Objectives At the end of this talk you


Objectives At the end of this talk you will be able to: • Describe the rationale for extended duration (30 days) thromboprophylaxis in abdominopelvic surgical cancer patients • Assess the thrombotic risk and bleeding risk abdominopelvic surgical cancer patients • Describe the evidence and guidelines supporting the use of extended duration thromboprophylaxis in abdominopelvic surgical patients • Assess the thrombotic risk and bleeding risk in urology patients • Explain the rationale for the order set developed by Thrombosis Canada for cancer patients having undergone abdominopelvic surgery

Talk Overview 1. Introduction 2. Rationale for Extended Duration Thromboprophylaxis in abdominopelvic Surgical Cancer Patients 3. Assessing the Bleeding Risk and Thrombotic Risk 4. Review key evidence-based clinical data and guidelines related to VTE prophylaxis in abdominopelvic surgical cancer patients 5. Considerations for urology, renal failure and obesity 6. Review the Thrombosis Canada protocol/Order Set for thromboprophylaxis of abdominopelvic surgical cancer patients

Introduction: Virchow’s Triad Venous Stasis • Vascular compression by tumor • Prolonged bed rest • Hypotension Rudolph Virchow Vascular Injury • Direct invasion • Central catheters • Endothelial damage • Surgery • Chemotherapy Hypercoagulability • Tumor procoagulants • Cytokines • Impaired endothelial cell defense • Cellular interactions Adapted from Joist JH. Semin Thromb Hemost. 1990; 16: 151 -157.

Introduction: Rationale for Post Surgical Thromboprophylaxis • Pulmonary embolism (PE) is the most common preventable cause of hospital death and the number-one strategy to improve patient safety in hospitals • Approximately one third of the 150, 000 to 200, 000 VTE-related deaths per year in the United States are post-surgical • A vast number of randomized clinical trials over the past 30 years have confirmed benefit, safety and cost effectiveness • Thromboprophylaxis effectively reduces: DVT/PE, Fatal PE, Death from any cause and Health care management costs Geerts WH et al. Chest. 2008 Jun; 133(6 Suppl): 381 S-453 S Gould et al. Chest. 2012; 141 e 227 S-77 S. Horlander KT, Mannino DM, Leeper KV. Arch Intern Med. 2003 28; 163(14): 1711 -7.

Introduction: 9 th ACCP Guideline Recommendations for Standard Surgical Thromboprophylaxis • Patients undergoing general and abdominal pelvic surgery should receive a risk assessment before their procedure to predict their risk of VTE • For patients at moderate risk for VTE (Caprini Score of 3 -4) who are not at high risk for major bleeding complications, guidelines suggest LMWH, LDUH (Grade 2 B) for 7 -10 days or mechanical prophylaxis (Grade 2 C) • For patients at high risk for VTE (Caprini Score of 5 or more) who are not at high risk for major bleeding complications, guidelines recommend pharmacologic prophylaxis with LMWH or LDUH (Grade 1 B) for 7 -10 days Gould MK, et al. Chest 2012; 141; e 227 S-e 277 S

