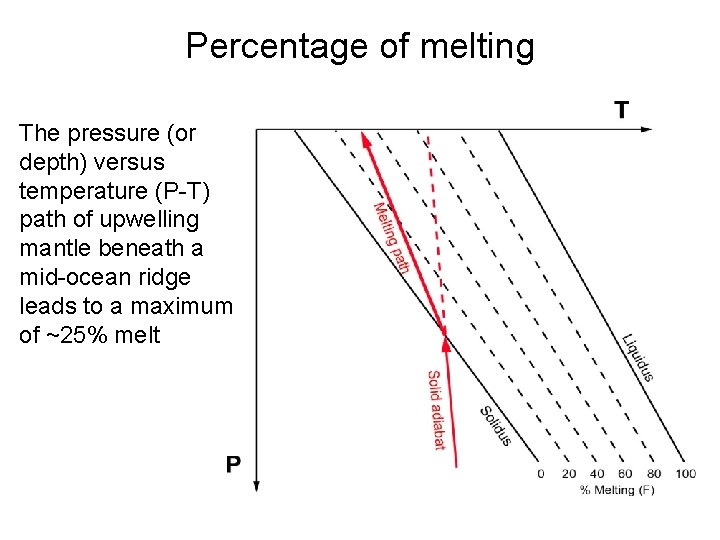
Percentage of melting The pressure (or depth) versus temperature (P-T) path of upwelling mantle beneath a mid-ocean ridge leads to a maximum of ~25% melt

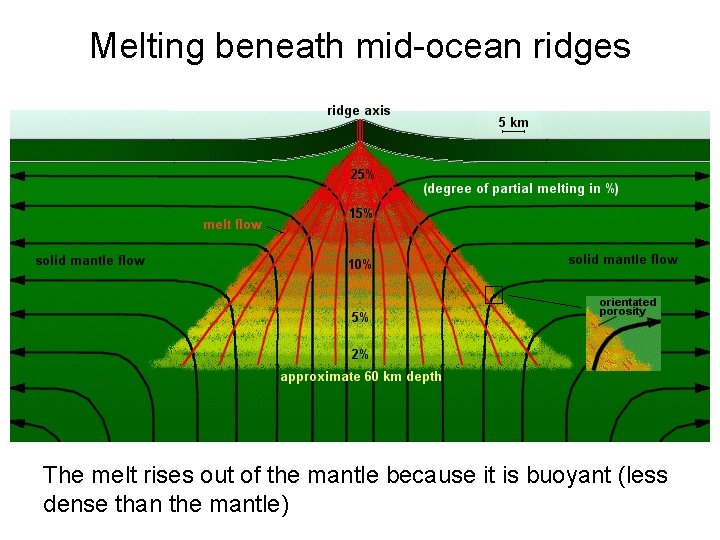
Melting beneath mid-ocean ridges The melt rises out of the mantle because it is buoyant (less dense than the mantle)

Na. Cl • Naturally occurring • Inorganic • Fixed chemical formula (or range of formulas) • Unique, orderly internal arrangement of atoms (crystalline)

Definitions • A system can be defined as that part of the universe that is arbitrarily or naturally isolated for the purposes of consideration or experimentation. For our purposes it will generally be a sample of a rock • A phase is defined as a chemically and physically homogeneous part of a system. Each mineral in a rock is a phase as is a melt. • Components are the minimum number of chemical constituents necessary to describe the system

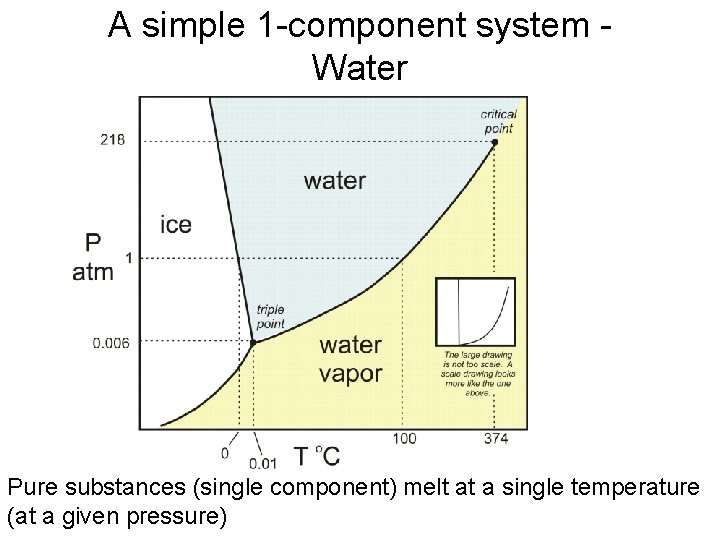
A simple 1 -component system - Water Pure substances (single component) melt at a single temperature (at a given pressure)

