Observations Measurements Regents Earth Science Topic 1 Observations

Observations & Measurements Regent’s Earth Science Topic #1

Observations • Using one or more senses to gather information. • Quantitative – involves numerical quantity (magnitude). Can be scalar or vector quantities. • Qualitative describes without a numerical quantity.

Scalar vs. Vector Quantities • Scalar Quantities- have magnitude only. Time, temperature, mass, volume are examples. • Vector Quantities- have both magnitude and direction. Any force (magnetic for example), wind velocity, ocean currents all have speed or force along with the direction so are vector quantities. Wind out of the south @ 15 knots.

Instruments • They extend senses. • Senses are extended so your measurements can be more accurate. • Instruments include meter sticks, graduated cylinders, balances, stopwatches, microscope s, telescopes & many more.

Inference • Interpretations or conclusions based on observations and/or previous experiences.

Prediction • An inference about what will happen in the future.

Measurement • Quantitative measurement. • Comparison to a known quantity • Always has magnitude (numerical quantity) and a unit of measure. Sometimes with a direction. • Length – measure of distance. Instrument: ruler/meter stick. • Mass – amount of matter present Instrument: balance. • Time – sense that events are sequential compared to sec, min, hr, yr. Instruments: clock/ calendar.

Metric (S. I. )Units • Length – mm, cm, m, km • Mass – g, kg • Time – sec, min, hr, yr

Volume • The space an object or substance takes up. (size) • Metric units – ml, L, cc, cm 3.

Volume (Rectangular Solids) Step #1 (formula) V= L x W x H Step #2 (substitution) V=4 x 3 x 2 Step #3 (simplify answer) V = 24. 0 cm 3 3 cm 4 cm 2 cm

Density • Concentration of matter. • Measures how tightly atoms are packed in the substance. • D = M/V

Density Calc. #1 • Small piece of quartz. Step #1 D = M/V Step #2 D = 14. 04 g/5. 2 cm 3 Step #3 D = 2. 7 g/cm 3

Density Calc. #2 • Large piece of quartz. Step #1 D = M/V Step #2 D = 405. 0 g/150. 0 cm 3 Step #3 D = 2. 7 g/cm 3

Density of Rectangular Solids Volume: Step #1 V= L x W x H Density: Step #1 D = M/V Step #2 V=2 x 2 x 5 Step #2 D = 50. 0 g/20. 0 cm 3 Step #3 V = 20. 0 cm 3 Step #3 D = 2. 5 g/cm 3
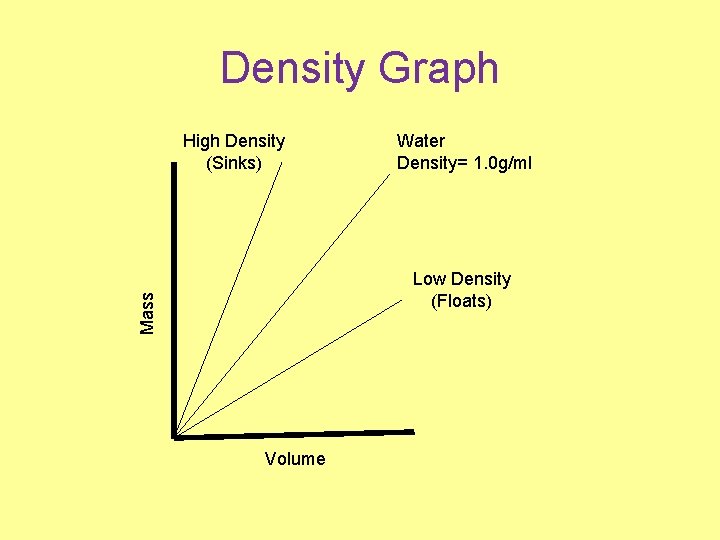
Density Graph High Density (Sinks) Water Density= 1. 0 g/ml Mass Low Density (Floats) Volume

Factors Affecting Density • Heat causes materials to expand because atoms move faster & farther apart, so it lowers the density. • Pressure pushes atoms closer together, so density increases. These 2 factors always will change density!

Density vs. Phase • Usually solids are most dense, liquids medium and gases are least dense. 2 Exceptions • Mercury is a liquid that is very dense. ~ 13 g/ml. ( 1 gallon > 100 lbs) • Water is denser than ice, because it expands when it freezes. Water is the only substance that does this.

Buoyancy (Float/Sink) • Low density materials float on high density materials. • Earth is layered by density. Atmosphere – Gases (Least Dense) Hydrosphere – Liquids Lithosphere – Solids (Most Dense)

Error (Not Mistakes!) • Called deviation or scientific error. • No measurement is perfect. • All measurements are limited by your senses and the precision (accuracy) of your instruments.

%Error Or % Deviation Step #1 % Error = difference from accepted value x 100 accepted value Step #2 % Error = 127. 5 – 125. 0 Step #3 % Error = 2. 0 % x 100 OR 2. 5 x 100 125. 0

Event • Change of properties of an object or within a system. • Events can happen quickly or take long periods of time.

