Obstetric Care for Women with Opioid Use Disorder

Obstetric Care for Women with Opioid Use Disorder: The AIM Bundle Ronald E Iverson, Jr. , MD, MPH Vice-Chair, Obstetrics, Boston Medical Center Director, Quality Improvement, OBGYN, Boston Medical Center Assistant Professor, Boston University September 7, 2018 Boston Medical Center is the primary teaching affiliate of the Boston University School of Medicine.

DISCLOSURE I have no financial relationships with a commercial entity producing health care related products or services related to this presentation.


WE CREATED THIS EPIDEMIC
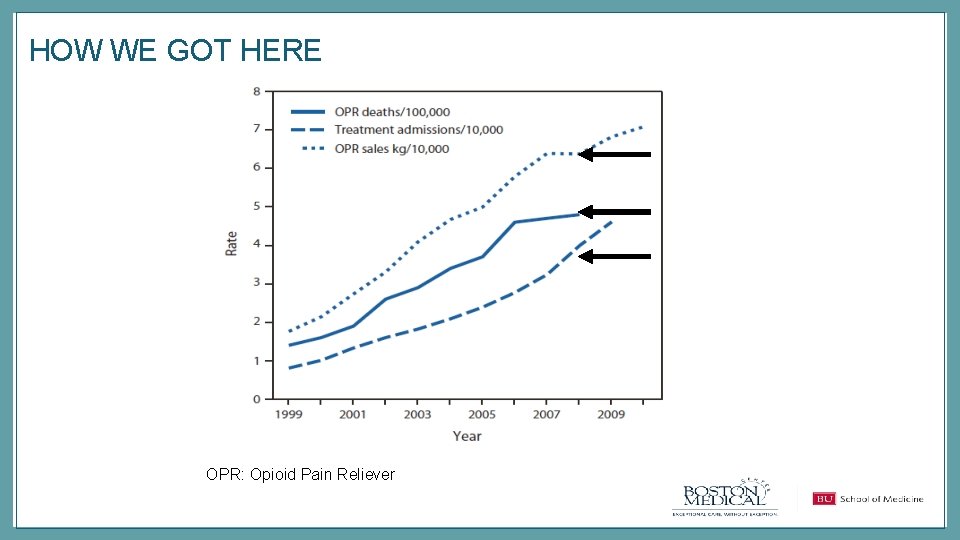
HOW WE GOT HERE OPR: Opioid Pain Reliever
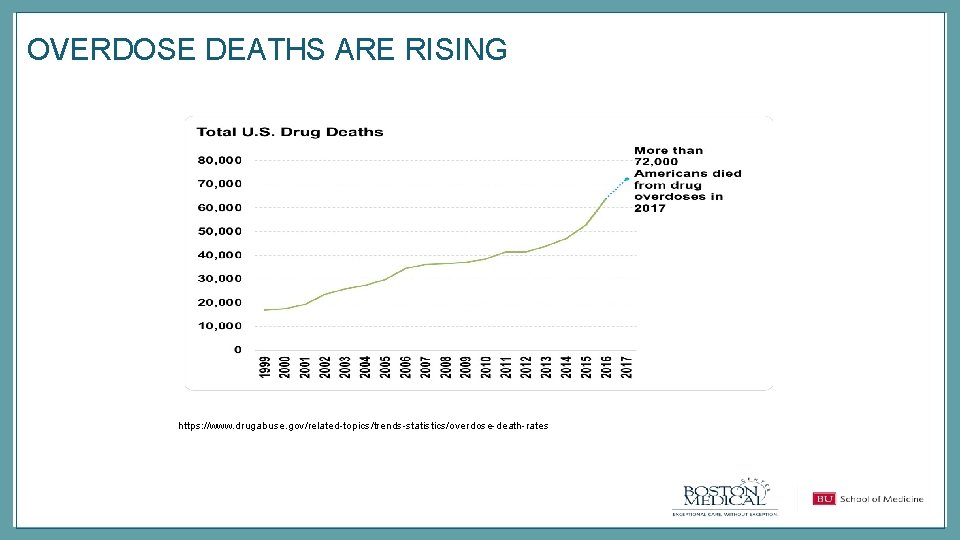
OVERDOSE DEATHS ARE RISING https: //www. drugabuse. gov/related-topics/trends-statistics/overdose-death-rates
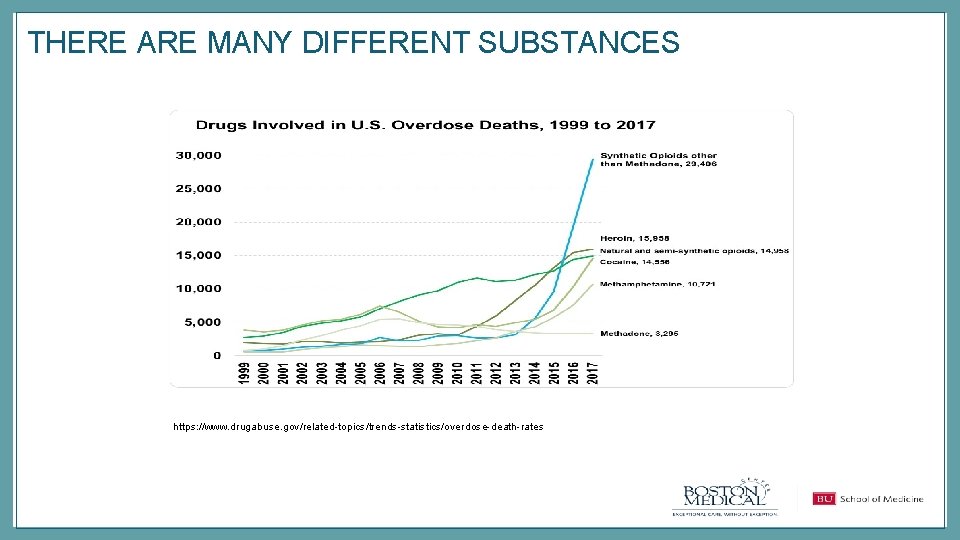
THERE ARE MANY DIFFERENT SUBSTANCES https: //www. drugabuse. gov/related-topics/trends-statistics/overdose-death-rates
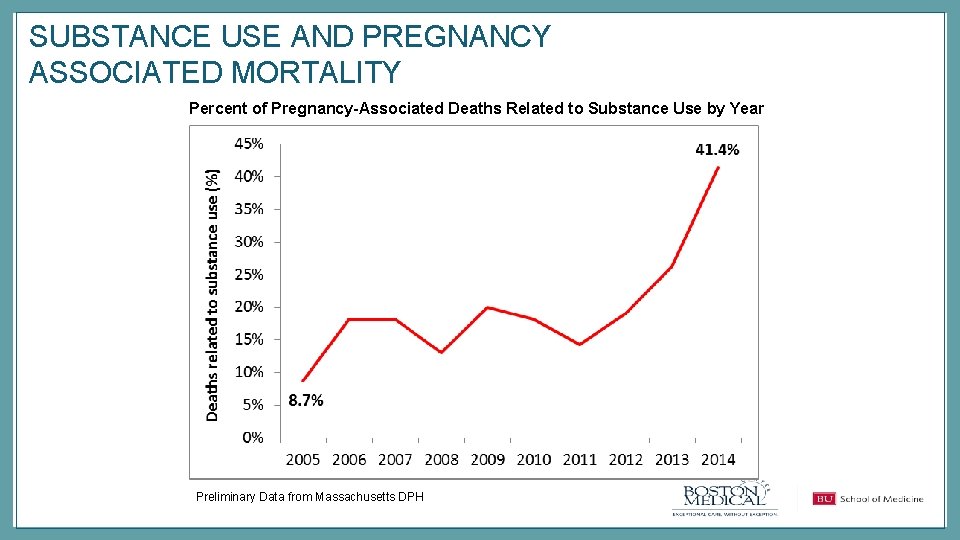
SUBSTANCE USE AND PREGNANCY ASSOCIATED MORTALITY Percent of Pregnancy-Associated Deaths Related to Substance Use by Year Preliminary Data from Massachusetts DPH
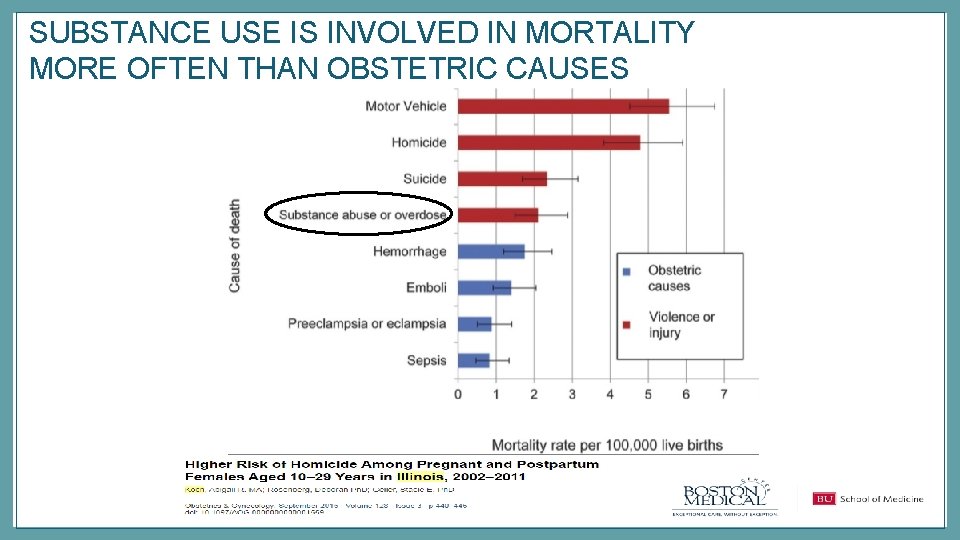
SUBSTANCE USE IS INVOLVED IN MORTALITY MORE OFTEN THAN OBSTETRIC CAUSES
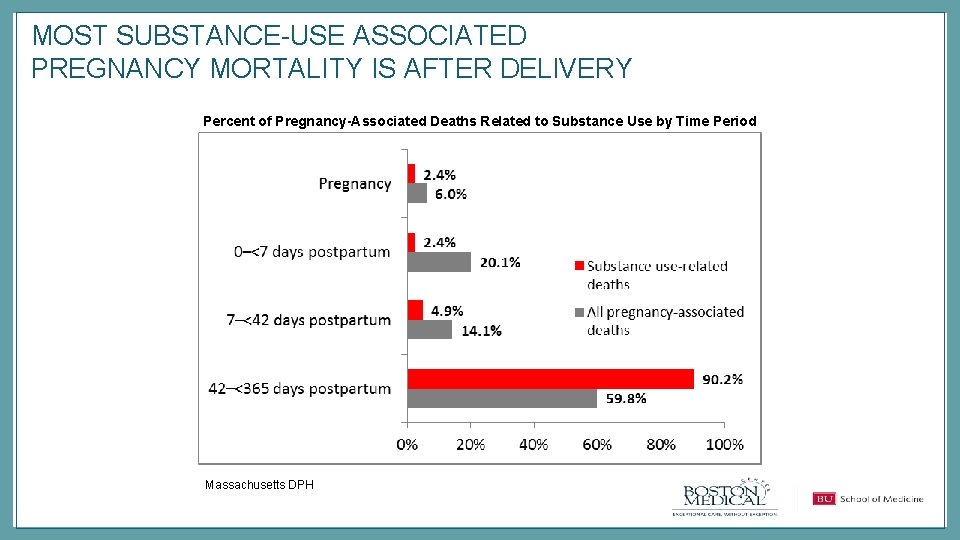
MOST SUBSTANCE-USE ASSOCIATED PREGNANCY MORTALITY IS AFTER DELIVERY Percent of Pregnancy-Associated Deaths Related to Substance Use by Time Period Massachusetts DPH

