OBJECTIVE QUESTIONS FOR NEET AIIMS JIPMER www wisedane

OBJECTIVE QUESTIONS FOR NEET AIIMS JIPMER www. wisedane. com

PHYSICS HEAT AND THERMODYNAMICS SET 2 MCQ’S

www. wisedane. com

ANSWER (D) EXPLANATION www. wisedane. com

Q 2. A slab consists of two portions of different materials of same thickness and having the conductivities K 1 and K 2. The equivalent thermal conductivity of the slab is www. wisedane. com

ANSWER (C) EXAPLANATION www. wisedane. com
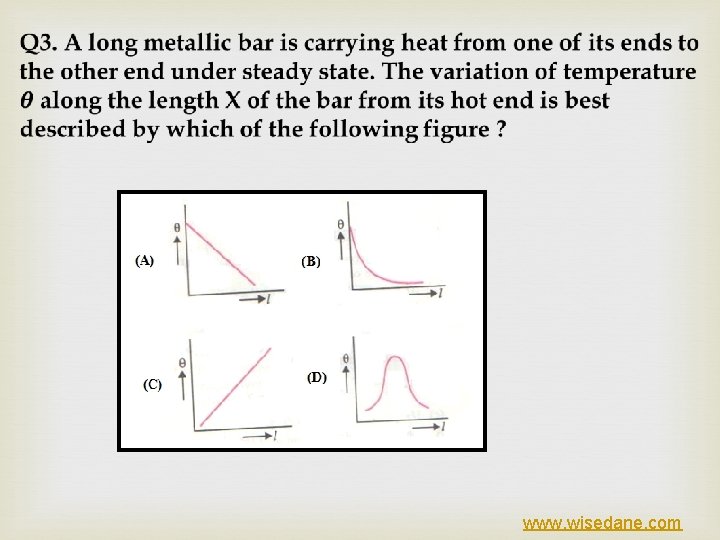
www. wisedane. com
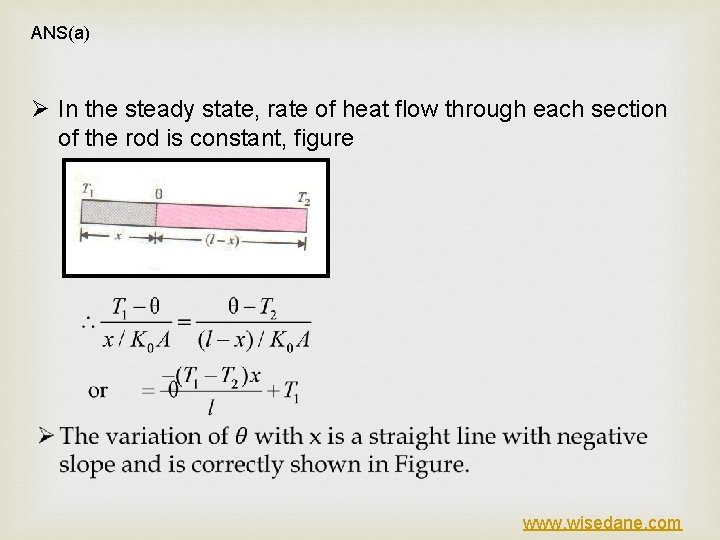
ANS(a) Ø In the steady state, rate of heat flow through each section of the rod is constant, figure www. wisedane. com
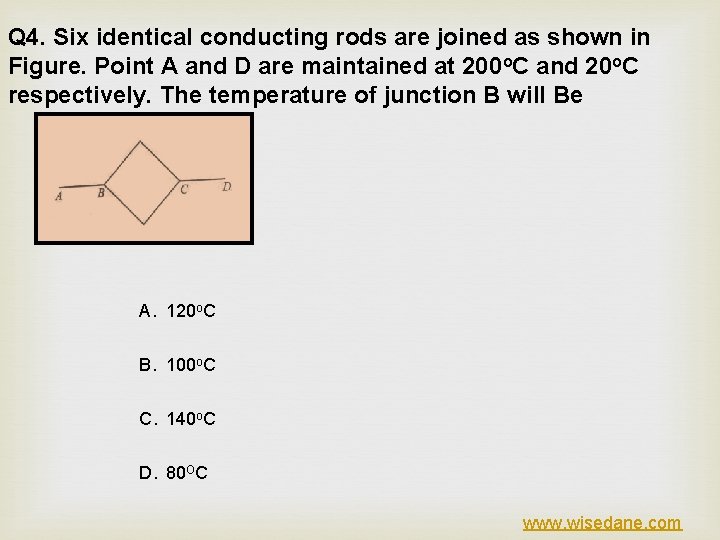
Q 4. Six identical conducting rods are joined as shown in Figure. Point A and D are maintained at 200 o. C and 20 o. C respectively. The temperature of junction B will Be A. 120 o. C B. 100 o. C C. 140 o. C D. 80 OC www. wisedane. com

