Objectives 1 Define buffers 2 Calculate the p

Objectives: 1. Define buffers 2. Calculate the p. H of various buffer solutions
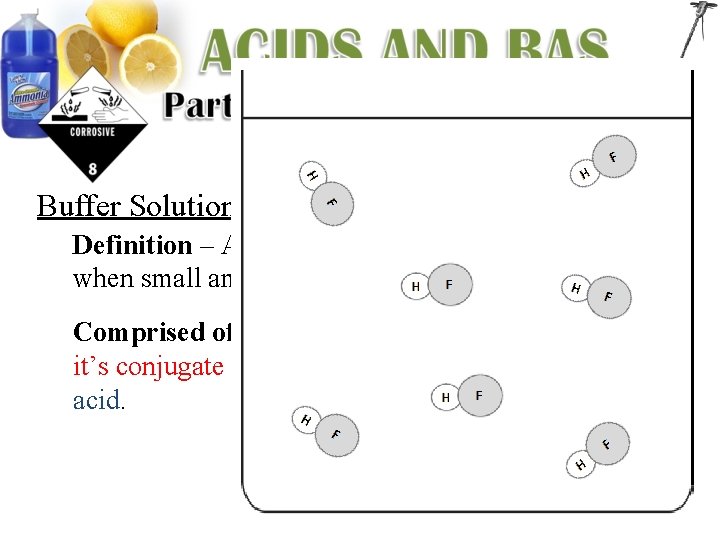
Buffer Solutions: Definition – Aqueous solutions that resists p. H changes when small amounts of acids or bases are added. Comprised of – A solution containing a weak acid and it’s conjugate base or a weak base and it’s conjugate acid.
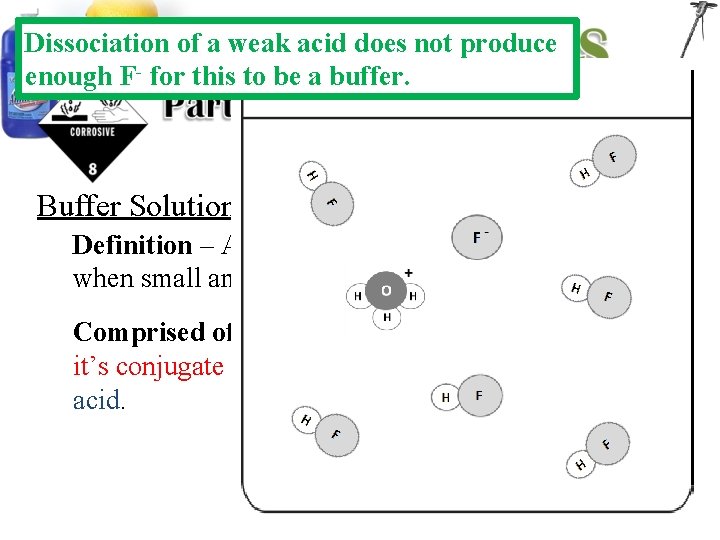
Dissociation of a weak acid does not produce enough F- for this to be a buffer. Buffer Solutions: Definition – Aqueous solutions that resists p. H changes when small amounts of acids or bases are added. Comprised of – A solution containing a weak acid and it’s conjugate base or a weak base and it’s conjugate acid.
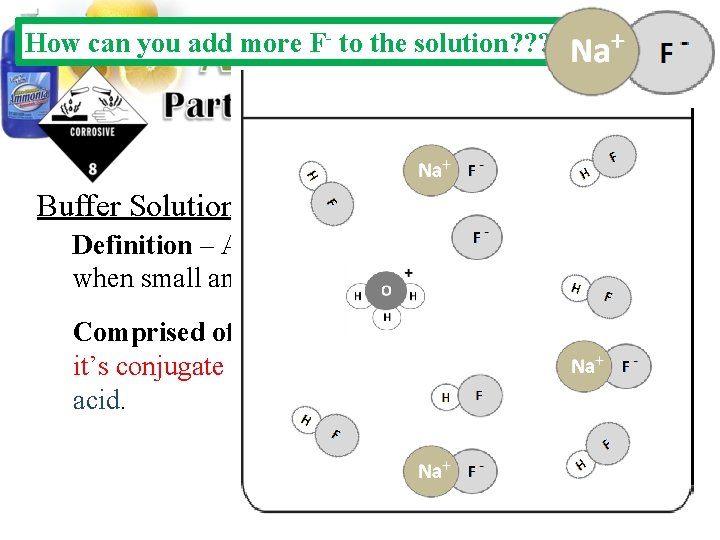
How can you add more F- to the solution? ? ? Buffer Solutions: Definition – Aqueous solutions that resists p. H changes when small amounts of acids or bases are added. Comprised of – A solution containing a weak acid and it’s conjugate base or a weak base and it’s conjugate acid.