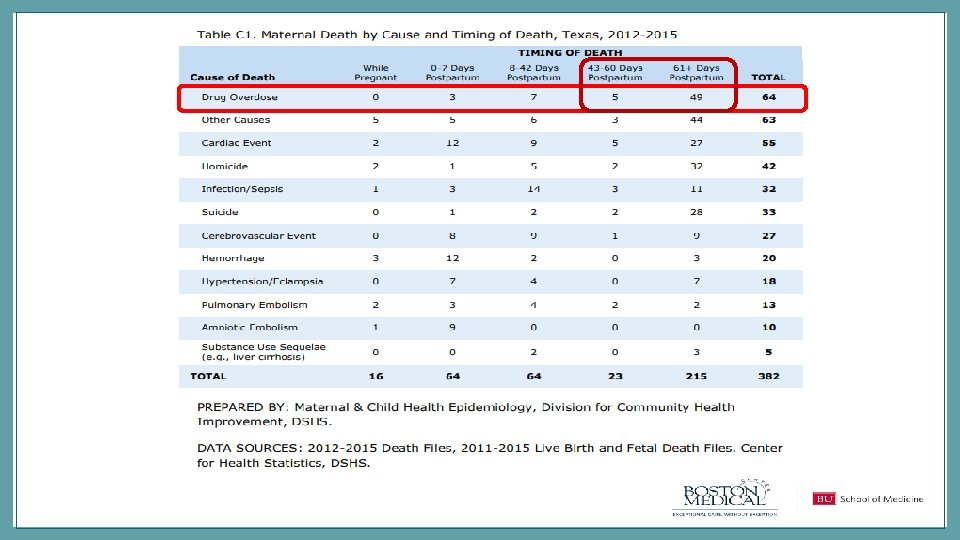
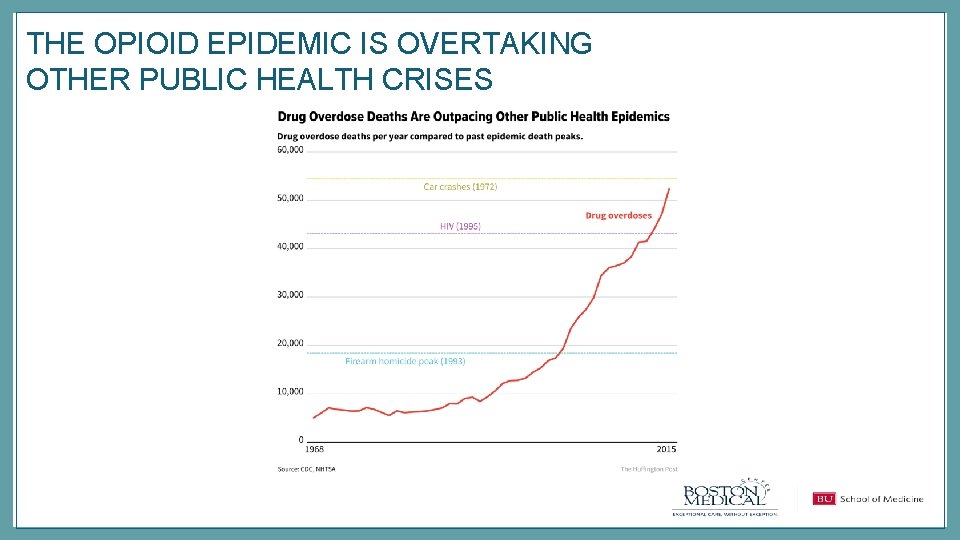
THE OPIOID EPIDEMIC IS OVERTAKING OTHER PUBLIC HEALTH CRISES
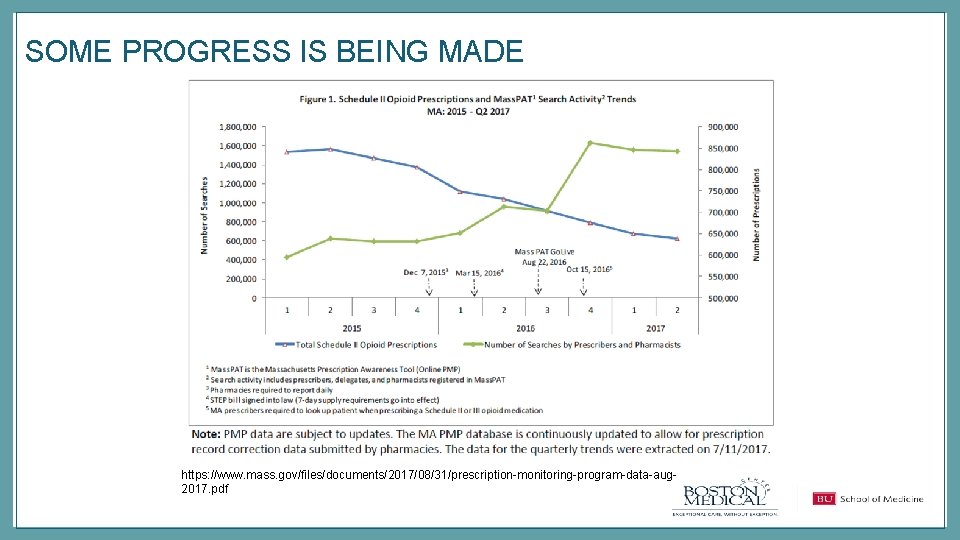
SOME PROGRESS IS BEING MADE https: //www. mass. gov/files/documents/2017/08/31/prescription-monitoring-program-data-aug 2017. pdf
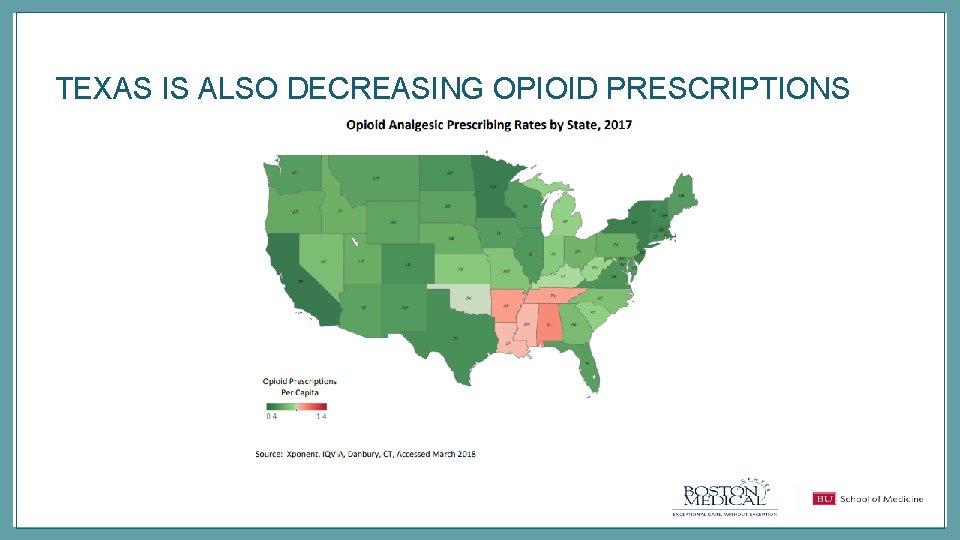
TEXAS IS ALSO DECREASING OPIOID PRESCRIPTIONS
ADDRESSING OPIOID USE ACROSS THE LIFE COURSE Family Pregnancy Newborn Infant Family