ANSWER (C) EXPLANATION Ø The equivalent electrical circuit of the arrangement is shown in Figure. www. wisedane. com
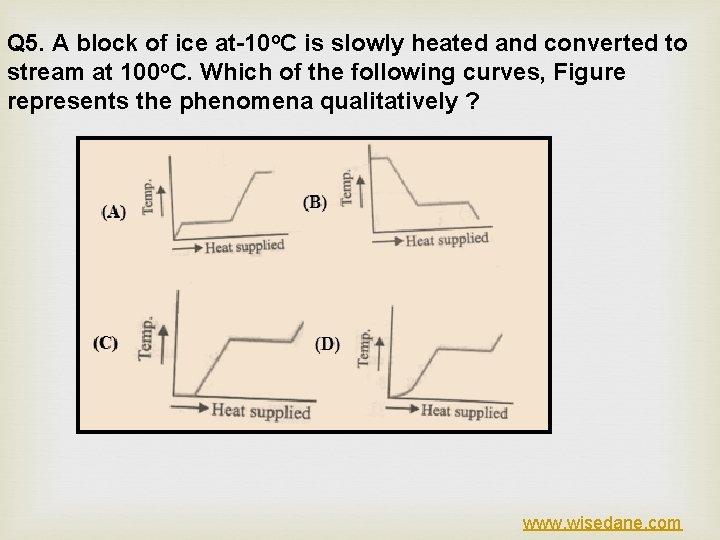
Q 5. A block of ice at-10 o. C is slowly heated and converted to stream at 100 o. C. Which of the following curves, Figure represents the phenomena qualitatively ? www. wisedane. com

ANSWER (A) EXPLANATION www. wisedane. com

Q 6. For the isothermal expansion of a gas, all the statements are correct except www. wisedane. com

ANSWER (C) EXPLANATION www. wisedane. com

Q 7. The ratio of the slopes of P-V graphs of adiabatic and isothermal changes is www. wisedane. com

ANSWER (D) EXPLANATION www. wisedane. com

Q 8. A monoatomic ideal gas at 27 o. C is suddenly compressed one tenth of its original volume. After compression, its temperature is A. 1265 o. K B. 1392 o. K C. 2009 o. K D. 7210 o. K www. wisedane. com

ANSWER (B) EXPLANATION www. wisedane. com

Q 9. If for hydrogen Cp-C V=m and for the nitrogen Cp-C V=n, Where Cp, C V refer to specific heats per unit mass respectively at constant pressure and constant volume. The relation between m and n is A. m = 14 n B. n= 7 m C. m= 7 n D. n=14 m www. wisedane. com