WHAT IS THE RATIONALE FOR EXTENDED DURATION (30 DAYS) THROMBOPROPHYLAXIS IN SURGICAL CANCER PATIENTS?
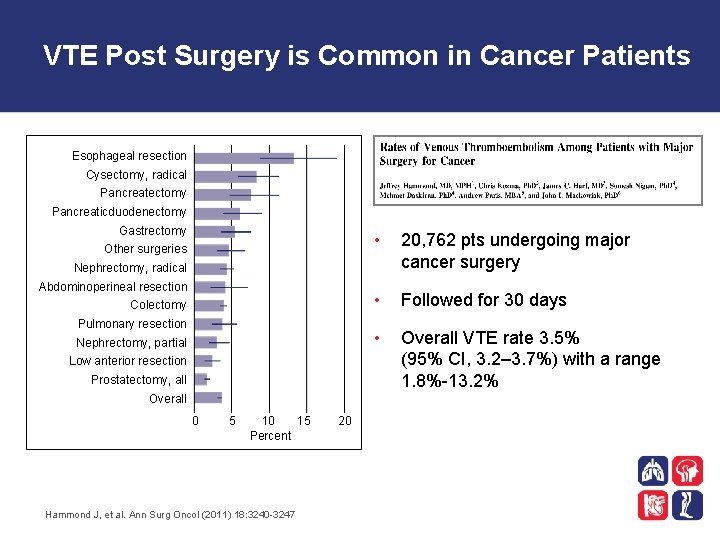
VTE Post Surgery is Common in Cancer Patients Esophageal resection Cysectomy, radical Pancreatectomy Pancreaticduodenectomy Gastrectomy Other surgeries • 20, 762 pts undergoing major cancer surgery • Followed for 30 days • Overall VTE rate 3. 5% (95% CI, 3. 2– 3. 7%) with a range 1. 8%-13. 2% Nephrectomy, radical Abdominoperineal resection Colectomy Pulmonary resection Nephrectomy, partial Low anterior resection Prostatectomy, all Overall 0 5 10 15 Percent Hammond J, et al. Ann Surg Oncol (2011) 18: 3240 -3247 20
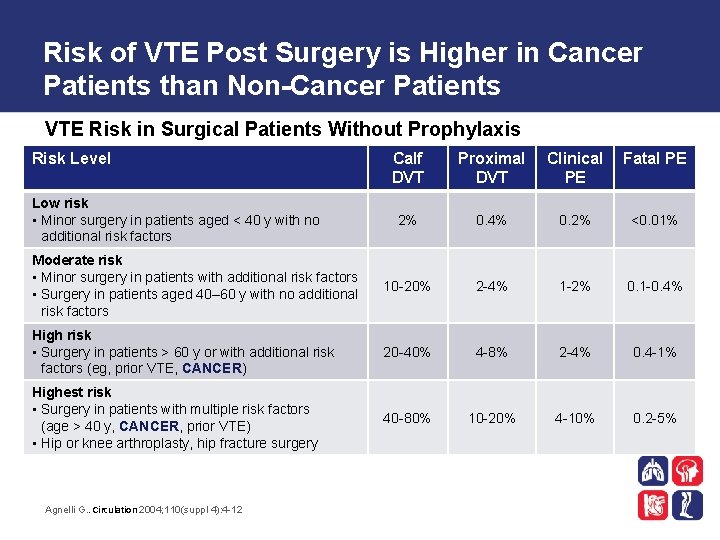
Risk of VTE Post Surgery is Higher in Cancer Patients than Non-Cancer Patients VTE Risk in Surgical Patients Without Prophylaxis Risk Level Calf DVT Proximal DVT Clinical PE Fatal PE 2% 0. 4% 0. 2% ˂0. 01% Moderate risk • Minor surgery in patients with additional risk factors • Surgery in patients aged 40– 60 y with no additional risk factors 10 -20% 2 -4% 1 -2% 0. 1 -0. 4% High risk • Surgery in patients ˃ 60 y or with additional risk factors (eg, prior VTE, CANCER) 20 -40% 4 -8% 2 -4% 0. 4 -1% Highest risk • Surgery in patients with multiple risk factors (age ˃ 40 y, CANCER, prior VTE) • Hip or knee arthroplasty, hip fracture surgery 40 -80% 10 -20% 4 -10% 0. 2 -5% Low risk • Minor surgery in patients aged ˂ 40 y with no additional risk factors Agnelli G. . Circulation 2004; 110(suppl 4): 4 -12
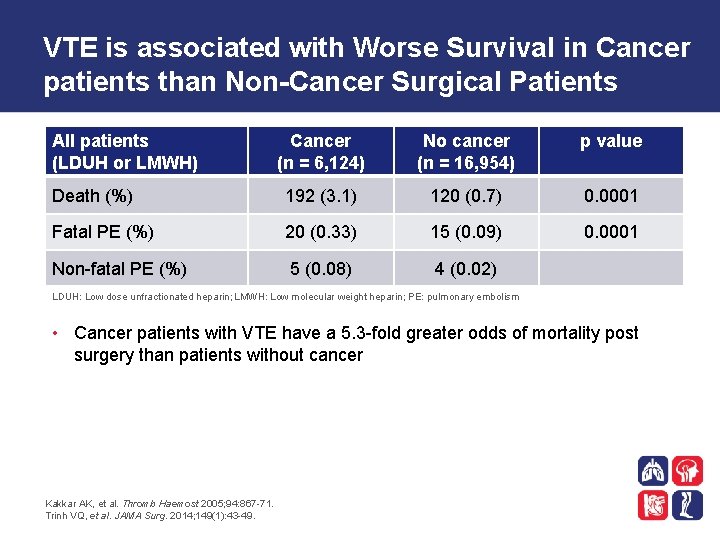
VTE is associated with Worse Survival in Cancer patients than Non-Cancer Surgical Patients All patients (LDUH or LMWH) Cancer (n = 6, 124) No cancer (n = 16, 954) p value Death (%) 192 (3. 1) 120 (0. 7) 0. 0001 Fatal PE (%) 20 (0. 33) 15 (0. 09) 0. 0001 Non-fatal PE (%) 5 (0. 08) 4 (0. 02) LDUH: Low dose unfractionated heparin; LMWH: Low molecular weight heparin; PE: pulmonary embolism • Cancer patients with VTE have a 5. 3 -fold greater odds of mortality post surgery than patients without cancer Kakkar AK, et al. Thromb Haemost 2005; 94: 867 -71. Trinh VQ, et al. JAMA Surg. 2014; 149(1): 43 -49.
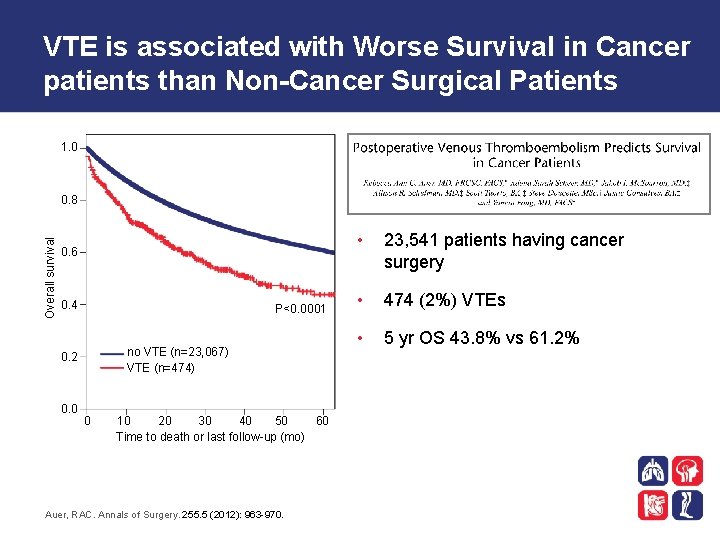
VTE is associated with Worse Survival in Cancer patients than Non-Cancer Surgical Patients 1. 0 Overall survival 0. 8 0. 6 0. 4 P<0. 0001 no VTE (n=23, 067) VTE (n=474) 0. 2 0. 0 0 10 20 30 40 50 60 Time to death or last follow-up (mo) Auer, RAC. Annals of Surgery. 255. 5 (2012): 963 -970. • 23, 541 patients having cancer surgery • 474 (2%) VTEs • 5 yr OS 43. 8% vs 61. 2%
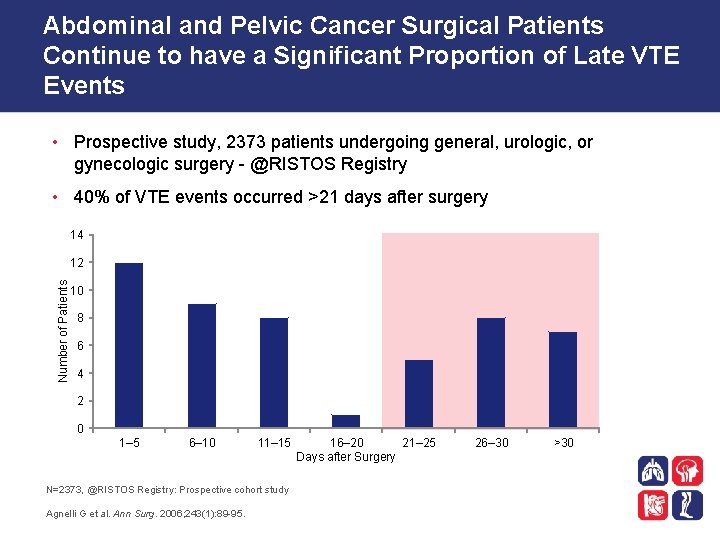
Abdominal and Pelvic Cancer Surgical Patients Continue to have a Significant Proportion of Late VTE Events • Prospective study, 2373 patients undergoing general, urologic, or gynecologic surgery - @RISTOS Registry • 40% of VTE events occurred >21 days after surgery 14 Number of Patients 12 10 8 6 4 2 0 1– 5 6– 10 11– 15 N=2373, @RISTOS Registry: Prospective cohort study Agnelli G et al. Ann Surg. 2006; 243(1): 89 -95. 16– 20 21– 25 Days after Surgery 26– 30 >30
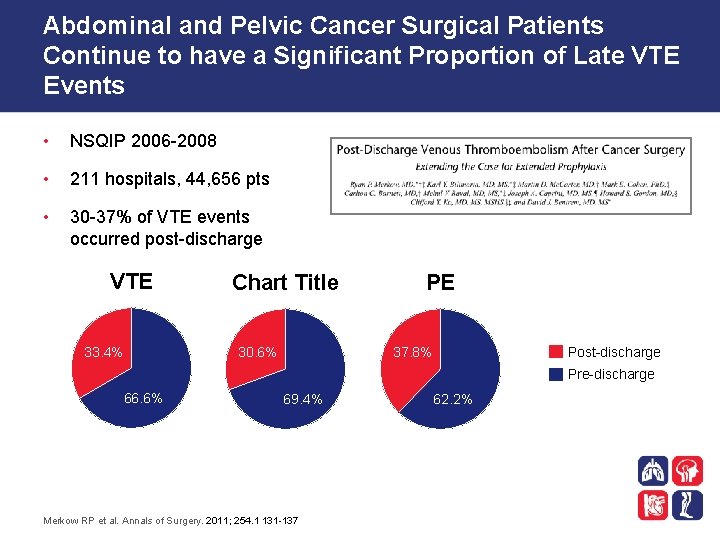
Abdominal and Pelvic Cancer Surgical Patients Continue to have a Significant Proportion of Late VTE Events • NSQIP 2006 -2008 • 211 hospitals, 44, 656 pts • 30 -37% of VTE events occurred post-discharge VTE 33. 4% Chart Title 30. 6% PE 37. 8% Post-discharge Pre-discharge 66. 6% 69. 4% Merkow RP et al. Annals of Surgery. 2011; 254. 1 131 -137 62. 2%

ASSESSING THE THROMBOTIC AND BLEEDING RISKS

Assessing the Thrombotic Risk Factors for Cancer-Associated VTE Cancer Related Treatment Related Patient related Biomarkers Primary site Chemotherapy Older Age Platelet count (≤ 350, 000/µL) Stage (higher for advanced stage) Antiangiogenic agents (eg, thalidomide, lenalidomide) Race (higher in African Americans; lower in Asians/Pacific Islanders) Leukocyte count (> 11, 000/µL) Cancer histology (higher for adenocarcinoma than squamous cell) Hormonal therapy Medical comorbidities (infection, renal disease, pulmonary disease, arterial thromboembolism) Hemoglobin (< 10 g/d. L) Time after initial diagnosis (highest in first 3 to 6 months) • Erythropoiesisstimulating agents • Transfusions • Indwelling venous access devices • Radiation therapy • Surgery > 60 min • Obesity • History of VTE • Diminished performance status • Inherited prothrombotic mutations VTE = venous thromboembolism Adapted from Lyman GH, et al. ASCO Update. J Clin Oncol. 2013; 31: 2189 -204.