AIM OUD BUNDLE GOALS • Improve identification and care of women with OUD through screening and linkage to treatment • Optimize Medical Care of Pregnant Women with OUD • Increase access to MAT for pregnant and postpartum women with OUD • Prevent opioid use disorder by reducing the number of opioids prescribed for deliveries • Optimize the care of Opioid Exposed Newborns by improving maternal engagement in infant management
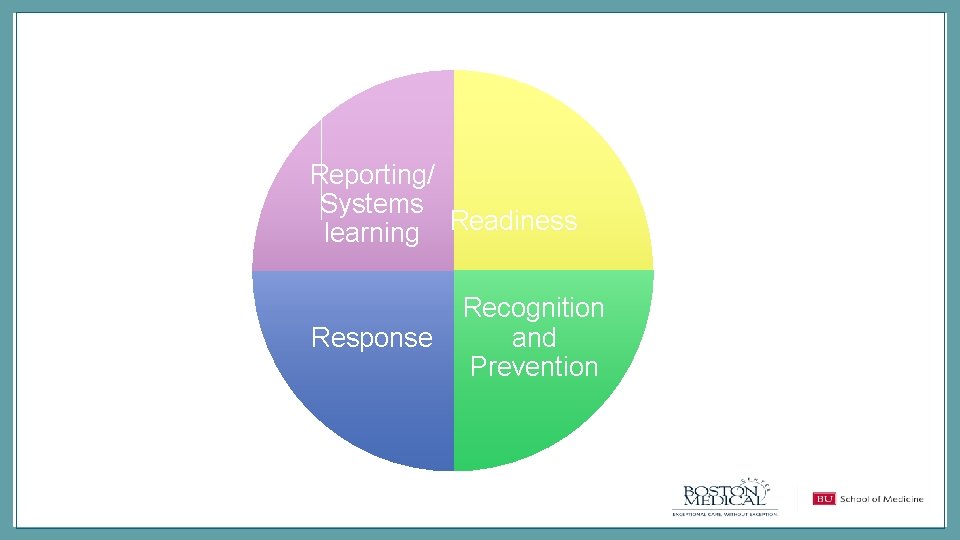
Reporting/ Systems learning Readiness Response Recognition and Prevention

READINESS: HEALTH SYSTEM Provide clinical and non-clinical staff education on SUDs ▪ SUDs are chronic medical conditions. ▪ Stigma, bias and discrimination negatively impact ▪ pregnant women with OUD. Provide training regarding trauma-informed care.
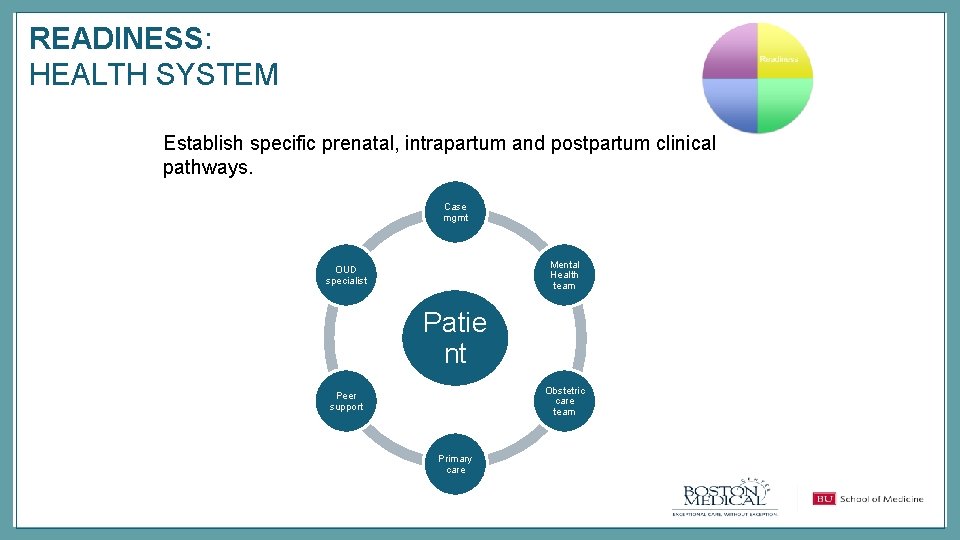
READINESS: HEALTH SYSTEM Establish specific prenatal, intrapartum and postpartum clinical pathways. Case mgmt Mental Health team OUD specialist Patie nt Obstetric care team Peer support Primary care