ANSWER (A) EXPLANATION www. wisedane. com

A. 1 : 16 B. 1 : 8 C. 1 : 4 D. 8 : 1 www. wisedane. com

ANSWER (D) EXPLANATION www. wisedane. com

A. 5/3 B. 3/2 C. 7/5 D. 4/3 www. wisedane. com

ANSWER (B) EXPLANATION www. wisedane. com
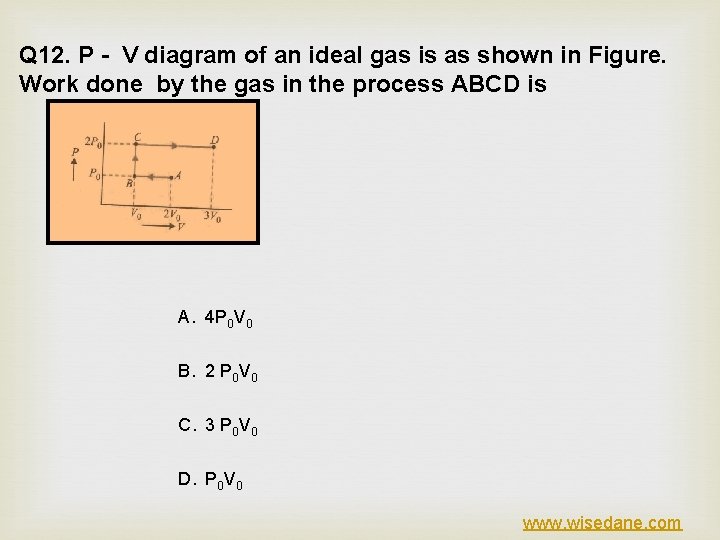
Q 12. P - V diagram of an ideal gas is as shown in Figure. Work done by the gas in the process ABCD is A. 4 P 0 V 0 B. 2 P 0 V 0 C. 3 P 0 V 0 D. P 0 V 0 www. wisedane. com

ANSWER (C) EXPLANATION www. wisedane. com
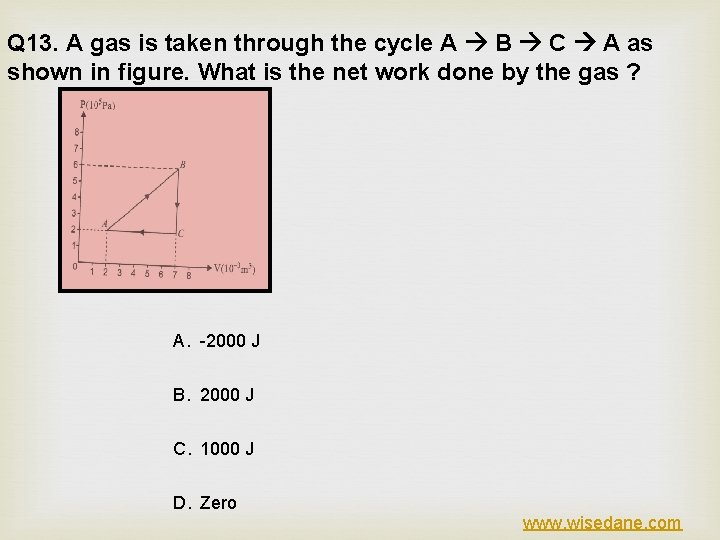
Q 13. A gas is taken through the cycle A B C A as shown in figure. What is the net work done by the gas ? A. -2000 J B. 2000 J C. 1000 J D. Zero www. wisedane. com

ANSWER (C) EXPLANATION www. wisedane. com

Q 14. By what percentage should the pressure of a given mass of a gas be increased so as to decrease its volume by 10% at a constant temperature ? A. 5% B. 7. 2% C. 12. 5% D. 11. 1% www. wisedane. com

ANSWER (D) EXPLANATION www. wisedane. com

Q 15. An ideal gas A and a real gas B have their volumes increased from V to 2 V under isothermal conditions. The increase in internal energy A. Of A will be more than B B. Of A will be less than B C. Will be same in both cases D. Will be zero in both cases. www. wisedane. com

ANSWER (D) EXPLANATION www. wisedane. com

Q 16. A sample of gas expands from volume V 1 to V 2, The amount of work done by the gas is max. when the expansion is A. Isothermal B. Adiabatic C. Isochoric D. Same in all the cases. www. wisedane. com

ANSWER (A) EXPLANATION Ø W = area under P –V curve = max. in isothermal expansion. www. wisedane. com