Assessing the Thrombotic Risk Who is high risk? • High risk patients require thromboprophylaxis, but who is high risk? • Based on the randomized controlled trials in this setting, the indications for extended duration thromboprophylaxis in patients undergoing open abdominal or pelvic surgery for cancer (i. e. high risk patients) would include: - Patients over 40 years of age or older requiring admission or - Patients over the age of 40 whose surgery lasts longer than an 30 minutes to an hour - Patients with alternate risk factors for example between ages of 18 -40 with additional risk factors shown in previous slide. 1. 2. 3. 4. Bergqvist D, et al. N Engl J Med 2002; 346: 975 -80 Rasumssuen MS, et al. J Thromb Haemost 2006; 4: 2384 -90 Kakkar VV, et al. J Thromb Haemost 2010; 8: 1223 -9 Gould, MK, et al. . Chest 2012; 141(2_suppl): e 227 S-e 277 S
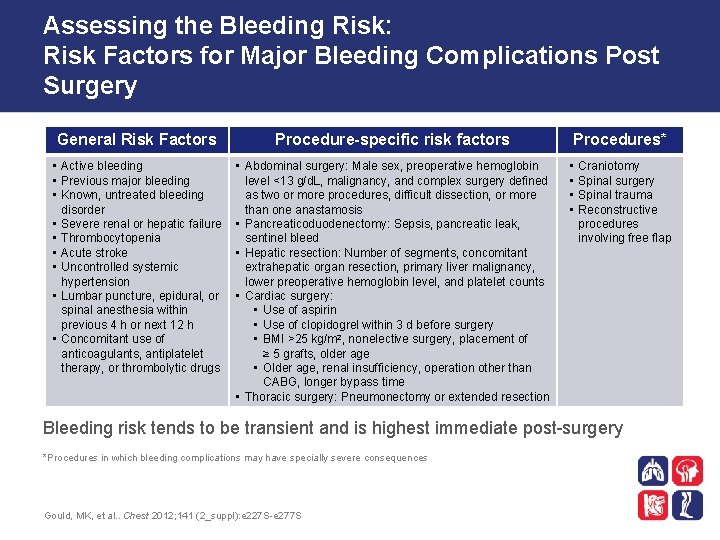
Assessing the Bleeding Risk: Risk Factors for Major Bleeding Complications Post Surgery General Risk Factors Procedure-specific risk factors • Active bleeding • Previous major bleeding • Known, untreated bleeding disorder • Severe renal or hepatic failure • Thrombocytopenia • Acute stroke • Uncontrolled systemic hypertension • Lumbar puncture, epidural, or spinal anesthesia within previous 4 h or next 12 h • Concomitant use of anticoagulants, antiplatelet therapy, or thrombolytic drugs • Abdominal surgery: Male sex, preoperative hemoglobin level ˂13 g/d. L, malignancy, and complex surgery defined as two or more procedures, difficult dissection, or more than one anastamosis • Pancreaticoduodenectomy: Sepsis, pancreatic leak, sentinel bleed • Hepatic resection: Number of segments, concomitant extrahepatic organ resection, primary liver malignancy, lower preoperative hemoglobin level, and platelet counts • Cardiac surgery: • Use of aspirin • Use of clopidogrel within 3 d before surgery • BMI >25 kg/m 2, nonelective surgery, placement of ≥ 5 grafts, older age • Older age, renal insufficiency, operation other than CABG, longer bypass time • Thoracic surgery: Pneumonectomy or extended resection Procedures* • • Craniotomy Spinal surgery Spinal trauma Reconstructive procedures involving free flap Bleeding risk tends to be transient and is highest immediate post-surgery *Procedures in which bleeding complications may have specially severe consequences Gould, MK, et al. . Chest 2012; 141 (2_suppl): e 227 S-e 277 S