TRAUMA-INFORMED CARE • Understand the neurobiology of trauma • Recognize the signs and symptoms of trauma in patients and families • Screen for physical and sexual violence • Coordinate care with behavioral health/psychiatric care teams • Prevent re-traumatization

READINESS: HEALTH SYSTEM Identify local SUD treatment facilities ▪ ▪ Know ▪ ▪ ▪ Develop pain control protocols ▪ ▪ Provide women-centered care. Wrap-around services such as housing, child care, transportation and home visitation. Drug and alcohol counseling. State reporting guidelines regarding the use of opioid pharmacotherapy and identification of illicit substance use during pregnancy. Federal, state and county reporting guidelines for substance-exposed infants. Understand “Plan of Safe Care” requirements. Account for increased pain sensitivity and avoidance of mixed agonist-antagonist opioid analgesics. Order sets. Remove agonist/antagonists from Pyxis.
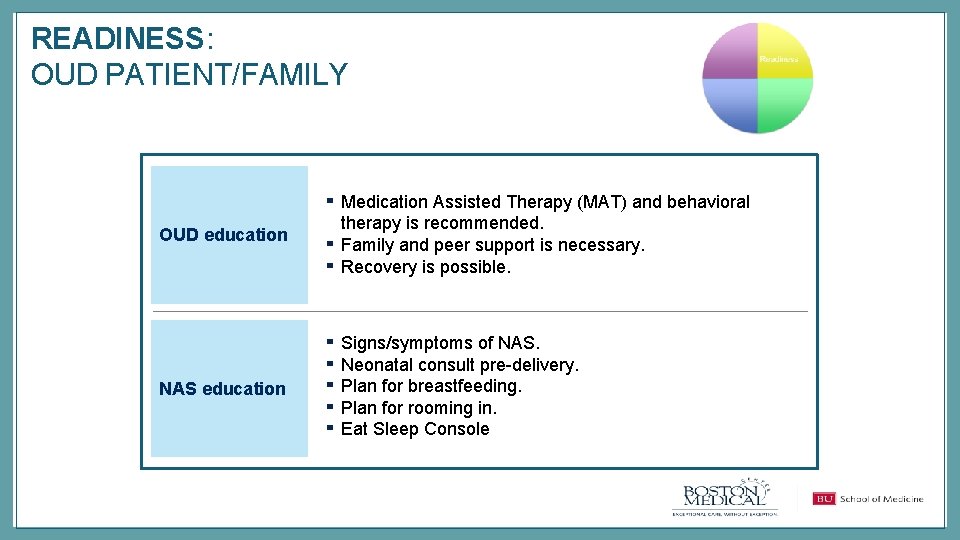
READINESS: OUD PATIENT/FAMILY ▪ OUD education NAS education ▪ ▪ Medication Assisted Therapy (MAT) and behavioral therapy is recommended. Family and peer support is necessary. Recovery is possible. ▪ ▪ ▪ Signs/symptoms of NAS. Neonatal consult pre-delivery. Plan for breastfeeding. Plan for rooming in. Eat Sleep Console
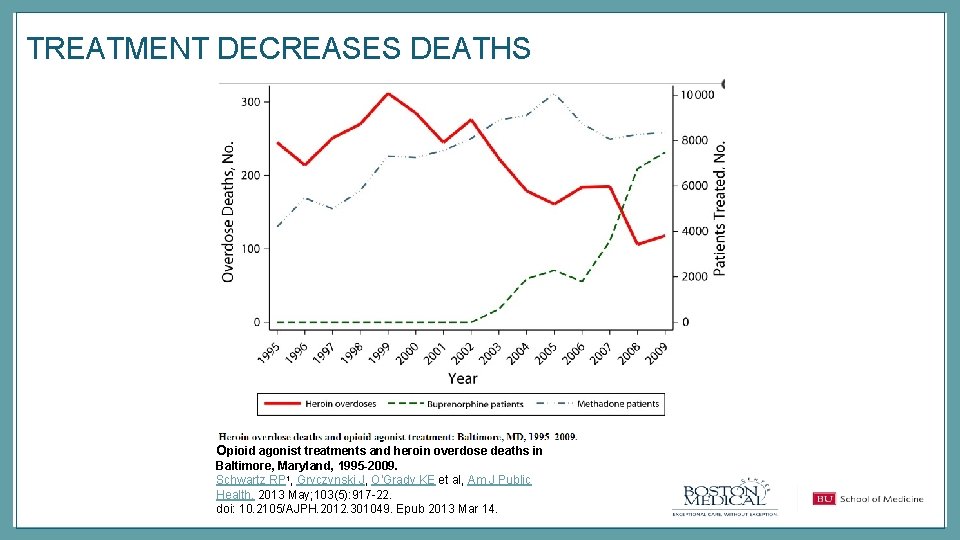
TREATMENT DECREASES DEATHS Opioid agonist treatments and heroin overdose deaths in Baltimore, Maryland, 1995 -2009. Schwartz RP 1, Gryczynski J, O'Grady KE et al, Am J Public Health. 2013 May; 103(5): 917 -22. doi: 10. 2105/AJPH. 2012. 301049. Epub 2013 Mar 14.
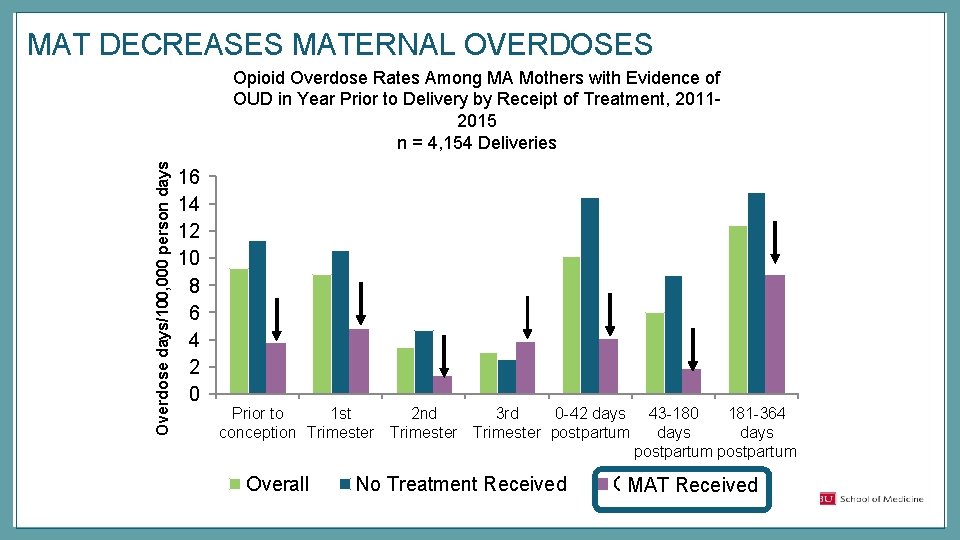
MAT DECREASES MATERNAL OVERDOSES Overdose days/100, 000 person days Opioid Overdose Rates Among MA Mothers with Evidence of OUD in Year Prior to Delivery by Receipt of Treatment, 20112015 n = 4, 154 Deliveries 16 14 12 10 8 6 4 2 0 Prior to 1 st 2 nd 3 rd 0 -42 days conception Trimester postpartum Overall No Treatment Received 43 -180 181 -364 days postpartum OAT Received MAT Received
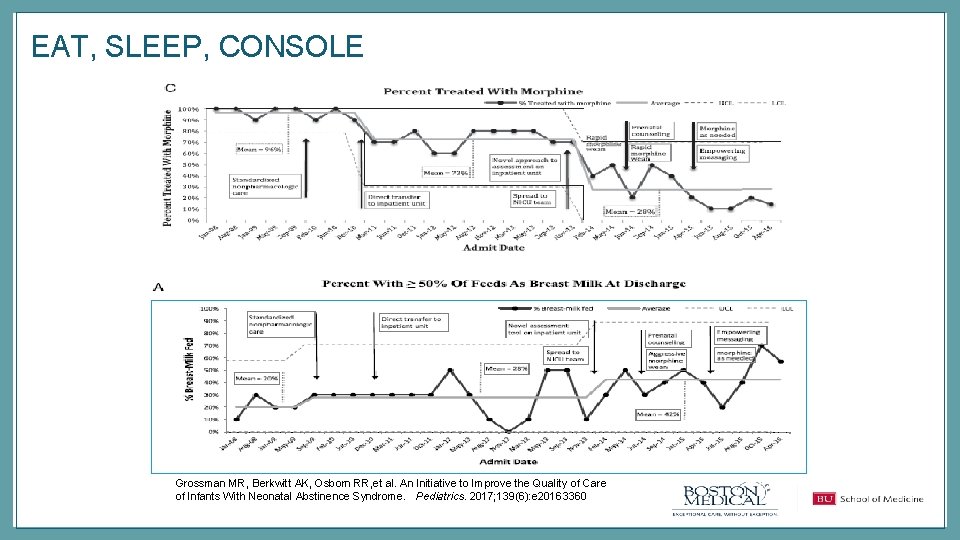
EAT, SLEEP, CONSOLE Grossman MR, Berkwitt AK, Osborn RR, et al. An Initiative to Improve the Quality of Care of Infants With Neonatal Abstinence Syndrome. Pediatrics. 2017; 139(6): e 20163360