Q 17. A thermo dynamical system goes from state (i) (P, V) to (2 P, V) and (ii) (P, V) to (P, 2 V). Work done in the two cases is A. Zero, zero B. Zero, PV C. PV, zero D. PV, PV www. wisedane. com

ANSWER (B) EXPLANATION www. wisedane. com
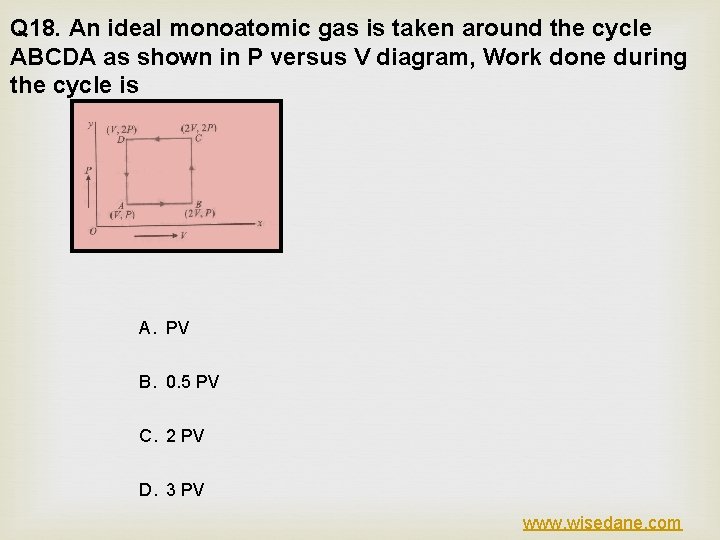
Q 18. An ideal monoatomic gas is taken around the cycle ABCDA as shown in P versus V diagram, Work done during the cycle is A. PV B. 0. 5 PV C. 2 PV D. 3 PV www. wisedane. com

ANSWER (A) EXPLANATION www. wisedane. com
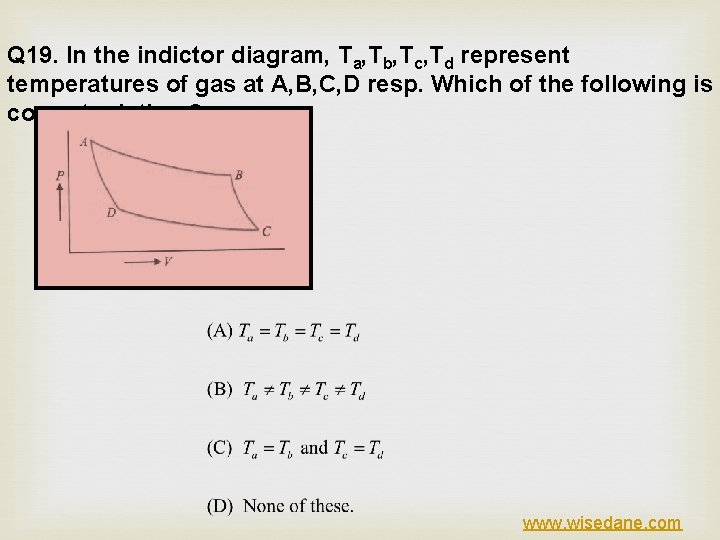
Q 19. In the indictor diagram, Ta, Tb, Tc, Td represent temperatures of gas at A, B, C, D resp. Which of the following is correct relation ? www. wisedane. com

ANSWER (C) EXPLANATION www. wisedane. com

Q 20. In the indicator diagram, AB is an isotherm and BC is an adiabatic, because A. AB and BC meet at B B. BC is shorter than AB C. Slope of AB is less than slope of BC D. None of the above. www. wisedane. com

ANSWER (C) EXPLANATION Ø Slope of isothermal curve (AB) is smaller than slope of adiabatic curve (BC). www. wisedane. com

More Mcq’s For All Subject Please Log on to http: //www. wisedane. com/ Exclusive Portal For Online Coaching NEET AIIMS JIPMER
- Slides: 43