WHAT IS THE EVIDENCE TO SUPPORT EXTENDED-DURATION (30 -DAYS) ANTICOAGULANT PROPHYLAXIS AFTER ABDOMINAL & PELVIC CANCER SURGERY?
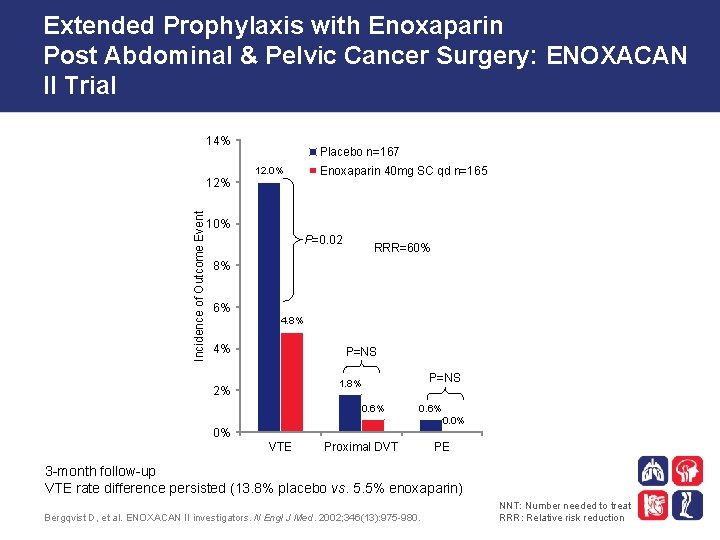
Extended Prophylaxis with Enoxaparin Post Abdominal & Pelvic Cancer Surgery: ENOXACAN II Trial 14% Incidence of Outcome Event 12% Placebo n=167 12. 0% Enoxaparin 40 mg SC qd n=165 10% P=0. 02 RRR=60% 8% 6% 4. 8% 4% P=NS 1. 8% 2% 0. 6% 0. 0% 0% VTE Proximal DVT PE 3 -month follow-up VTE rate difference persisted (13. 8% placebo vs. 5. 5% enoxaparin) Bergqvist D, et al. ENOXACAN II investigators. N Engl J Med. 2002; 346(13): 975 -980. NNT: Number needed to treat RRR: Relative risk reduction
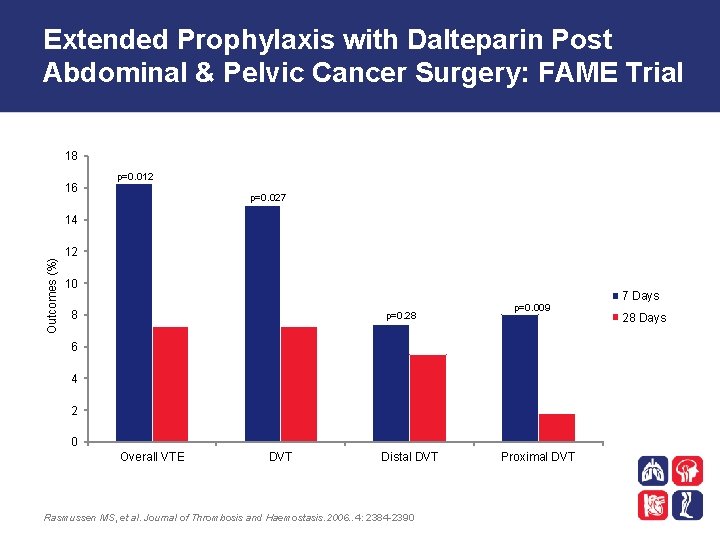
Extended Prophylaxis with Dalteparin Post Abdominal & Pelvic Cancer Surgery: FAME Trial 18 16 p=0. 012 p=0. 027 Outcomes (%) 14 12 10 8 p=0. 28 p=0. 009 6 4 2 0 Overall VTE DVT Distal DVT Rasmussen MS, et al. Journal of Thrombosis and Haemostasis. 2006. . 4: 2384 -2390 Proximal DVT 7 Days 28 Days
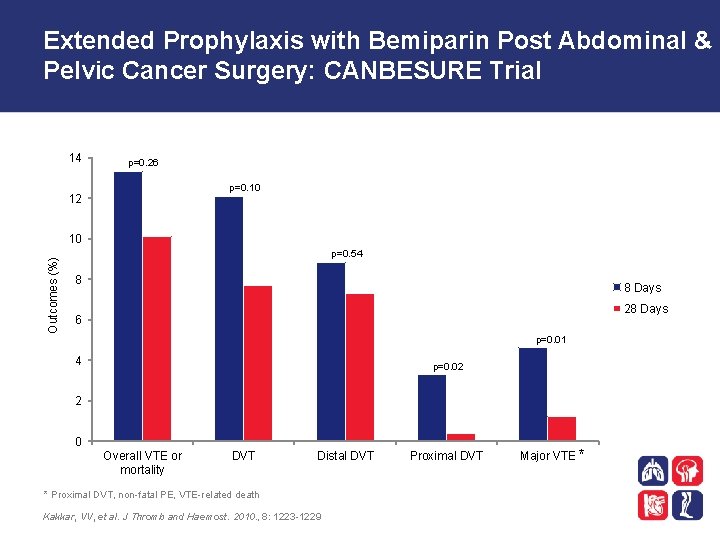
Extended Prophylaxis with Bemiparin Post Abdominal & Pelvic Cancer Surgery: CANBESURE Trial 14 p=0. 26 p=0. 10 12 Outcomes (%) 10 p=0. 54 8 8 Days 28 Days 6 p=0. 01 4 p=0. 02 2 0 Overall VTE or mortality DVT Distal DVT * Proximal DVT, non-fatal PE, VTE-related death Kakkar, VV, et al. J Thromb and Haemost. 2010. , 8: 1223 -1229 Proximal DVT Major VTE *
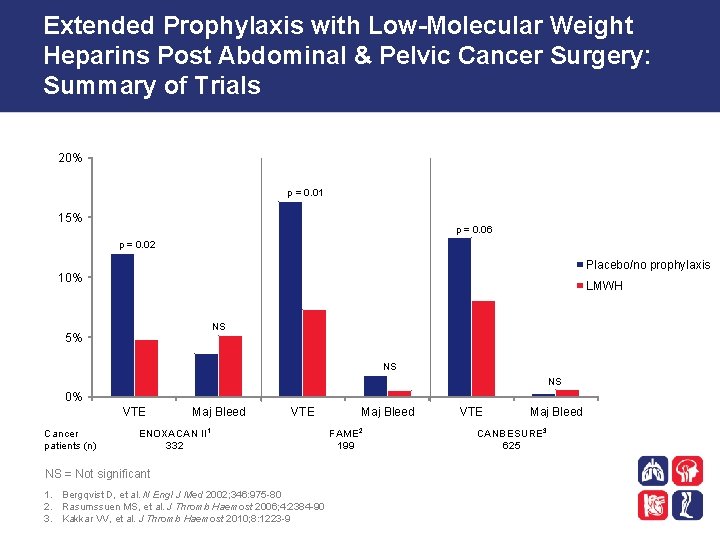
Extended Prophylaxis with Low-Molecular Weight Heparins Post Abdominal & Pelvic Cancer Surgery: Summary of Trials 20% p = 0. 01 15% p = 0. 06 p = 0. 02 Placebo/no prophylaxis 10% LMWH NS 5% NS NS 0% VTE Cancer patients (n) Maj Bleed VTE ENOXACAN II 1 332 NS = Not significant 1. Bergqvist D, et al. N Engl J Med 2002; 346: 975 -80 2. Rasumssuen MS, et al. J Thromb Haemost 2006; 4: 2384 -90 3. Kakkar VV, et al. J Thromb Haemost 2010; 8: 1223 -9 Maj Bleed FAME 2 199 VTE Maj Bleed CANBESURE 3 625
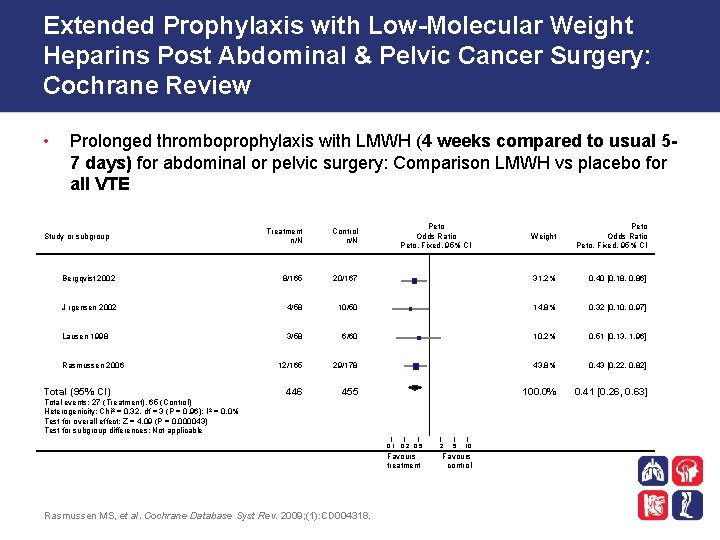
Extended Prophylaxis with Low-Molecular Weight Heparins Post Abdominal & Pelvic Cancer Surgery: Cochrane Review • Prolonged thromboprophylaxis with LMWH (4 weeks compared to usual 57 days) for abdominal or pelvic surgery: Comparison LMWH vs placebo for all VTE Weight Peto Odds Ratio Peto, Fixed, 95% CI 20/167 31. 2% 0. 40 [0. 18, 0. 86] 4/58 10/50 14. 8% 0. 32 [0. 10, 0. 97] 3/58 6/60 10. 2% 0. 51 [0. 13, 1. 96] 12/165 29/178 43. 8% 0. 43 [0. 22, 0. 82] 446 455 100. 0% 0. 41 [0. 26, 0. 63] Treatment n/N Control n/N Bergqvist 2002 8/165 J rgensen 2002 Lausen 1998 Study or subgroup Rasmussen 2006 Total (95% CI) Peto Odds Ratio Peto, Fixed, 95% CI Total events: 27 (Treatment), 65 (Control) Heterogenicity: Chi 2 = 0. 32, df = 3 (P = 0. 96); I 2 = 0. 0% Test for overall effect: Z = 4. 09 (P = 0. 000043) Test for subgroup differences: Not applicable 0. 1 0. 2 0. 5 Favours treatment Rasmussen MS, et al. Cochrane Database Syst Rev. 2009; (1): CD 004318. 2 5 10 Favours control
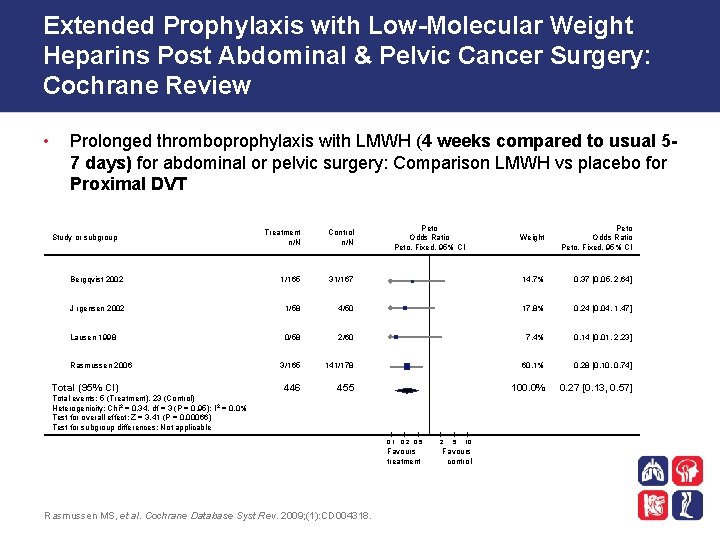
Extended Prophylaxis with Low-Molecular Weight Heparins Post Abdominal & Pelvic Cancer Surgery: Cochrane Review • Prolonged thromboprophylaxis with LMWH (4 weeks compared to usual 57 days) for abdominal or pelvic surgery: Comparison LMWH vs placebo for Proximal DVT Weight Peto Odds Ratio Peto, Fixed, 95% CI 31/167 14. 7% 0. 37 [0. 05, 2. 64] 1/58 4/50 17. 8% 0. 24 [0. 04, 1. 47] 0/58 2/60 7. 4% 0. 14 [0. 01, 2. 23] 3/165 141/178 60. 1% 0. 28 [0. 10, 0. 74] 446 455 100. 0% 0. 27 [0. 13, 0. 57] Treatment n/N Control n/N Bergqvist 2002 1/165 J rgensen 2002 Lausen 1998 Study or subgroup Rasmussen 2006 Total (95% CI) Peto Odds Ratio Peto, Fixed, 95% CI Total events: 5 (Treatment), 23 (Control) Heterogenicity: Chi 2 = 0. 34, df = 3 (P = 0. 95); I 2 = 0. 0% Test for overall effect: Z = 3. 41 (P = 0. 00066) Test for subgroup differences: Not applicable 0. 1 0. 2 0. 5 Favours treatment Rasmussen MS, et al. Cochrane Database Syst Rev. 2009; (1): CD 004318. 2 5 10 Favours control
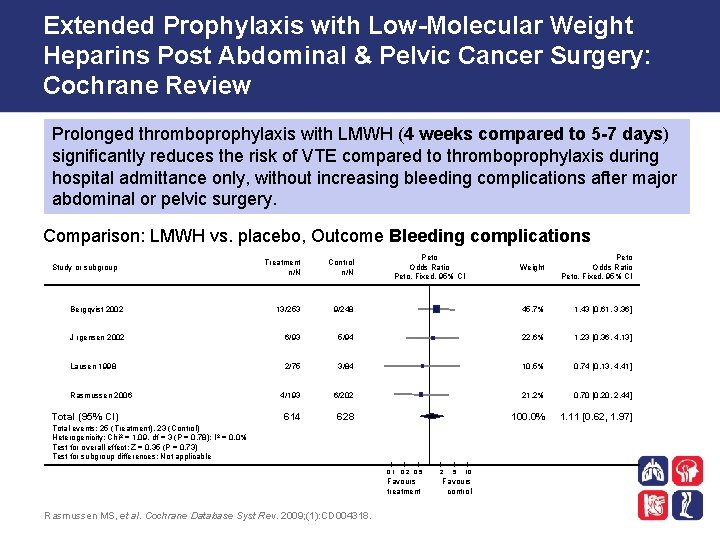
Extended Prophylaxis with Low-Molecular Weight Heparins Post Abdominal & Pelvic Cancer Surgery: Cochrane Review Prolonged thromboprophylaxis with LMWH (4 weeks compared to 5 -7 days) significantly reduces the risk of VTE compared to thromboprophylaxis during hospital admittance only, without increasing bleeding complications after major abdominal or pelvic surgery. Comparison: LMWH vs. placebo, Outcome Bleeding complications Weight Peto Odds Ratio Peto, Fixed, 95% CI 9/248 45. 7% 1. 43 [0. 61, 3. 36] 6/93 5/94 22. 6% 1. 23 [0. 36, 4. 13] 2/75 3/84 10. 5% 0. 74 [0. 13, 4. 41] 4/193 6/202 21. 2% 0. 70 [0. 20, 2. 44] 614 628 100. 0% 1. 11 [0. 62, 1. 97] Treatment n/N Control n/N Bergqvist 2002 13/253 J rgensen 2002 Lausen 1998 Study or subgroup Rasmussen 2006 Total (95% CI) Peto Odds Ratio Peto, Fixed, 95% CI Total events: 25 (Treatment), 23 (Control) Heterogenicity: Chi 2 = 1. 09, df = 3 (P = 0. 78); I 2 = 0. 0% Test for overall effect: Z = 0. 35 (P = 0. 73) Test for subgroup differences: Not applicable 0. 1 0. 2 0. 5 Favours treatment Rasmussen MS, et al. Cochrane Database Syst Rev. 2009; (1): CD 004318. 2 5 10 Favours control