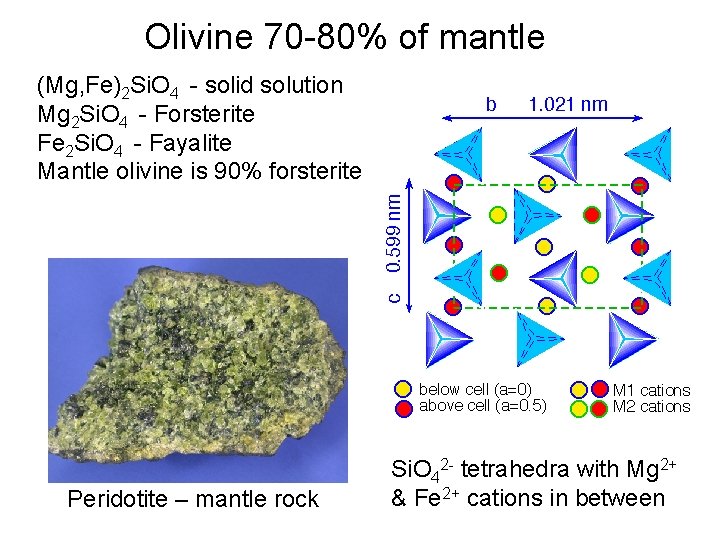
Olivine 70 -80% of mantle (Mg, Fe)2 Si. O 4 - solid solution Mg 2 Si. O 4 - Forsterite Fe 2 Si. O 4 - Fayalite Mantle olivine is 90% forsterite Peridotite – mantle rock Si. O 42 - tetrahedra with Mg 2+ & Fe 2+ cations in between

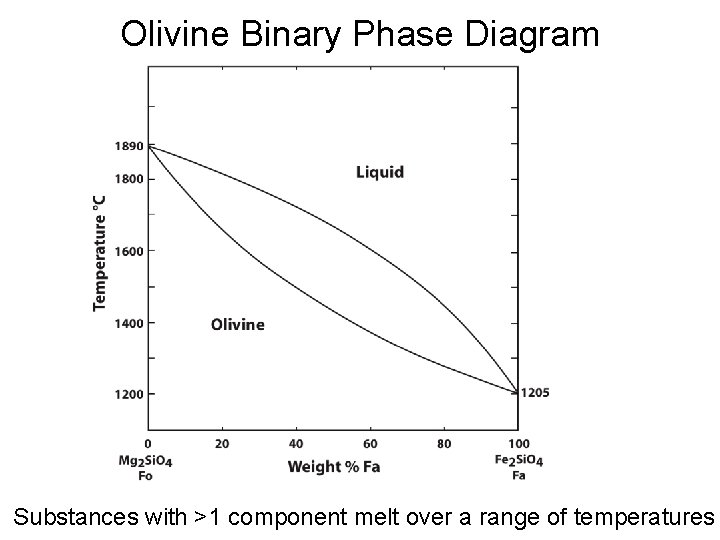
Olivine Binary Phase Diagram Substances with >1 component melt over a range of temperatures

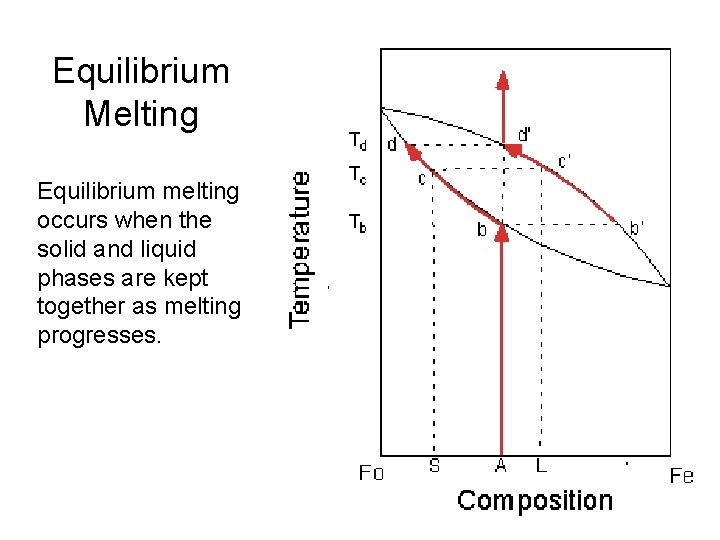
Equilibrium Melting Equilibrium melting occurs when the solid and liquid phases are kept together as melting progresses.