READINESS: OUD PATIENT/FAMILY ▪ Child Abuse Prevention and Treatment Act Develop a “plan of safe care” for mom & baby ▪ (CAPTA) Ensure the safety and well-being of infants affected by substance use following release from health care providers.

RECOGNITION AND PREVENTION: CLINICAL SETTING Assess all pregnant women for SUDs Recognitio n and Prevention ▪ Drug and alcohol use. ▪ Screening, Brief Intervention and Referral to ▪ Treatment (SBIRT) Screen for polysubstance use among women with OUD.
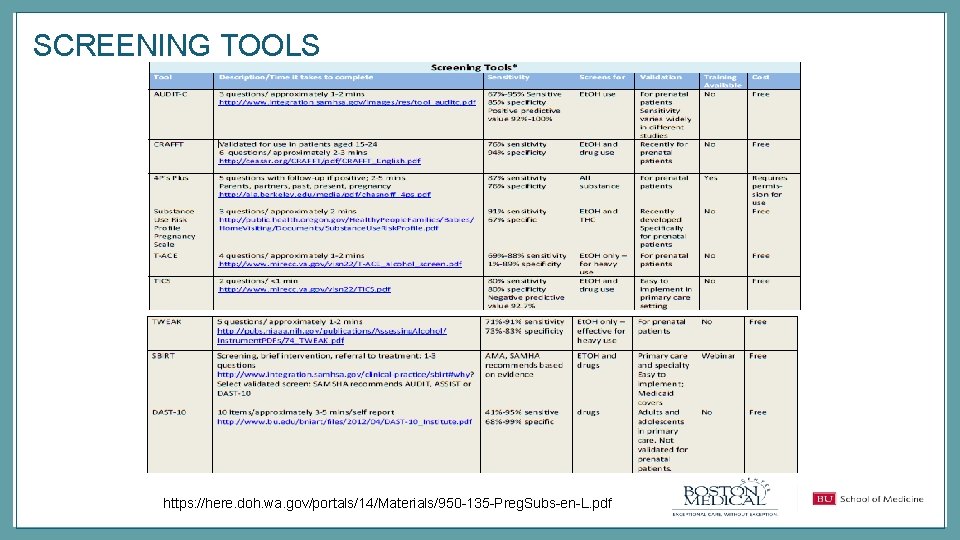
SCREENING TOOLS https: //here. doh. wa. gov/portals/14/Materials/950 -135 -Preg. Subs-en-L. pdf
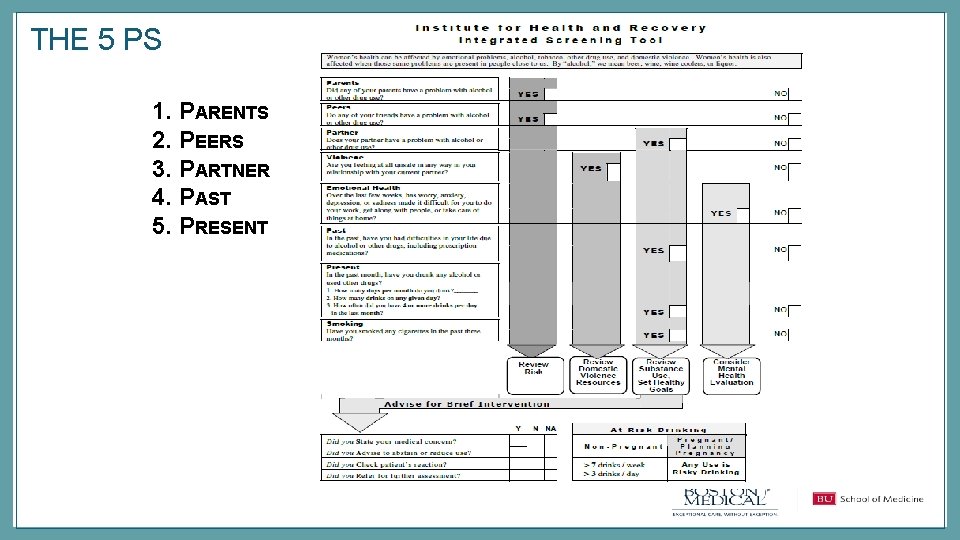
THE 5 PS 1. PARENTS 2. PEERS 3. PARTNER 4. PAST 5. PRESENT
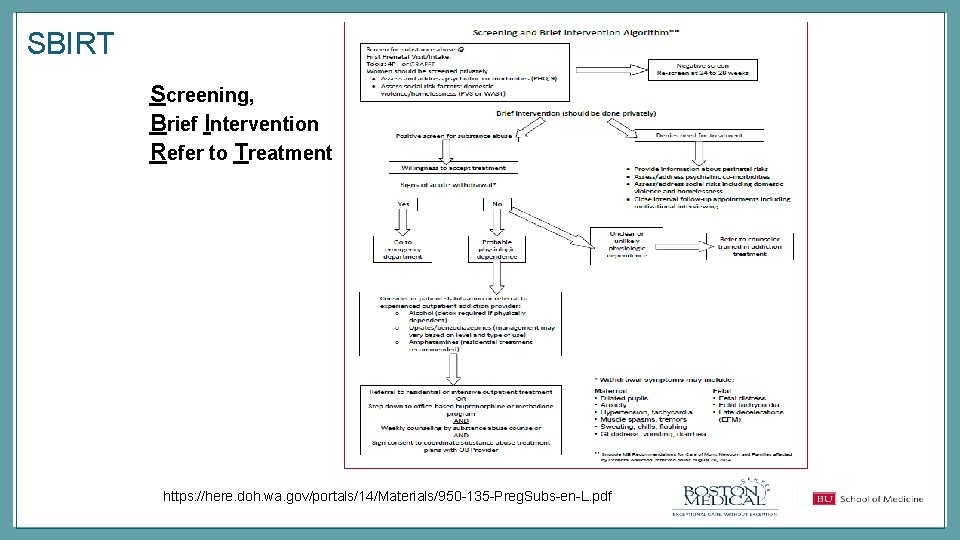
SBIRT Screening, Brief Intervention Refer to Treatment https: //here. doh. wa. gov/portals/14/Materials/950 -135 -Preg. Subs-en-L. pdf