WHAT DO THE GUIDELINES SAY ABOUT EXTENDED-DURATION (30 -DAYS) ANTICOAGULANT PROPHYLAXIS AFTER CANCER SURGERY?

Guideline Recommendations for SURGICAL VTE Prophylaxis ASCO 2013 Pharmacological thromboprophylaxis to all patients with malignant disease undergoing major surgical interventions • Prophylaxis should be commenced preoperatively, should be continued for at least 7 to 10 days. • Extended prophylaxis with LMWH for up to 4 weeks postoperatively should be considered for patients undergoing major abdominal or pelvic surgery for cancer who have high-risk features such as restricted mobility, obesity, history of VTE, or with additional risk factors NCCN Out-of-hospital primary VTE prophylaxis is recommended for up to 4 weeks postoperatively (particularly for high-risk abdominal or pelvic cancer surgery patients ) ESMO 2011 Pharmacological thromboprophylaxis to all cancer patients undergoing major cancer surgery • patients having a laparotomy, laparoscopy, thoracotomy or thoracoscopy lasting >than 30 min, consider s. c. LMWH for at least 10 days postoperatively. • Cancer patients undergoing elective major abdominal or pelvic surgery should receive in hospital and post-discharge prophylaxis with s. c. LMWH for up to 1 month after surgery ACCP 2012 General and abdominal-pelvic surgery patients for cancer • High risk for VTE/not at high bleeding risk → extended duration, 4 weeks , prophylaxis with LMWH • LDUH, LMWH or mechanical prophylaxis No good quality studies have been done using unfractionated heparin for extended duration thromboprophylaxis Lyman GH, et al. J Clin Oncol. 2013; 31: 2189 -204 Streiff MB, et al. JNCCN 2011; 9: 714– 777 Madnala M, et al. Annals Oncology 2011; 22 (Supplement 6): vi 85–vi 92 Gould, MK, et al. 9 th Chest. 2012; 141(2_suppl): e 227 S-e 277 S

WHAT ANTICOAGULANT REGIMENS AND DOSES ARE APPROVED IN CANADA FOR DVT PROPHYLAXIS AFTER SURGERY?

