Ocean Acidification Lecture Hilary Palevsky palevskyuw edu Absorbing

Ocean Acidification Lecture Hilary Palevsky palevsky@uw. edu
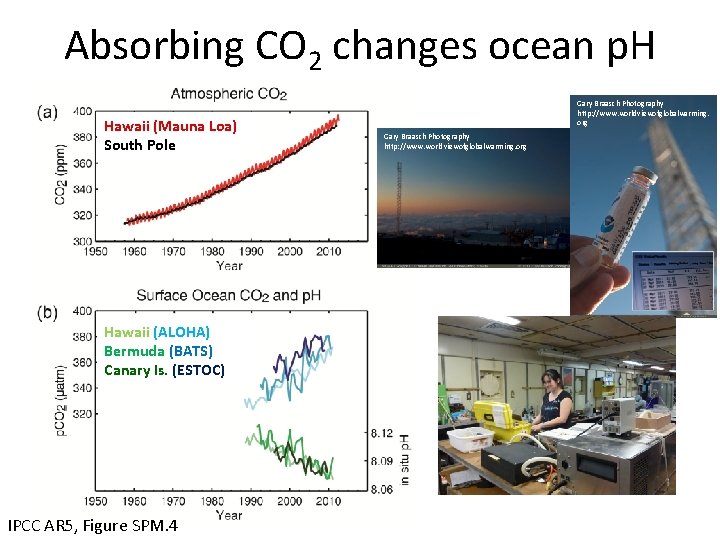
Absorbing CO 2 changes ocean p. H Hawaii (Mauna Loa) South Pole Hawaii (ALOHA) Bermuda (BATS) Canary Is. (ESTOC) IPCC AR 5, Figure SPM. 4 Gary Braasch Photography http: //www. worldviewofglobalwarming. org
![What is p. H all about anyway? • p. H = -log[H+] • Decrease What is p. H all about anyway? • p. H = -log[H+] • Decrease](http://slidetodoc.com/presentation_image/5db02ebd466c4f3fe8c32603a2f6db05/image-3.jpg)
What is p. H all about anyway? • p. H = -log[H+] • Decrease in p. H = increase in acidity • Logarithmic scale – A decrease of 1 unit is a 10× increase in [H+] – A decrease of 0. 1 unit (observed to date) is a 26% increase in [H+]
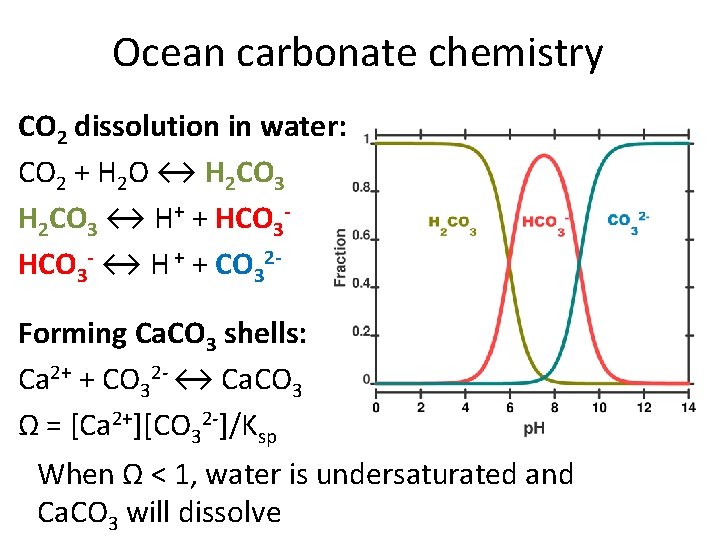
Ocean carbonate chemistry CO 2 dissolution in water: CO 2 + H 2 O ↔ H 2 CO 3 ↔ H+ + HCO 3 - ↔ H + + CO 32 Forming Ca. CO 3 shells: Ca 2+ + CO 32 - ↔ Ca. CO 3 Ω = [Ca 2+][CO 32 -]/Ksp When Ω < 1, water is undersaturated and Ca. CO 3 will dissolve

Calcifying organisms Geoduck Blue mussel Pink calcifying algae Urchins Dungeness crab Juvenile king crab • Examples of Puget Sound organisms that build shells, skeletons or other hard parts from Ca. CO 3 Images from Washington State Blue Ribbon Panel on Ocean Acidification Report, 2012