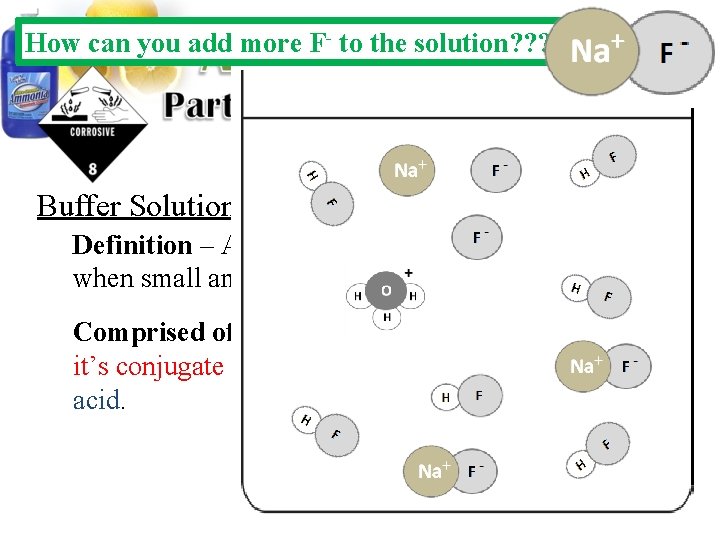
How can you add more F- to the solution? ? ? Buffer Solutions: Definition – Aqueous solutions that resists p. H changes when small amounts of acids or bases are added. Comprised of – A solution containing a weak acid and it’s conjugate base or a weak base and it’s conjugate acid.
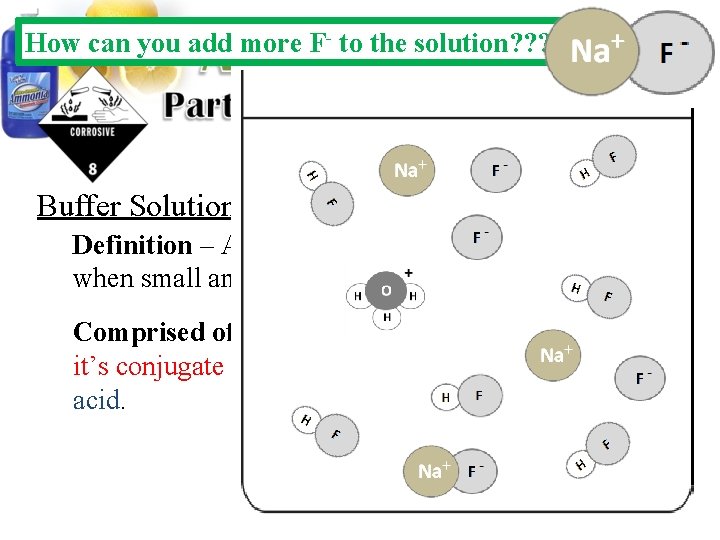
How can you add more F- to the solution? ? ? Buffer Solutions: Definition – Aqueous solutions that resists p. H changes when small amounts of acids or bases are added. Comprised of – A solution containing a weak acid and it’s conjugate base or a weak base and it’s conjugate acid.
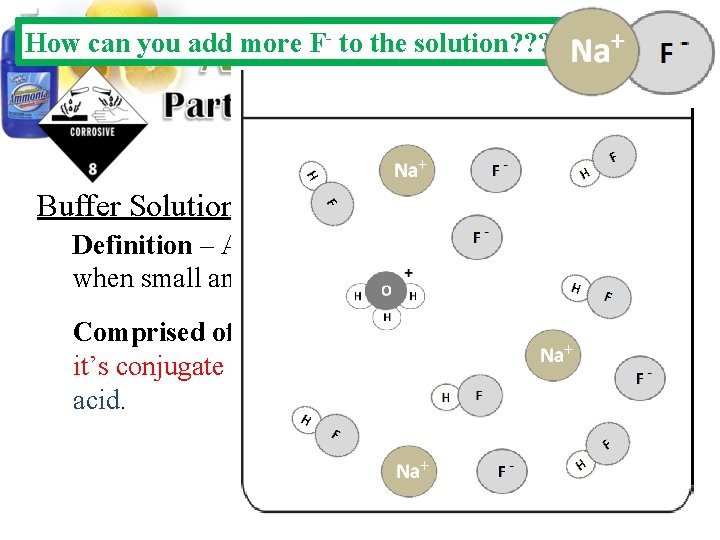
How can you add more F- to the solution? ? ? Buffer Solutions: Definition – Aqueous solutions that resists p. H changes when small amounts of acids or bases are added. Comprised of – A solution containing a weak acid and it’s conjugate base or a weak base and it’s conjugate acid.

