OBJECTIVES 1 Nomenclature of nucleic acids a nucleosides


OBJECTIVES: 1. Nomenclature of nucleic acids: a. nucleosides* b. nucleotides 2. Structure and function of purines and pyrimidines. 3. Origin of atoms in the purine ring and in the pyrimidine ring. 4. Essential features of purine and pyrimidine metabolism (anabolism and catabolism). 5. Diseases associated with metabolic malfunction. *Keywords are highlighted in yellow

Chemical compound composed of three components: (1) heterocyclic base; (2) sugar (pentose; ribose); and (3) one or more phosphate groups Glycosidic bond Base Phosphate Pentose sugar Adenosine monophosphate (AMP)

RNA is sensitive to alkaline degradation Base Ribonucleoside Ribonucleotide Deoxyribonucleotide Adenine Adenosine Adenylate Deoxyadenylate Guanine Guanosine Guanylate Deoxyguanylate Cytosine Cytidylate Deoxycytidylate Thymine Thymidine Ribothymidylate Thymidylate Uracil Uridine Uridylate Deoxyuridylate Hypoxanthine Inosinate Deoxyinosinate Xanthine Xanthosine Xanthylate Deoxanthylate


The Nitrogenous Bases In DNA: Adenine Guanine *Thymine* Cytosine In RNA: Adenine Guanine *Uracil* Cytosine
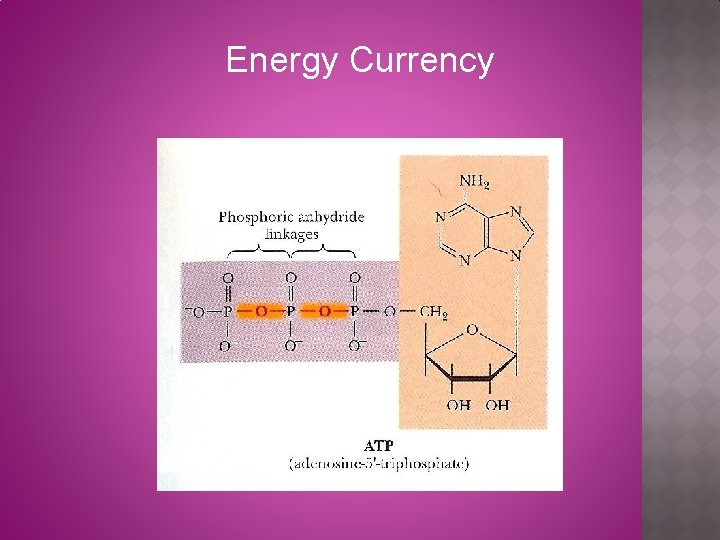
Energy Currency

Carriers for Activated Intermediates
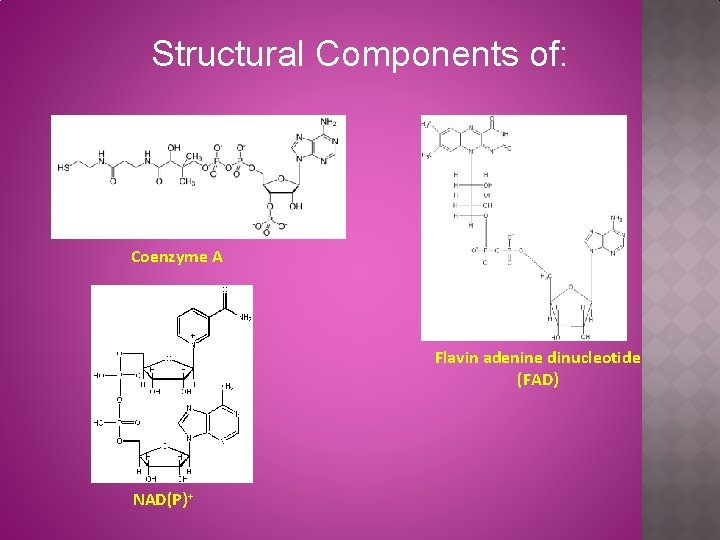
Structural Components of: Coenzyme A Flavin adenine dinucleotide (FAD) NAD(P)+

Signaling Molecules

Important metabolic intermediates; not typically found in either DNA or RNA. Hypoxanthine Xanthine


� Two ways: �De Novo Pathway: means from scratch; nucleotide bases are produced from simpler compounds Purines: base is synthesized in segments, in order, directly onto the ribose structure Pyrimidines: base is synthesized first and then assembled onto the ribose structure �Salvage Pathway: “a process whereby a metabolite is reutilized for biosynthesis of a compound from which the metabolite was derived”

De novo purine synthesis AMP IMP GMP ADP Adenosine monophosphate kinase GDP Guanosine monophosphate kinase Adenosine diphosphate kinase Guanosine diphosphate kinase ATP GTP De novo pyrimidine synthesis UMP Uridine monophosphate kinase d. UMP UDP Uridine diphosphate kinase d. UDP UTP CTP Thymidylate synthase d. TMP d. TDP Thymidine monophosphate kinase Thymidine diphosphate kinase d. TTP


� Purine ring: synthesized by a series of 12 reactions; carbon and nitrogen atoms added to a pre-formed ribose-5 -phosphate. � Ribose-5 -phosphate: Hexose Mono. Phosphate Pathway. � In humans: enzymes found in the cytoplasm of the cell.