RECOGNITION AND PREVENTION: CLINICAL SETTING Screen for commonly occurring comorbidities for all patients with OUD ▪ Infectious diseases ▪ Psychiatric disorders ▪ Physical and sexual violence ▪ Smoking cessation Recognitio n and Prevention

CHECKLIST http: //www. nnepqin. org/wp-content/uploads/2017/08/Toolkit. OUD
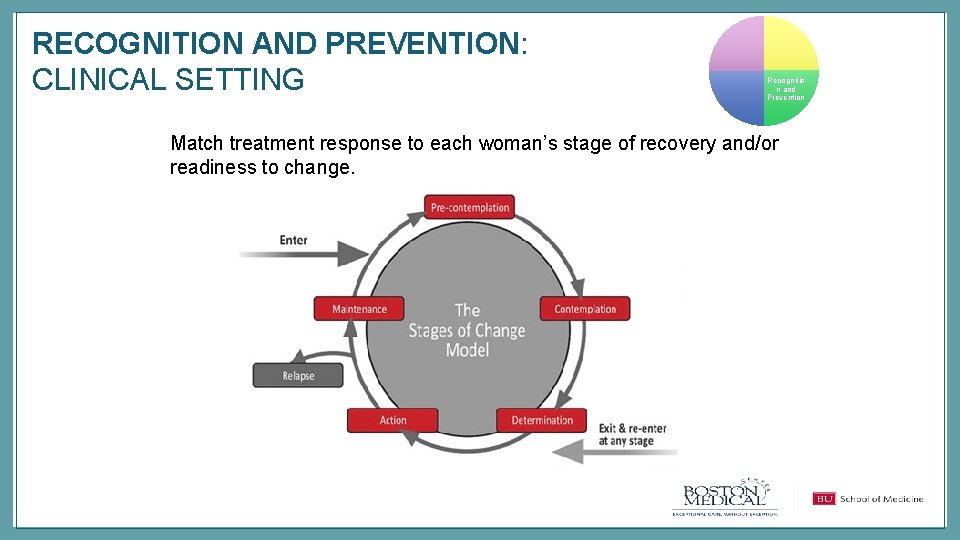
RECOGNITION AND PREVENTION: CLINICAL SETTING Recognitio n and Prevention Match treatment response to each woman’s stage of recovery and/or readiness to change.

RESPONSE: MEDICATION ASSISTED TREATMENT (MAT) Response For pregnant women with an opioid use disorder, opioid agonist pharmacotherapy is the recommended therapy and is preferable to medically supervised withdrawal because withdrawal is associated with high relapse rates, which lead to worse outcomes. More research is needed to assess the safety (particularly regarding maternal relapse), efficacy, and long-term outcomes of medically supervised withdrawal.
RESPONSE: HEALTH SYSTEM All patients with OUD are enrolled in a womancentered OUD treatment program Incorporate key counseling, education and resources into care pathways Response ▪ ▪ Establish clinic relationships. Link to local resources that support recovery. ▪ ▪ Breastfeeding and lactation support. Immediate postpartum contraceptive (LARC) options. Pain management strategies Infant care

BREASTFEEDING EDUCATION http: //pcssmat. org/wp-content/uploads/2013/10/ASAM-WAGBrochure-Opioid-Labor
RESPONSE: HEALTH SYSTEM Response Coordinate among providers during pregnancy, postpartum and the interconception period Engage child welfare services ▪ ▪ ▪ Referrals to providers for co-morbid conditions. Lead provider responsible for care coordination. Communication strategy. ▪ Develop safe care protocols tailored to the patient and family’s OUD treatment and resource needs.
REPORTING & SYSTEMS LEARNING: HEALTH SYSTEM Reporting/ Systems Learning Develop mechanisms to collect data and monitor process and outcome metrics Create multidisciplinary case review teams Develop learning opportunities for providers and staff ▪ ▪ Inpatient and outpatient Data dashboard measures – Outcome – Process ▪ Evaluate patient, provider and system-level issues. ▪ Use data and events to educate teams

AIM OUTCOME MEASURES Reporting/ Systems Learning • Pregnancy Associated Opioid Deaths • Average length of stay for newborns with Neonatal Abstinence Syndrome (NAS)

AIM PROCESS MEASURES Reporting/ Systems Learning • Percent of OEN receiving mother’s milk at newborn discharge • Percent of women with OUD during pregnancy who receive medication assisted treatment MAT or behavioral health treatment • Percent of OEN who go home to biological mother

AIM STRUCTURE MEASURES Reporting/ Systems Learning • Percent of Prenatal Care Sites which have implemented a universal screening protocol for OUD • Percent of delivery sites using post-delivery and discharge pain management prescribing practices for routine vaginal and cesarean births focused on limiting opioid prescription • Percent of delivery sites with OUD specific pain management and opioid prescribing guidelines

AIM STATE SURVEILLANCE MEASURES Reporting/ Systems Learning • Percent of newborns diagnosed as affected by maternal use of opiates • Percent of newborns diagnosed with NAS

AIM GLOSSARY • • • Newborn Neonatal Abstinence Syndrome (NAS) Opioid Use Disorder (OUD)/Pregnant woman with OUD Medication Assisted Treatment (MAT) Opioid Exposed Newborn (OEN) Mother’s milk at discharge Screening Testing Ongoing OUD training

REPORTING & SYSTEMS LEARNING: HEALTH SYSTEM Connect other stakeholders with clinical providers and health systems to share outcomes and identify ways to improve systems of care ▪ Child welfare services ▪ Public health agencies ▪ Court systems ▪ Law enforcement Reporting/ Systems Learning

BUNDLE IMPLEMENTATION • • Multidisciplinary team Agreement on Aims PDSA testing of Bundle Components Follow Process and Balancing Measures Incorporate into the EMR Education/Simulations Incorporate into Policy

47 TOOLS AND RESOURCES: SLIDE DECKS Stigma Screening NAS

48 TOOLS AND RESOURCES: CLINICAL PATHWAYS Checklist Screening Tool Chart

49 TOOLS AND RESOURCES: COLLABORATIVE CHART Opioid Collaborative Aim (Bundle Component) Improve identification and care of women with OUD through screening and linkage to treatment RECOGNITION Key Drivers Interventions Universal Prenatal Screening Brief Intervention Referral to Treatment Map local resources: Resources Metrics List of Validated Screening Tools 1. (S 1) Percent of Prenatal Care Sites which have implemented a -Identify existing models AIM Provider Education Slide universal screening protocol for Decks: OUD (Required AIM Structure -Identify partners to receive referrals AIM Stigma Slides Measure) AIM Screening Slides -Establish protocols for referral ACOG Committee Opinions: Select validated screening tool ACOG CO 711 “Opioid Use and Opioid Use Disorder During Train staff to use screening tool Pregnancy” Assess screen positive women for OUD Screening resources: Link women with OUD to OB and OUD -NNEPQIN local resource mapping template treatment services -SAMHSA Guidance -Snuggle. ME -SBIRT Oregon -WA State Screening Guidelines -NNEPQIN Guidelines -WHO Guidelines Other Resources: ACOG District II Slide Deck Denominator: Number of PNC sites associated with delivery sites Numerator: Among the denominator, those sites using universal screening for OUD with all pregnant patients

BUNDLES = COMPLETE CARE Reporting/ Systems learning Readiness Response Recognition and Prevention

THANK YOU



READINESS: PLAN OF SAFE CARE • Child Abuse Prevention and Treatment Act (CAPTA) • Ensure the safety and well-being of infants affected by substance use following release from health care providers • Address the health and substance use disorder treatment needs of the infant and family • Refer and deliver appropriate services to the infant and affected family or caregiver