Frame of Reference • Used to describe change by comparing to something. • 2 most common are time or space.

Time • Compares when changes occur. • How fast the change occurs. Space • Compares locations or where the change occurs. • What direction the change is progressing.

Cyclic Change • Orderly change that repeats over & over. Examples include: • • • Seasons/Temperature Apparent Sun Diameter Moon phases Eclipses River Discharge Erosion &Deposition Rates

Rate of Change = Change in field value Time See E. S. R. T. Page #1

Time Referenced Rate Change Step #1 (formula) Rate of Change = Change in field value Time Step #2 (substitution) Rate of Change = 25°-18°C OR 7°C 2 hr. 2 hr. Don’t Step #3 (simplify answer) Forget Rate of Change = 3. 5°C/hr. Units!

Gradient (Space Referenced Rate Change) Step #1 (formula) Rate of Change = ∆ Elevation Distance Step #2 (substitution) Rate of Change = 2000 m-1200 m OR 800 m 5 km. 5 km. Don’t Step #3 (simplify answer) Forget Rate of Change = 160. 0 m/km. Units!
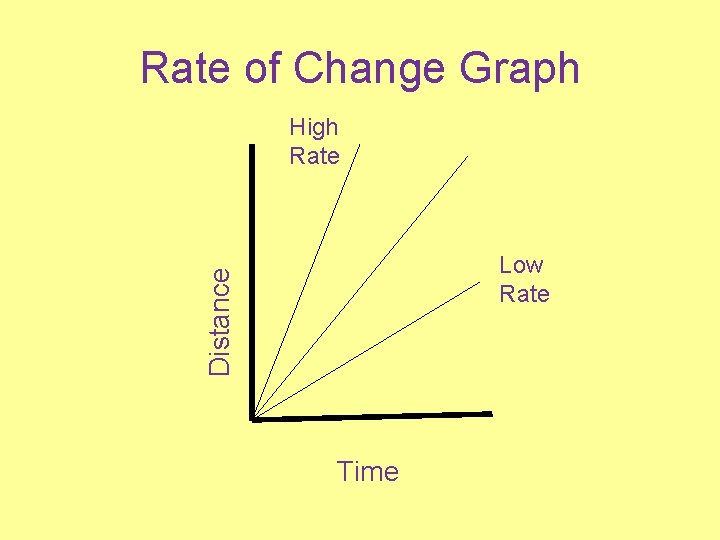
Rate of Change Graph High Rate Distance Low Rate Time

Energy & Change • Change occurs when there is energy flow. • Energy always flows from places with more to places with less. (From a source to a sink. ) Example: Energy flows from the Sun to Earth.

Interface • A boundary between areas with different properties. • The surface separating two different materials. Example: Earth’s surface, where your feet are, is the boundary between the lithosphere and atmosphere.
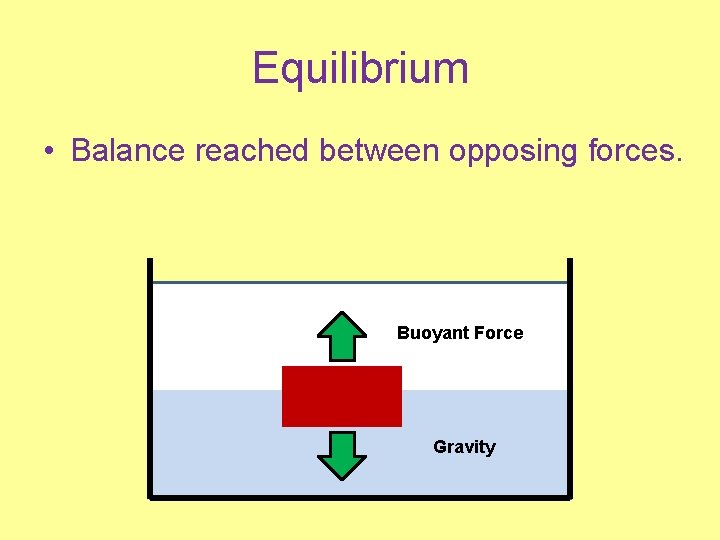
Equilibrium • Balance reached between opposing forces. Buoyant Force Gravity

Dynamic Equilibrium • A balance of changes in the environment. • A condition where constant change balances out.

Environmental Equilibrium • A balance of changes in the environment. • Small changes easy to balance. • Bigger changes are harder to balance. Examples: • A cold front passing lowers temperatures then they quickly go back to normal. • The start of an ice age is more difficult to normalize.

Pollution • Any adverse concentration of a material or energy. • Harms the environment by harming one or more populations within the environment. • Some have a natural origin and some are man- made.

Man-Made Pollution • • Industrial Waste Thermal Pollution Sound Pollution Light Pollution

Natural Pollution • • Pollen Dust Smoke Volcanic Gases
- Slides: 36