Source For Ribose-5 -Phosphate

Conversion of Ribose-5 -phosphate to PRPP • Ribose: Pentose sugar; may be reduced to deoxyribose (DNA). • 5 -Phosphoribosyl-1 -pyrophosphate (PRPP): also involved in pyrimidine synthesis, NAD+, and histidine biosynthesis.


� From normal turnover of cellular nucleic acids � Obtained from the diet � Reutilization of adenine, hypoxanthine, and guanine �Two enzymes: 1. Adenine phosphoribosyltransferase 2. Hypoxanthine-guanine phosphoribosyltransferase

METHOTREXATE � Mode of Action �Dihydrofolate � reductase Adverse events: � Anemia, scaly skin, GI tract disturbances (diarrhea), Baldness � Resistance: Amplification of dihydrofolate reductase gene � Other indications: � Rheumatoid � Psoriasis arthritis (lower doses; inhibition of salvage pathways; increased adenosine, inhibits T cell activation.


Can synthesize folate Cannot synthesize folate

High levels shut down de novo purine synthesis Mycophenolic acid

�KEY: Feedback �Purine Inhibition biosynthesis: 3 sites: 1) glutamine phosphoribosyl amidotransferase 2) the reactions leading away from inosinate 3) the reciprocal substrate relationship between GTP and ATP

Another Look at Regulation Fig 26. 6

Lesch-Nyhan Syndrome • Build up of hypoxanthine and guanine • Degradation of hypoxanthine and guanine results in increased uric acid • Excess uric acid in urine often results in orange crystals in the diaper of affected children • Severe mental retardation • Self-mutilation • Involuntary movements • Gout

1. Sulfonamides inhibit purine synthesis in bacteria by interfering with folate synthesis. 2. Methotrexate inhibits dihydrofolate reductase. 3. IMP, end product of de novo purine synthesis. 4. AMP, GMP, and IMP inhibit; PRPP is an activator. 5. Rate limiting step of the pathway and source of atoms for the purine ring. 6. Requires 4 ATP molecules.


� Pyrimidine ring: completely synthesized, then attached to a ribose-5 -phosphate donated by PRPP � Source of carbons and nitrogens less diverse than purines.

(Carbamoyl-P)

Enzymatic functions from one large protein (215, 000 Mr) Enzymatic functions from one large protein

� Carbamoyl-phosphate synthetase II, Aspartate transcarbamoylase, Dihydroorotase, i. e. the CAD Complex (in mammals); located on the outer face of the inner mitochondrial membrane. � Orotate phosphoribosyltransferase and Orotidylate decarboxylase, i. e. , the UMP Synthase

Urea Synthesis Pyrimidine Synthesis

The Urea Cycle CPS-1 carbamoyl phosphate synthetase I OTC Ornithine transcarbamylase ASS argininosuccinate synthetase ASL argininosuccinate lyase ARG 1 arginase 1

The reactions of the urea cycle Step Reactants Products Catalyzed by Location 1 NH 4+ + HCO 3− + 2 ATP carbamoyl phosphate + 2 ADP CPS 1 + Pi mitochondria 2 carbamoyl phosphate + ornithine citrulline + Pi mitochondria 3 citrulline + aspartate + ATP argininosuccinate ASS + AMP + PPi 4 argininosuccinate Arg + fumarate ASL cytosol 5 Arg + H 2 O ARG 1 cytosol ornithine + urea OTC cytosol

�KEY: Feedback Inhibition �Pyrimidine Biosynthesis In bacteria: Aspartate Transcarbamoylase In both prokaryotes and eukaryotes: Carbamoyl phosphate synthetase

1. CPSII, aspartate transcarbamoylase, and dihydroorotase are three enzymatic functions in one protein. 2. Orotate phosphoribosyltransferase and OMP decarboxylase are two enzymatic functions in one protein; deficiency = Orotic Aciduria. 3. Orotate, 1 st pyrimidine base made, then attached to a PRPP.

Very Important!
![• • High [ATP] • plenty of energy, make DNA • activation of • • High [ATP] • plenty of energy, make DNA • activation of](http://slidetodoc.com/presentation_image_h2/b493474eb40ada4555875de16ab858c1/image-40.jpg)
• • High [ATP] • plenty of energy, make DNA • activation of ribonucleotide reductase is active (ON) ATP • in specificity site S favors CDP or UDP in catalytic site C [d. CDP] and [d. UDP] ↑ • d. CDP and d. UDP become metabolized to d. TTP • [d. TTP]↑, occupies specificity site favoring GDP in catalytic site; [d. GP]↑ [d. GTP]↑ • [d. GTP]↑, occupies specificity site, favors ADP in catalytic site, [d. ADP]↑ replace ATP in activity site and turn enzyme off

� Purines: �Synthesis begins with PRPP, from Ribose 5 -PO 4 � 12 steps, from nine sources � 2 nucleotides �Two-ringed structures � Pyrimidines: �Synthesis begins with the pyrimidine ring, then attached to Ribose 5 -PO 4 � 6 to 7 steps, from three sources � 3 nucleotides �Single ringed structures
- Slides: 41