Dosing Regimens for Extended Duration Thromboprophylaxis VTE in Abdomino-Pelvic Cancer Surgical Patients – Canadian Labeling Drug Regimen Dalteparin 2500 U 2 -4 hours preoperatively and 5000 U once daily thereafter or 5000 U 10 -12 hours preoperatively and 5000 U once daily thereafter Enoxaparin 20 mg 2 -4 hours preoperatively and 40 mg once daily thereafter or 40 mg 10 -12 hours preoperatively and 40 mg once daily thereafter Tinzaparin 3500 IU SC 2 hours before surgery followed by 4500 IU once daily Fragmin (dalteprain) Product Monograph; Jan. 6, 2014. Lovenox (enoxaparin) Product Monograph; Dec. 20, 2013. Innohep (tinzaparin) Product Monograph; Feb. 3, 2011.

CONSIDERATIONS IN UROLOGY PATIENTS

VTE in Patients Undergoing Urological Procedures VTE is • Among the most common causes of non-surgical death in patients undergoing urologic surgery • Preventable • DVT prophylaxis has been identified by a number of organizations as a marker of good quality of patient care Quek ML, et al. J Urol 2006; 175: 886 -90 Forrest JB, et al. J Urol 2009; 181: 1170 -7 DVT = Deep vein thrombosis

Risk of VTE With Radical Prostatectomy in the SEER Database: 1994– 2009 • 35, 104 patients with nonmetastatic prostate cancer who underwent radical prostatectomy • 30 days after radical prostatectomy : - 931 (2. 7%) VTEs - 87 (0. 25%) deaths • 90 days after radical prostatectomy : - 1, 112 (3. 2%) VTEs - 121 (0. 3%) deaths

Risk of Major Bleeding Following Urologic Surgery • The risk of major bleeding in urologic surgery is derived from the RCTs where urologic surgery was included as part of all abdominal-pelvic surgeries where the bleeding risk was shown to be low as in the previous slides. 1. Bergqvist D, et al. N Engl J Med 2002; 346: 975 -80 2. Rasumssuen MS, et al. J Thromb Haemost 2006; 4: 2384 -90 3. VV, SLIDE et al. J 22 Thromb Haemost 2010; 8: 1223 -9 1. Kakkar SAME AS Tikkinen KA, et al. Syst Rev 2014; 3: 150

Timing of Major Bleeding and VTE Following All Urologic Surgeries • All urologic surgeries were assessed • The risk of bleeding in urology surgery is transient, within the first few days, however the risk of VTE is progressive over a 3 month period - see following 2 slides Tikkinen KA, et al. Syst Rev 2014; 3: 150
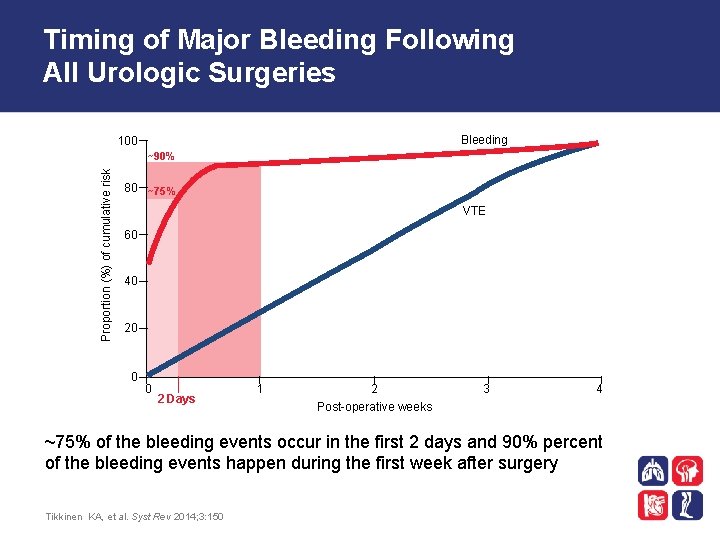
Timing of Major Bleeding Following All Urologic Surgeries Bleeding 100 Proportion (%) of cumulative risk ~90% 80 ~75% VTE 60 40 20 0 0 2 Days 1 2 3 4 Post-operative weeks ~75% of the bleeding events occur in the first 2 days and 90% percent of the bleeding events happen during the first week after surgery Tikkinen KA, et al. Syst Rev 2014; 3: 150
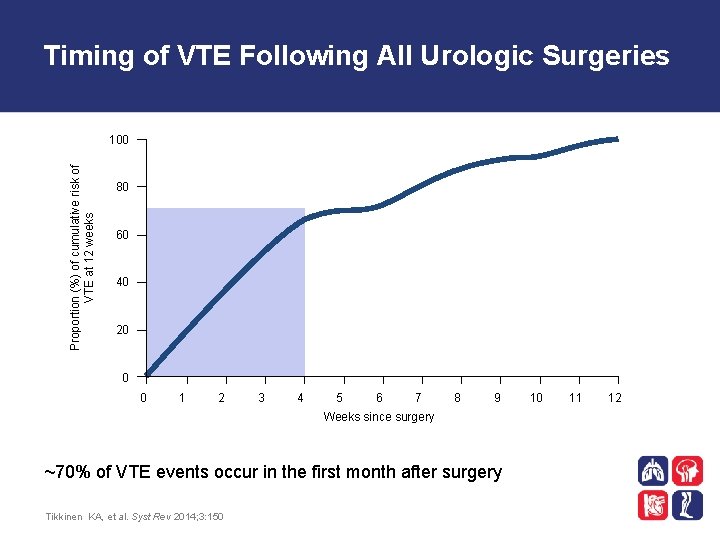
Timing of VTE Following All Urologic Surgeries Proportion (%) of cumulative risk of VTE at 12 weeks 100 80 60 40 20 0 0 1 2 3 4 5 6 7 8 9 Weeks since surgery ~70% of VTE events occur in the first month after surgery Tikkinen KA, et al. Syst Rev 2014; 3: 150 10 11 12