S – solid composition L – liquid composition A – system composition We can write fraction x of solid as x. S + (1 -x)L = A which can also be written as x (A –S) = (1 -x)(L-A) This is analogous to an old fashioned balance scale as shown in the figure We can solve the above equations to get the proportion of solid x = (A – L) / (S – L) Lever Rule x 1 -x

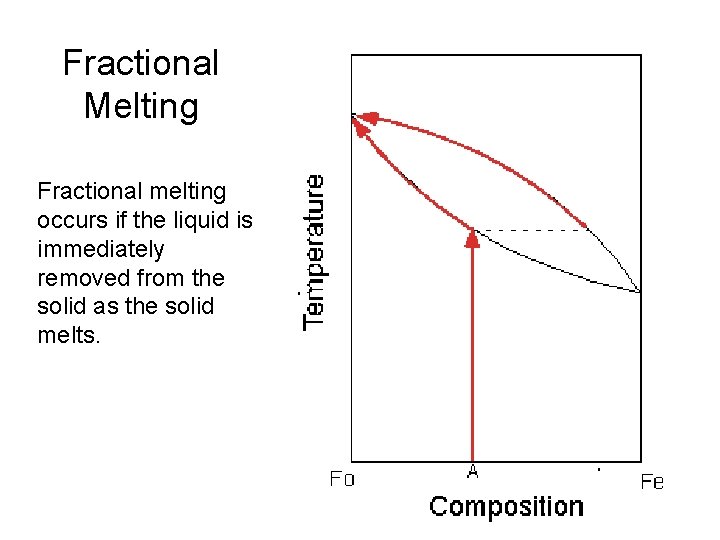
Fractional Melting Fractional melting occurs if the liquid is immediately removed from the solid as the solid melts.

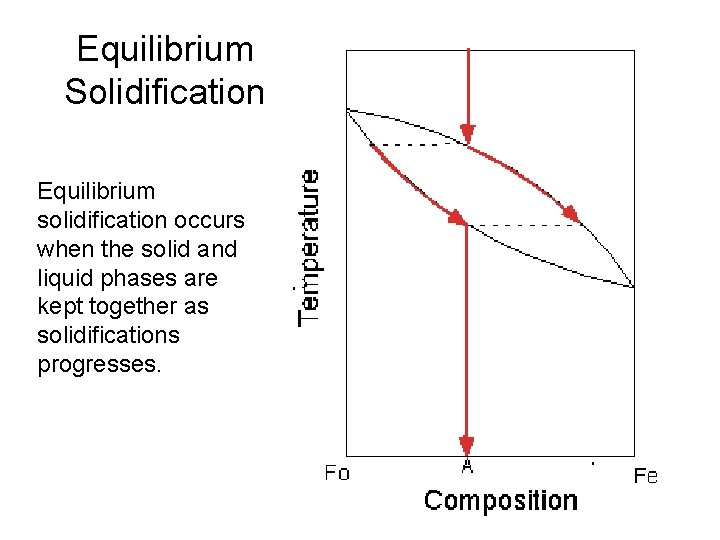
Equilibrium Solidification Equilibrium solidification occurs when the solid and liquid phases are kept together as solidifications progresses.

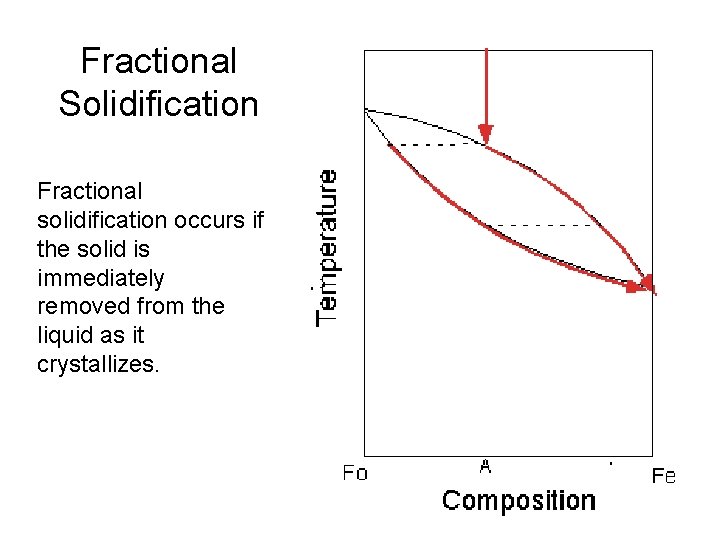
Fractional Solidification Fractional solidification occurs if the solid is immediately removed from the liquid as it crystallizes.