Reportin g/ Systems Learning AIM OUTCOME MEASURES Data Source Outcome Measures (O) Description Frequency Notes O 1: Pregnancy Associated Opioid Deaths Denominator: Number of women delivering in that year State Data Annually Maybe consistently delayed by 1 -2 years based on state data Numerator: Among the denominator, those with opioid related death within 1 availability year of delivery O 2: Average length of stay for Denominator: Number of newborns ≥ 35 weeks gestation diagnosed with NAS Hospital Data Form/ Annually newborns 1 with Neonatal Numerator: Among the denominator, total number of hospital days State Data and ICD Abstinence Syndrome (NAS)2 10 code/ Linked LOS P 96. 1 Neonatal withdrawal symptoms from maternal use of drugs of addiction
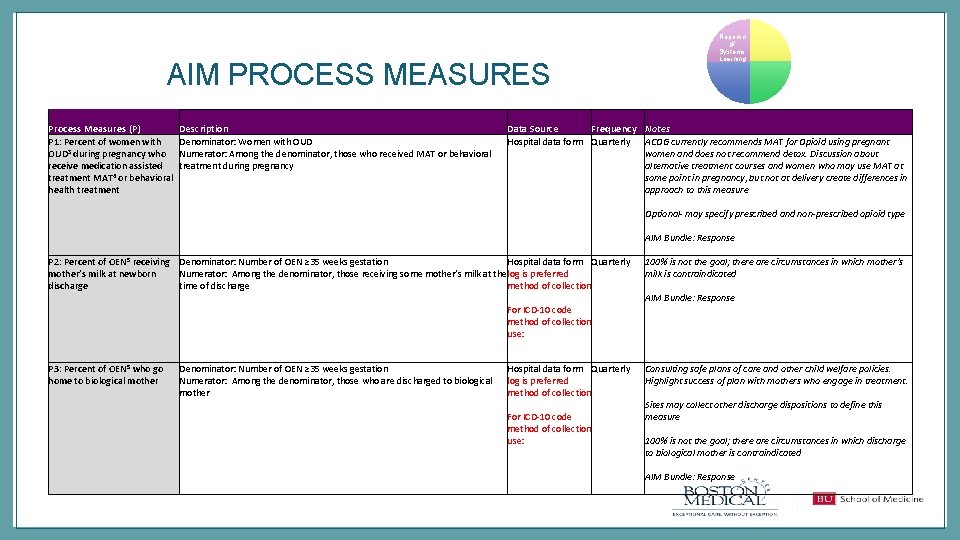
AIM PROCESS MEASURES Process Measures (P) P 1: Percent of women with OUD 3 during pregnancy who receive medication assisted treatment MAT 4 or behavioral health treatment Description Denominator: Women with OUD Numerator: Among the denominator, those who received MAT or behavioral treatment during pregnancy Reportin g/ Systems Learning Data Source Frequency Notes Hospital data form Quarterly ACOG currently recommends MAT for Opioid using pregnant women and does not recommend detox. Discussion about alternative treatment courses and women who may use MAT at some point in pregnancy, but not at delivery create differences in approach to this measure Optional- may specify prescribed and non-prescribed opioid type AIM Bundle: Response P 2: Percent of OEN 5 receiving Denominator: Number of OEN ≥ 35 weeks gestation Hospital data form Quarterly mother’s milk at newborn Numerator: Among the denominator, those receiving some mother’s milk at the log is preferred discharge time of discharge method of collection For ICD-10 code method of collection use: P 3: Percent of OEN 5 who go home to biological mother Denominator: Number of OEN ≥ 35 weeks gestation Hospital data form Quarterly Numerator: Among the denominator, those who are discharged to biological log is preferred mother method of collection For ICD-10 code method of collection use: 100% is not the goal; there are circumstances in which mother’s milk is contraindicated AIM Bundle: Response Consulting safe plans of care and other child welfare policies. Highlight success of plan with mothers who engage in treatment. Sites may collect other discharge dispositions to define this measure 100% is not the goal; there are circumstances in which discharge to biological mother is contraindicated AIM Bundle: Response
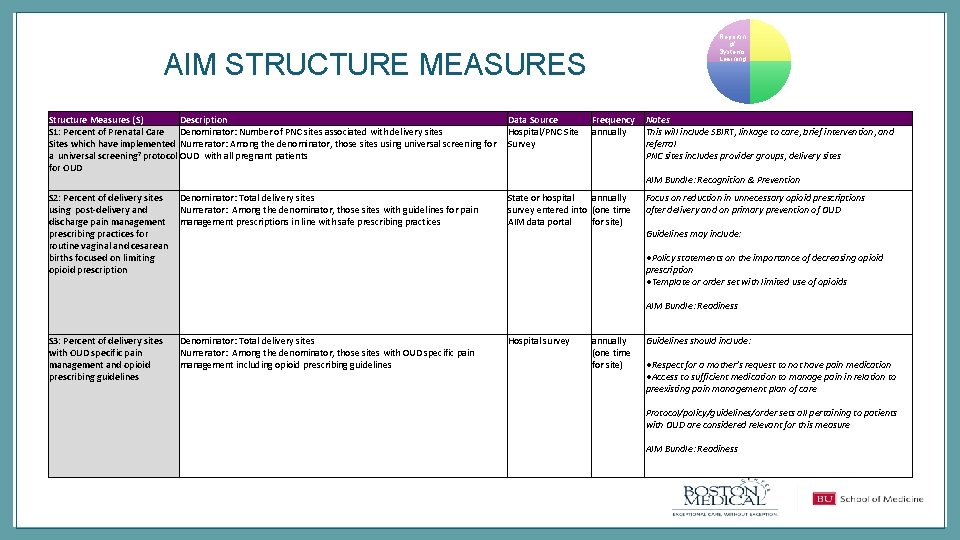
Reportin g/ Systems Learning AIM STRUCTURE MEASURES Structure Measures (S) Description Data Source S 1: Percent of Prenatal Care Denominator: Number of PNC sites associated with delivery sites Hospital/PNC Sites which have implemented Numerator: Among the denominator, those sites using universal screening for Survey a universal screening 7 protocol OUD with all pregnant patients for OUD S 2: Percent of delivery sites using post-delivery and discharge pain management prescribing practices for routine vaginal and cesarean births focused on limiting opioid prescription Denominator: Total delivery sites Numerator: Among the denominator, those sites with guidelines for pain management prescriptions in line with safe prescribing practices Frequency annually Notes This will include SBIRT, linkage to care, brief intervention, and referral PNC sites includes provider groups, delivery sites AIM Bundle: Recognition & Prevention State or hospital annually survey entered into (one time AIM data portal for site) Focus on reduction in unnecessary opioid prescriptions after delivery and on primary prevention of OUD Guidelines may include: ●Policy statements on the importance of decreasing opioid prescription ●Template or order set with limited use of opioids AIM Bundle: Readiness S 3: Percent of delivery sites with OUD specific pain management and opioid prescribing guidelines Denominator: Total delivery sites Numerator: Among the denominator, those sites with OUD specific pain management including opioid prescribing guidelines Hospital survey annually (one time for site) Guidelines should include: ●Respect for a mother’s request to not have pain medication ●Access to sufficient medication to manage pain in relation to preexisting pain management plan of care Protocol/policy/guidelines/order sets all pertaining to patients with OUD are considered relevant for this measure AIM Bundle: Readiness
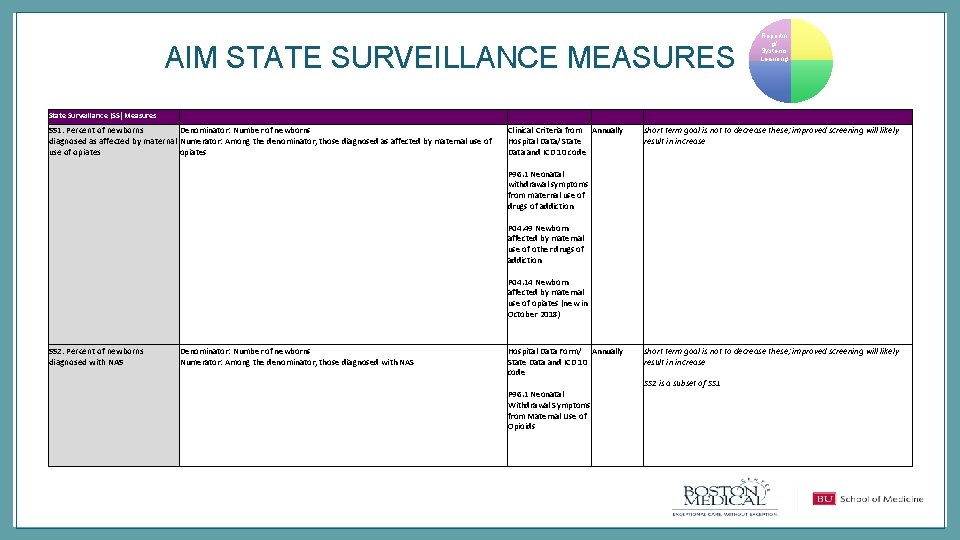
AIM STATE SURVEILLANCE MEASURES State Surveillance (SS) Measures SS 1: Percent of newborns Denominator: Number of newborns diagnosed as affected by maternal Numerator: Among the denominator, those diagnosed as affected by maternal use of opiates Clinical Criteria from Annually Hospital Data/ State Data and ICD 10 code Reportin g/ Systems Learning short term goal is not to decrease these; improved screening will likely result in increase P 96. 1 Neonatal withdrawal symptoms from maternal use of drugs of addiction P 04. 49 Newborn affected by maternal use of other drugs of addiction P 04. 14 Newborn affected by maternal use of opiates (new in October 2018) SS 2: Percent of newborns diagnosed with NAS Denominator: Number of newborns Hospital Data Form/ Annually Numerator: Among the denominator, those diagnosed with NAS State Data and ICD 10 code P 96. 1 Neonatal Withdrawal Symptoms from Maternal Use of Opioids short term goal is not to decrease these; improved screening will likely result in increase SS 2 is a subset of SS 1