European Association of Urology (EAU) Thromboprophylaxis Guideline Recommendations • The EAU compiled some thromboprophylaxis guidelines published in 2017 • The panel was made up of urologists, internists, hematologists and clinical epidemiologists • They reviewed the risks of bleeding and venous thromboembolism after a urologic surgery and came up with recommendations • The recommendations for open cancer urologic surgery are outlined here n KAO, et al. uroweb. org/guideline/thromboprophylaxis. March 2017
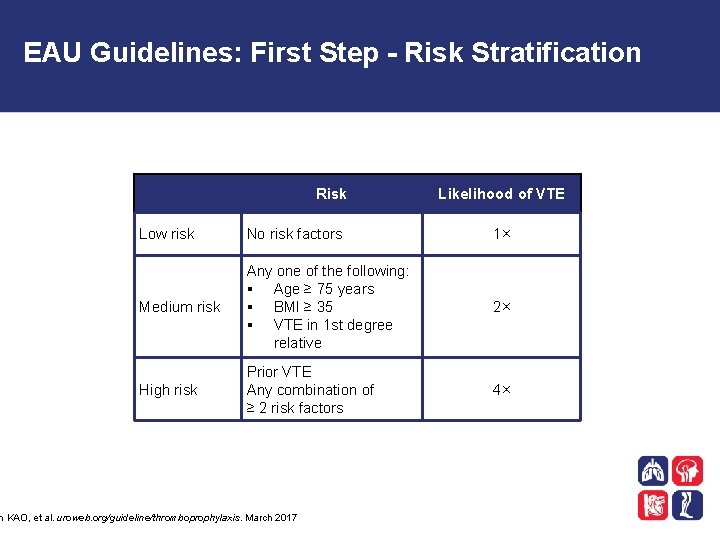
EAU Guidelines: First Step - Risk Stratification Risk Likelihood of VTE Low risk No risk factors 1× Medium risk Any one of the following: § Age ≥ 75 years § BMI ≥ 35 § VTE in 1 st degree relative 2× High risk Prior VTE Any combination of ≥ 2 risk factors 4× n KAO, et al. uroweb. org/guideline/thromboprophylaxis. March 2017
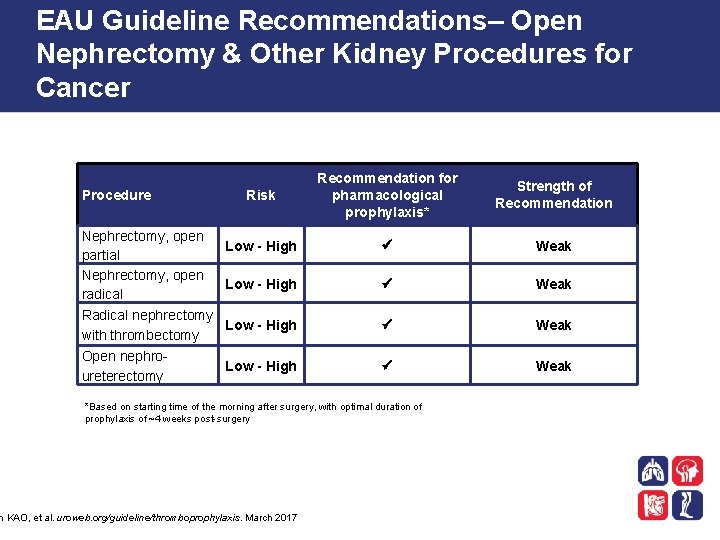
EAU Guideline Recommendations– Open Nephrectomy & Other Kidney Procedures for Cancer Risk Recommendation for pharmacological prophylaxis* Strength of Recommendation Low - High Weak Radical nephrectomy Low - High with thrombectomy Weak Open nephroureterectomy Weak Procedure Nephrectomy, open partial Nephrectomy, open radical Low - High *Based on starting time of the morning after surgery, with optimal duration of prophylaxis of ~4 weeks post-surgery n KAO, et al. uroweb. org/guideline/thromboprophylaxis. March 2017
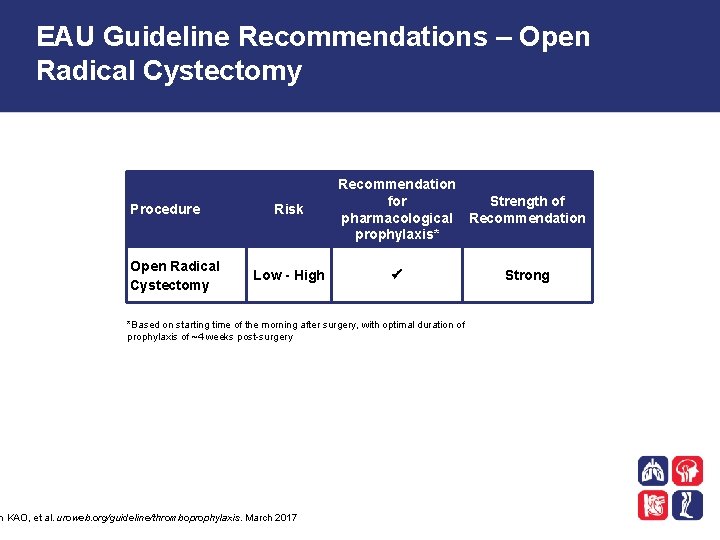
EAU Guideline Recommendations – Open Radical Cystectomy Procedure Open Radical Cystectomy Risk Low - High Recommendation for Strength of pharmacological Recommendation prophylaxis* *Based on starting time of the morning after surgery, with optimal duration of prophylaxis of ~4 weeks post-surgery n KAO, et al. uroweb. org/guideline/thromboprophylaxis. March 2017 Strong
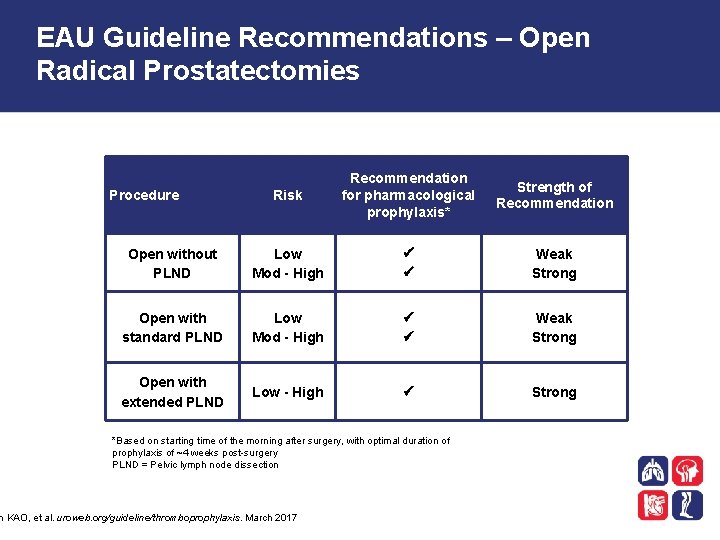
EAU Guideline Recommendations – Open Radical Prostatectomies Risk Recommendation for pharmacological prophylaxis* Strength of Recommendation Open without PLND Low Mod - High Weak Strong Open with standard PLND Low Mod - High Weak Strong Open with extended PLND Low - High Strong Procedure *Based on starting time of the morning after surgery, with optimal duration of prophylaxis of ~4 weeks post-surgery PLND = Pelvic lymph node dissection n KAO, et al. uroweb. org/guideline/thromboprophylaxis. March 2017

EAU Guideline Recommendations – Open Primary Nerve-sparing Retroperitoneal Lymph Node Dissection Procedure Primary Nervesparing Retroperitoneal Lymph Node Dissection Risk Low - High Recommendation for Strength of pharmacological Recommendation prophylaxis* *Based on starting time of the morning after surgery, with optimal duration of prophylaxis of ~4 weeks post-surgery n KAO, et al. uroweb. org/guideline/thromboprophylaxis. March 2017 Weak

EAU Guideline Recommendations • For open urologic cancer surgery there were no deviations from the ACCP guidelines or any other major guidelines that thromboprophylaxis for four weeks with low-molecular-weight heparin is required after surgery • Thromboprophylaxis for other urologic procedures was also addressed this guideline, but would be empiric n KAO, et al. uroweb. org/guideline/thromboprophylaxis. March 2017

