Ocean Chemistry and Composition Brian Schuster Chemical Properties

Ocean Chemistry and Composition Brian Schuster

Chemical Properties of Sea Water • polar: unequal sharing of electrons • hydrogen bonding: intermolecular dipole-dipole interaction between H 2 O molecules • cohesion: sticking of H 2 O molecules to each other resulting from hydrogen bonding; causes surface tension • adhesion: sticking of H 2 O molecules to other polar materials • capillarity: movement of water up a small tube • latent heat: energy stored in water that doesn’t change it’s temperature

Acidity/Alkalinity • • • ocean: p. H 7. 5 to 8. 5 due to carbonate (CO 3 -2) from dissociation of calcium carbonate freshwater: p. H 6. 5 to 7. 5 due to carbonic acid (H 2 CO 3) bicarbonate buffering, resistance to p. H change – – – H 2 O + CO 2 H 2 CO 3 (carbonic acid=weak acid) H 2 CO 3 H+ + HCO 3 - (bicarbonate=buffer) Ca. CO 3 Ca+2 + CO 3 -2 (carbonate=base)
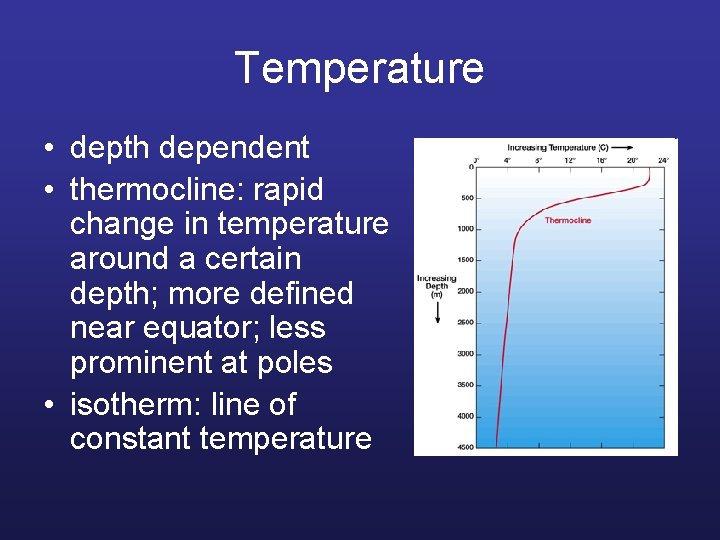
Temperature • depth dependent • thermocline: rapid change in temperature around a certain depth; more defined near equator; less prominent at poles • isotherm: line of constant temperature

Global isotherms display currents

Salinity • isohaline: line of constant salinity • residence time: average time a molecule spends in a certain reservoir • salinometers: determine salinity through conductivity • halocline: rapid change in salinity • constancy of composition: major ion constituents exist in constant proportions • water is the “universal solvent” • ocean salinity: 35 ppt, brackish: 17 ppt, brine: >50 ppt • saltiest sea: Dead Sea

Salinity (continued) • Long-term sources: – hydrothermal vents & volcanoes – weathering of rocks • cation: positively charged ion • anion: negatively charged ion • salinity effected by: – precipitation, evaporation – freezing, thawing – river input • Top six constituents – – – chlorine (Cl-) 55% sodium (Na+) 31% sulfate (SO 4 -2) 8% magnesium (Mg+2) 4% calcium (Ca+2) 1% potassium (K+) 1%

Desalination 1. 2. 3. 4. reverse osmosis freezing & thawing ion exchange distillation (evaporation & condensation)

Density • pycnocline: rapid change in density • freshwater most dense at 4 °C, saltwater most dense just before freezing • saltwater freezes at -2 °C (freezing point depression) • density affected by: – temperature – salinity – depth, pressure: minimally • isopycnal: surface of constant density

Pressure and Gases • rises by 1 atm (14. 7 psi, 101. 3 k. Pa) for every 10 m (33 ft) depth • absolute pressure at 20 m is 3 atm • gauge pressure at 20 m is 2 atm • isobar: line of constant pressure

Dissolved Gases • Henry’s Law: more gas can be dissolved under high pressure • more gas is dissolved in deep, cold water • oxygen minimum zone: depth depends on productivity and aerobic respiration • atmospheric gases resemble ocean’s due to mixing

Calcium Carbonate (Ca. CO 3) • used in shells, compound of limestone and calcite, base of coral reef • sources and sinks – chemical precipitation, dissolution – weathering of limestone and calcite – organisms • solubility increased by: – greater depths – higher acidity (lower p. H) • carbonate compensation depth (CCD): dissolution rate of Ca. CO 3 equals supply rate

Nutrients • eutrophication: excessive addition of nutrients • oligotrophication: excessive removal of nutrients • iron is the limiting nutrient for most algae South Atlantic phytoplankton bloom

Heat Transfer • conduction: molecules speed each other up by physically bumping • convection: carried by movement of fluid • radiation: electromagnetic radiation is absorbed and raises temperature • heat is conducted faster in water than in air, so divers can get cold faster

Sound • in water, speed of sound = ~1500 m/s (3500 mph), 5 x speed in air • speed changes with density • sound travels farther in water than light, so it is good for cetacean communication • SOFAR (sound fixing and ranging) channel: sound travels slower around 1000 m, and can go farther; sound gets stuck in this channel
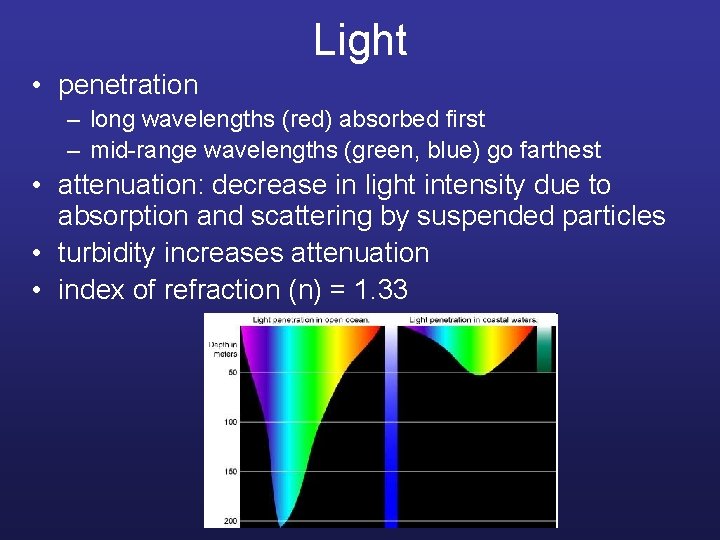
Light • penetration – long wavelengths (red) absorbed first – mid-range wavelengths (green, blue) go farthest • attenuation: decrease in light intensity due to absorption and scattering by suspended particles • turbidity increases attenuation • index of refraction (n) = 1. 33

Other • conservative property: mostly affected by mixing and diffusion (ex: salinity) • non-conservative property: affected mostly by processes other than mixing and diffusion (ex: dissolved oxygen relating to productivity) • temperature-salinity diagram: unique to different bodies of water; shows lines of constant density in sigma units
- Slides: 17