AIM GLOSSARY Reportin g/ Systems Learning TERM DEFINITION 1. Newborn Infant admitted at 0 days old, transfer admission up to 1 week old, or readmission from home/ER/clinic up to 1 week old *Admitted at less than 7 days old 2. Neonatal Abstinence Syndrome (NAS) Refer to ICD 10 Code P 96. 1 Neonatal Withdrawal Symptoms from Maternal Use of Drugs of Addiction 3. Opioid Use Disorder (OUD)/ Pregnant Woman with Opioid Use Disorder Clinical Criteria: All women delivering at your hospital with: • positive self-report screen or positive opioid toxicology screen during pregnancy and assessed to have OUD, or • Patient endorses or reports misuse of opioids / opioid use disorder, or • using non-prescribed opioids during pregnancy, or • using prescribed opioids chronically for longer than a month in the third trimester 4. Medication Assisted Treatment (MAT): the use of FDA- approved medications, in combination with counseling and behavioral therapies, to provide a “whole-patient” approach to the treatment of substance use disorders (SAMHSA, 2018) 5. Opioid Exposed Newborn ≥ 35 weeks (OEN) Clinical Criteria: All infants of mothers with opioid use disorder if mother has: • positive self-report screen or positive opioid toxicology screen during pregnancy and assessed to have OUD, or • Patient endorses or reports misuse of opioids / opioid use disorder, or • using non-prescribed opioids during pregnancy, or • using prescribed opioids chronically for longer than a month in the third trimester, or • if newborn has an unanticipated positive neonatal cord, urine, or meconium screen for opioids. ● if newborn affected by maternal use of opioids including NAS Using ICD-10 data will not be as accurate as clinical criteria above and will require a linkage of mother and baby discharge codes for best estimate and so is not recommended for routine use. Log created from hospital data form is preferred method of data collection. If using ICD-10 data, check both infant and maternal diagnoses: Newborn affected by maternal use of opiates P 96. 1 Neonatal withdrawal symptoms from maternal use of drugs of addiction P 04. 49 Newborn affected by maternal use of other drugs of addiction P 04. 14 Newborn affected by maternal use of opiates (new in October 2018) And Maternal codes for Opioid abuse, dependency, or use: F 11. xx *Note: For process measures that use OEN ≥ 35 weeks as the denominator, this is limited to those OEN ≥ 35 who were discharged home (i. e. exlclude those who were discharged to another NICU/ death, etc)* 6. Mother's milk at discharge 7. Screening 8. Testing 9. Ongoing Opioid Use Disorder (OUD) Training Mother's Milk at Discharge: Any ongoing plan for use of some mother's milk after discharge Screening: Verbal and written questions regarding opiate use. Screening tests include NIDA, 4 Ps, 5 Ps and others; refer to AIM screening tool guide A biologic test of serum, urine, hair for presence of opioids Structured education completed every 2 years
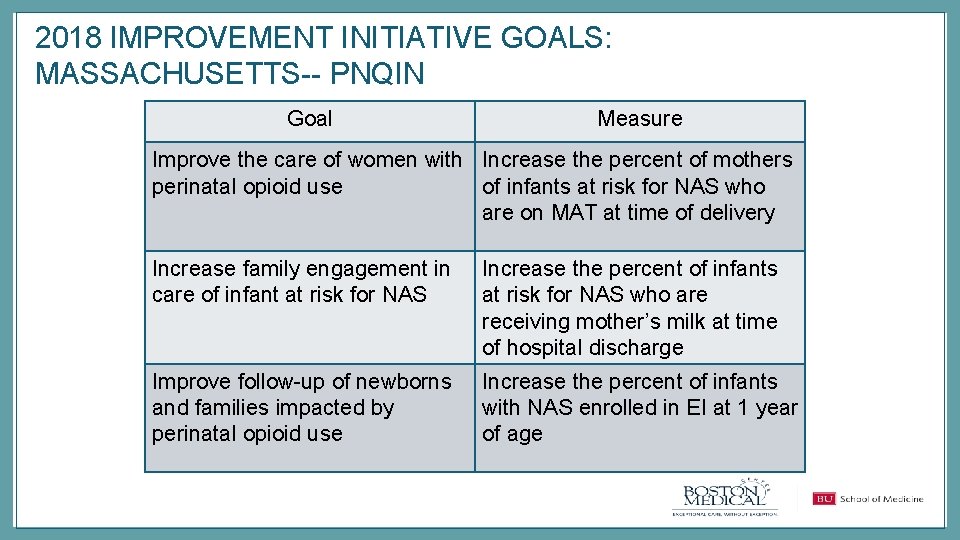
2018 IMPROVEMENT INITIATIVE GOALS: MASSACHUSETTS-- PNQIN Goal Measure Improve the care of women with Increase the percent of mothers perinatal opioid use of infants at risk for NAS who are on MAT at time of delivery Increase family engagement in care of infant at risk for NAS Increase the percent of infants at risk for NAS who are receiving mother’s milk at time of hospital discharge Improve follow-up of newborns and families impacted by perinatal opioid use Increase the percent of infants with NAS enrolled in EI at 1 year of age
PERINATAL OPIOID USE: GOALS AND MEASURES IN MASSACHUSETTS Family Pregnancy Newborn Infant Family Improve care of women with opioid use disorder (OUD) • Percent of mothers with OUD in medication assisted treatment (MAT)* • Percent of mothers screened for OUD in pregnancy Improve care of opioid-exposed newborns (OEN) by increasing family engagement • Percent of OENs receiving mother’s milk at discharge* • Percent of OENs rooming in during hospitalization • Percent of OENs requiring pharmacologic therapy for NAS Improve follow-up and support of families impacted by perinatal opioid use • Percent of infants with NAS enrolled in EI at 1 year of age* • Readmission rate for infants with NAS • Percent of mothers with OUD receiving MAT 1 year after delivery * Primary outcome measures
- Slides: 61