Pteropods: OA poster child • • Zooplankton, swimming snail Food for young salmon Shells dissolve in low p. H waters Observed dissolution in situ in Southern Ocean (Bednaršek et al. , 2012) Pteropods dissolve in water with p. H and carbonate chemistry expected for 2100
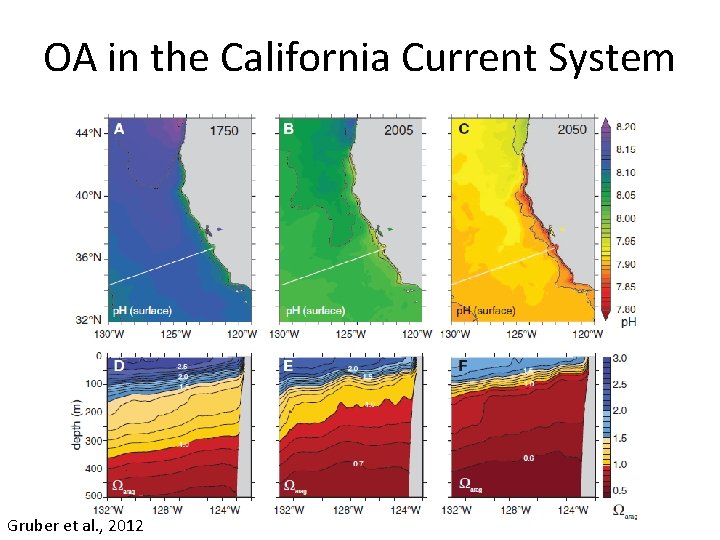
OA in the California Current System Gruber et al. , 2012
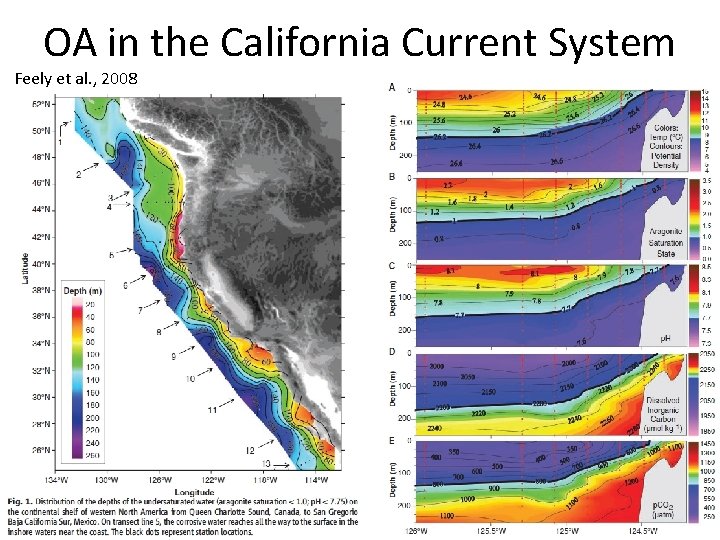
OA in the California Current System Feely et al. , 2008
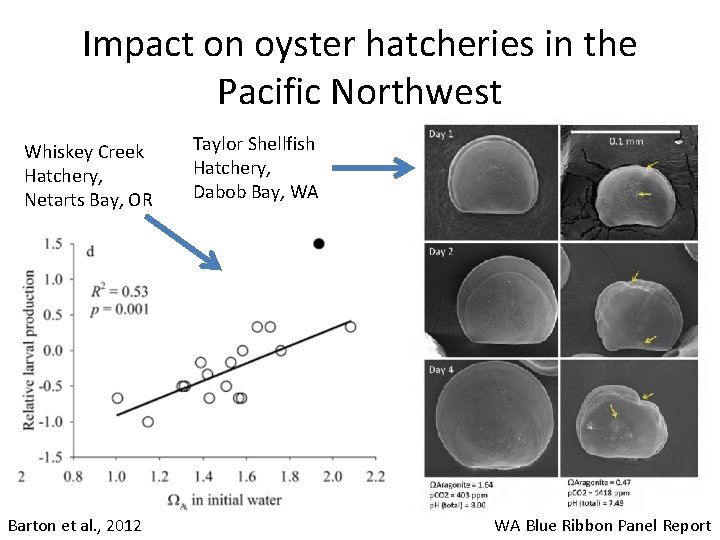
Impact on oyster hatcheries in the Pacific Northwest Whiskey Creek Hatchery, Netarts Bay, OR Barton et al. , 2012 Taylor Shellfish Hatchery, Dabob Bay, WA WA Blue Ribbon Panel Report
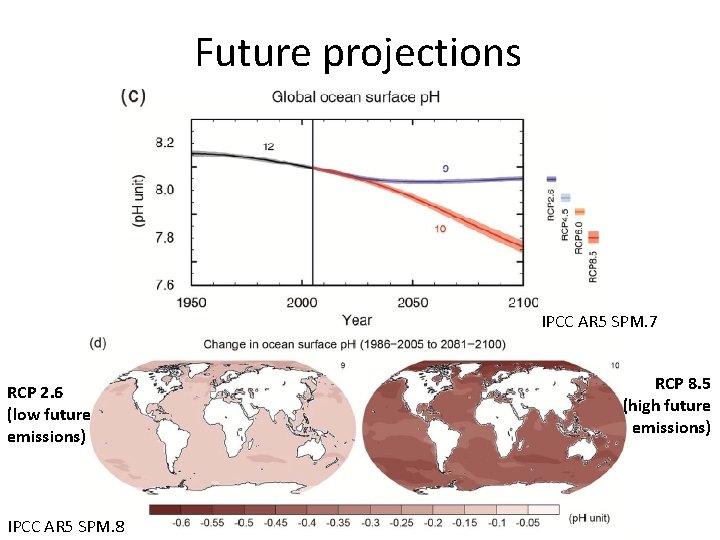
Future projections IPCC AR 5 SPM. 7 RCP 2. 6 (low future emissions) IPCC AR 5 SPM. 8 RCP 8. 5 (high future emissions)
- Slides: 10