ADDITIONAL CONSIDERATIONS REFLECTED ON THE EXTEND ORDER SET: RENAL IMPAIRMENT, OBESITY & CONCOMITANT MEDICATIONS
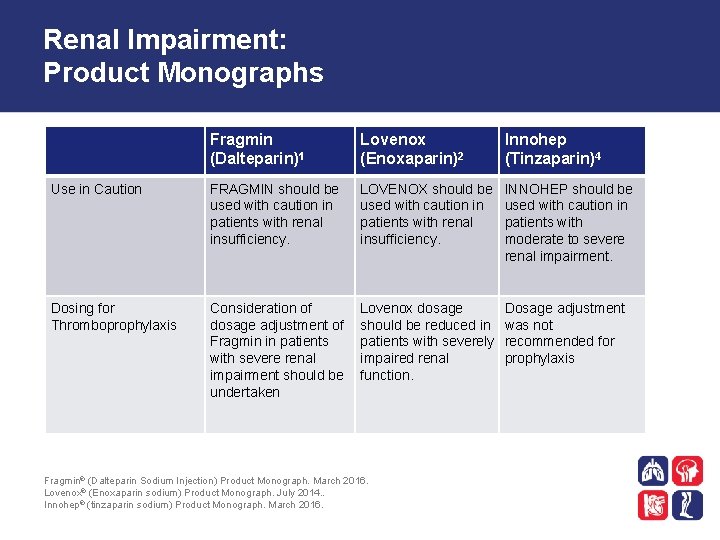
Renal Impairment: Product Monographs Fragmin (Dalteparin)1 Lovenox (Enoxaparin)2 Innohep (Tinzaparin)4 Use in Caution FRAGMIN should be used with caution in patients with renal insufficiency. LOVENOX should be used with caution in patients with renal insufficiency. INNOHEP should be used with caution in patients with moderate to severe renal impairment. Dosing for Thromboprophylaxis Consideration of dosage adjustment of Fragmin in patients with severe renal impairment should be undertaken Lovenox dosage should be reduced in patients with severely impaired renal function. Dosage adjustment was not recommended for prophylaxis Fragmin® (Dalteparin Sodium Injection) Product Monograph. March 2016. Lovenox® (Enoxaparin sodium) Product Monograph. July 2014. . Innohep® (tinzaparin sodium) Product Monograph. March 2016.

Obesity: Altered Drug Distribution and Metabolism in Obese Patients • Obesity is an independent risk factor for VTE • Bariatric surgery is an effective weight loss strategy in the morbidly obese (BMI > 40 kg/m 2) and can significantly improve health outcomes • Data on dosing for thromboprophylaxis in obesity is not of high quality • Obesity has been associated with increased clearance and altered volume of distribution of low-molecular-weight heparins - Functional and structural renal changes and glomerular hyperfiltration in obesity increases glomerular filtration by up to 51% and renal plasma flow by up to 31% Patel JP, et al. Br J Haematol. 2011; 155: 137 -49

Obesity: Altered Dosing May be Required in Obese Patients • Pharmacokinetic study • 28 morbidly obese (BMI ≥ 35 kg/m 2), medically ill patients • Enoxaparin 0. 5 mg/kg sc once daily • Peak anti-Xa levels measured ~4– 6 hours after each dose - Average daily dose 67 mg - Average peak anti-Xa level 0. 25 units/m. L • Peak anti-Xa levels did not significantly correlate with weight or BMI • No bleeding events, symptomatic VTE, or significant thrombocytopenia • Increased dosing in the obese was associated with no increase in bleeding events and no symptomatic VTE, suggested that their weight-based regimen is safe and effective Rondina MT, et al. Thromb Res 2010; 125: 220 -3.

Concomitant Medications • There is no good data to show that antiplatelet therapy provides thromboprophylaxis for this group of patients post surgery • The use of aspirin, clopidogrel or other non-steroidal anti-inflammatory drugs post-surgery is not a contraindication to the use of standard thromboprophylaxis Gould, MK, et al. . Chest 2012; 141(2_suppl): e 227 S-e 277 S

PRACTICAL CONSIDERATIONS

Practicalities of Implementing Extended Duration Thromboprophylaxis Discharge Pathway: How does the patient get coverage of the drug? • Private Insurance or • Government coverage (by a Government Drug Card) or • Compassionate coverage or • If a patient does not qualify for any of the above then they pay out of pocket

Practicalities of Implementing Extended Duration Thromboprophylaxis Discharge Pathway: How does the patient administer the drug? • The patient is taught to inject • If the patient is unwilling/unable to inject, then a family member is taught • If patient and family are unable to, then a referral is sent to the community nurse

Extended Thromboprophylaxis after Abdomino. Pelvic Surgery Patient Order set This order set was designed for the point of discharge, but may also be used immediately post-op

Extended Thromboprophylaxis after Abdomino. Pelvic Surgery Patient Order set

Extended Thromboprophylaxis after Abdomino. Pelvic Surgery Patient Order set
Entry. Point View

Patient Handout

Clinical Case • A 65 yr old obese man has stage 1 adenocarcinoma of the colon • His weight is 125 kg • He has a WCC of 15, Hemoglobin of 95, MCV of 76 and platelets of 490 • He undergoes a 1 hr 20 min laparotomy surgery and the surgeon and pathologist are confident that the entire mass was resected • He is admitted to the floor and is expected to remain admitted for 5 days • He has a low risk of bleeding • He is on metformin and aspirin 81 mg orally daily for diabetes mellitus type II

Clinical Case Would he be a candidate for thromboprophylaxis with low-molecular weight heparin? A. Yes B. No C. Don’t know

Clinical Case Would he be a candidate for thromboprophylaxis with low-molecular weight heparin? A. Yes B. No C. Don’t know A. Yes. Because he is admitted, he would require thromboprophylaxis. His other indication would be a patient over the age of 40 yrs with surgery lasting > 1 hour

Clinical Case You decide that he will receive thromboprophylaxis with enoxaparin. What dose should he receive? A. 30 mg s. c. daily B. 40 mg s. c. daily C. 60 mg s. c. daily D. 40 mg s. c. 2 x daily E. 120 mg s. c. daily

Clinical Case You decide that he will receive thromboprophylaxis with enoxaparin. What dose should he receive? A. 30 mg s. c. daily B. 40 mg s. c. daily C. 60 mg s. c. daily D. 40 mg s. c. 2 x daily E. 120 mg s. c. daily Because of his weight of 125 kg, his enoxaparin dose could be increased to: C. 60 mg (or up to 80 mg) sc once daily OR D. up to 40 mg twice daily

Clinical Case For how long should he receive thromboprophylaxis? A. 5 days B. 7 days C. 10 days D. Till discharge E. 28 days

Clinical Case For how long should he receive thromboprophylaxis? A. 5 days B. 7 days C. 10 days D. Till discharge E. 28 days E. 28 days. Because he has had open abdominal surgery for cancer, he requires 28 days of thromboprophylaxis with a low-molecular-weight heparin despite being on aspirin.

A Canadian Resource…
Concise Clinical Guides
Free Mobile App

Summary: Extended Duration Thromboprophylaxis VTE in Abdomino-Pelvic Cancer Surgical Patients • VTE Post Surgery is More Common in Cancer Patients than Non. Cancer Patients • VTE is associated with Worse Survival in Cancer patients than Non. Cancer Surgical Patients • Cancer Surgical Patients Continue to have a Significant Proportion of Late VTE Events • When present the bleeding risk is usually transient over days but the thrombotic risk persists up to 3 months

Summary: Extended Duration Thromboprophylaxis VTE in Abdomino-Pelvic Cancer Surgical Patients • Extended-duration (30 -days) anticoagulant prophylaxis with a lowmolecular-weight heparin after Abdominal & Pelvic cancer surgery in high risk patients at low bleeding risk has been shown to be effective and safe • Extended-duration (30 -days) anticoagulant prophylaxis with a lowmolecular-weight heparin after Abdominal & Pelvic cancer surgery in high risk patients at low bleeding risk has been recommended by the leading guideline bodies on thromboprophylaxis

Summary: Extended Duration Thromboprophylaxis VTE in Abdomino-Pelvic Cancer Surgical Patients • Urologic Surgery cancer patients are also at increased risk of venous thromboembolism extended-duration thromboprophylaxis with a lowmolecular-weight heparin has been shown to be safe • Reduced dosing is generally recommended for patients with renal failure • Increased dosing may be necessary for obese patients • Order sets/protocols ensure that a large number of patients receive an intervention that is appropriate to that patient
- Slides: 69