Buffer Solutions: For example – You blood is strongly buffered by H 2 CO 3 and HCO 3 - at eqm with one another. How do they work … 1. If a strong acid is added to a buffer A-(aq) + H 3 O+(aq) --> H 2 O(l) + HA(aq) The strong acid reacts with the conjugate base of the buffer solution. This results in a decrease in the amount of conjugate base present and an increase in the amount of the weak acid. The p. H of the buffer solution decreases by a very small amount because of this ( a lot less than if the buffer system was not present). This buffering action is also referred to as the “common ion effect”

2. If a strong base is added to a buffer OH-(aq) + HA(aq) --> H 2 O(l) + A-(aq) When a strong base (OH-) is added to a buffer solution, the hydroxide ions reacts with the weak acid of the buffer solution forming water and the weaker conjugate base of the acid. The amount of the weak acid decreases while the amount of the conjugate base increases. This prevents the p. H of the solution from significantly rising, which it would if the buffer system was not present.

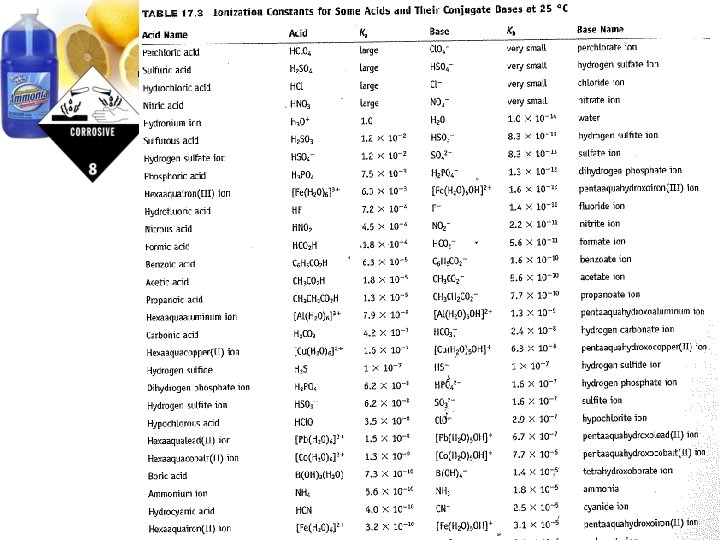
Finding the p. H of a buffer solution: What is the p. H of a 100 m. L buffer solution that is 0. 05 M acetic acid and 0. 05 M sodium acetate? (Ka of acetic acid = 1. 7 x 10 -5) How? ICE Tables and K values!!!

Finding the p. H of a buffer solution: Assume you have a 0. 30 M solution of formic acid (HCO 2 H) and have added enough sodium formate (Na. HCO 2) to make the solution 0. 1 M. Calculate the p. H of the buffer produced. Ka of HCO 2 H = 1. 8 x 10 -4 ; Kb of HCO 2 - = 5. 6 x 10 -11 Half the class will use the Ka, the other half use the Kb!


Finding the p. H of a buffer solution: Assume you have a 0. 30 M solution of formic acid (HCO 2 H) and have added enough sodium formate (Na. HCO 2) to make the solution 0. 1 M. Calculate the p. H of the buffer produced. Ka of HCO 2 H = 1. 8 x 10 -4 ; Kb of HCO 2 - = 5. 6 x 10 -11 Or use the Henderson-Hasselbach Equation!!!

Finding the p. H of a buffer solution: A buffer solution is made up of 75. 0 m. L of 0. 10 M NH 4+ and 25. 0 m. L of 0. 10 M NH 3. Find the p. H using the Henderson-Hasselbach equation. (NH 4+ Ka=5. 56 x 10 -10)
Acids and Bases
- Slides: 15