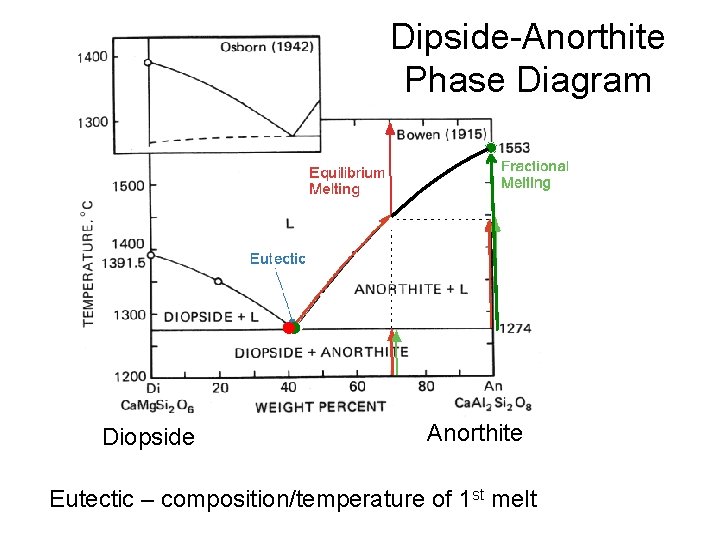
Diopside (Clinopyroxene) – Anorthite (Plagioclase) Diopside (Ca. Mg. Si 2 O 6) Dark mineral Gabbro (coarse grained equivalent of basalt) – Oceanic Crust Anorthite (Ca. Al 2 Si 2 O 8) Light mineral

Dipside-Anorthite Phase Diagram Diopside Anorthite Eutectic – composition/temperature of 1 st melt

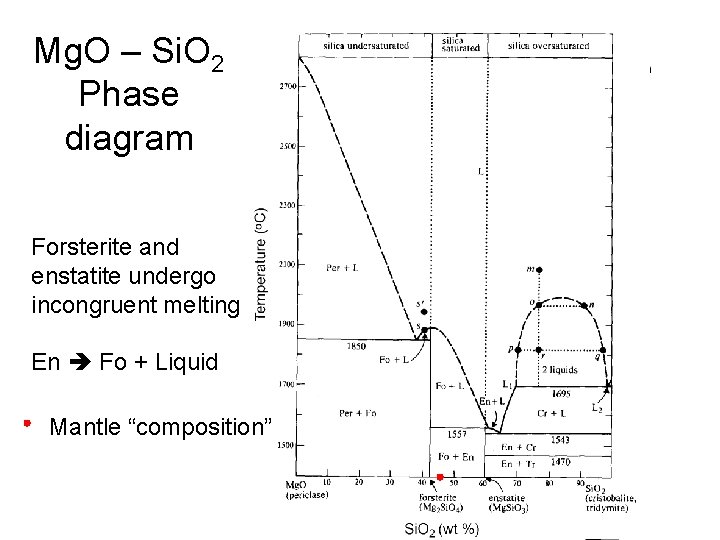
Enstatite (Bronzite, Orthopyroxene) Mg. Si. O 3 The second most common mineral in the mantle (20%)

Mg. O – Si. O 2 Phase diagram Forsterite and enstatite undergo incongruent melting En Fo + Liquid Mantle “composition”

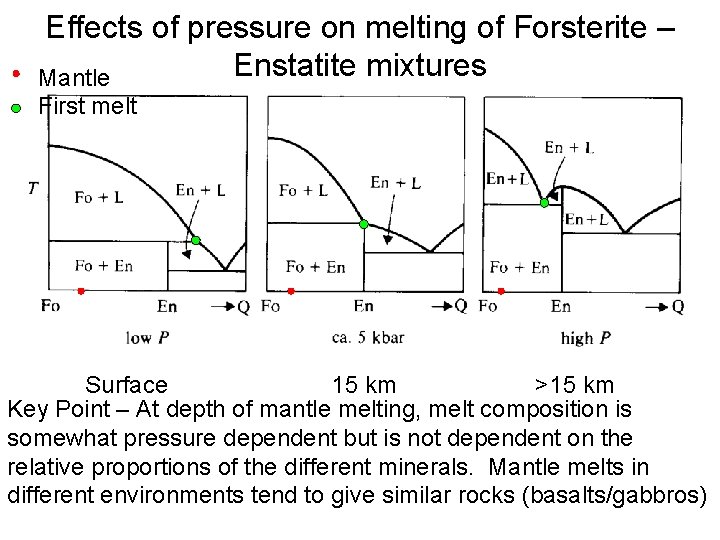
Effects of pressure on melting of Forsterite – Enstatite mixtures Mantle First melt 15 km Surface >15 km Key Point – At depth of mantle melting, melt composition is somewhat pressure dependent but is not dependent on the relative proportions of the different minerals. Mantle melts in different environments tend to give similar rocks (basalts/gabbros)