OBJECTIVE QUESTIONS FOR NEET AIIMS JIPMER www wisedane

OBJECTIVE QUESTIONS FOR NEET AIIMS JIPMER www. wisedane. com

CHEMISTRY P-BLOCK ELEMENTS SET 1 MCQ’s

Q 1. Mark the oxide which is amphoteric in character. A. CO 2 B. Si. O 2 C. Sn. O 2 D. Ca. O

ANSWER (C) EXPLANATION Ø Sn. O 2 is amphoteric. It dissolves in acids as well as in alkalies e. g.

Q 2. Which one of the following compounds has the smallest bond angle in its molecule ? A. OH 2 B. H 2 S C. NH 3 D. SO 2
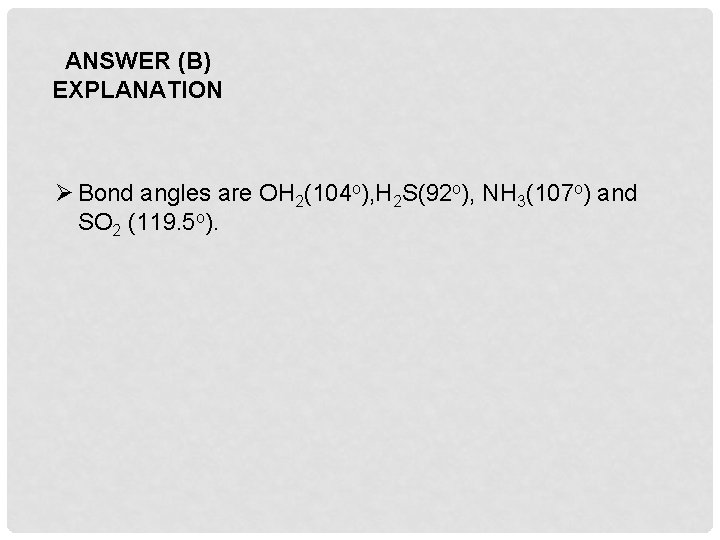
ANSWER (B) EXPLANATION Ø Bond angles are OH 2(104 o), H 2 S(92 o), NH 3(107 o) and SO 2 (119. 5 o).

Q 3. Boric acid is called acid because its molecule A. Contains replaceable H+ ion B. Gives up a proton C. Accepts OH from water releasing proton D. Combines with proton from water molecule

ANSWER (C) EXPLANATION Ø Due to the small size of boron atom and presence of six electrons. Ø Boric acid accepts a pair of electron from OH ion of H 2 O thereby releasing a proton.

Q 4. Strong reducing behaviour of H 3 PO 2 is due to A. Low oxidation state of phosphorus B. Presence of two – OH groups and one P – H bond C. Presence of one – OH group and two P – H bonds D. High electron gain enthalpy of phosphorus

ANSWER (C) EXPLANATION Ø Due to the presence of One – OH group, H 3 PO 2 act as reducing agent.

A. Addition of cis 1, 2 diol B. Addition of borax C. Addition of trans 1, 2 – diol D. Addition of Na 2 HPO 4
![ANSWER (A) EXPLANATION Ø This reaction is reversible reaction because sodium metaborate, Na+[B(OH)4] formed ANSWER (A) EXPLANATION Ø This reaction is reversible reaction because sodium metaborate, Na+[B(OH)4] formed](http://slidetodoc.com/presentation_image/44cbd54dab4a5413cdd015ee91c5670b/image-12.jpg)
ANSWER (A) EXPLANATION Ø This reaction is reversible reaction because sodium metaborate, Na+[B(OH)4] formed by the reaction between B(OH)3 and Na. OH gets hydrolysed to regenerate B (OH)3 and Na. OH. Ø If some quantity of polyhydroxy compounds like cis 1, 2 diol, catechol, glycerol etc. , is added to the reaction mixture, then the B(OH)3 combines with such polyhydroxy compounds to give chelated complex compound. Ø Due to complex compound formation, stability increases and due to higher stability of complex, reaction moves in forward direction.

Q 6. Heating of an aqueous solution of aluminium chloride to dryness will give A. Al(OH)Cl 2 B. Al 2 O 3 C. Al 2 Cl 6 D. Al. Cl 3

ANSWER (B) EXPLANATION

Q 7. Graphite is a soft, solid lubricant, extremely difficult to melt. The reason for this anomalous behaviour is that graphite A. Is an allotropic form of diamond B. Has molecules of variable molecular masses like polymers C. Has carbon atoms arranged in large plates of rings of strongly bound carbon atoms with weak interpolate bonds D. Is a non – crystalline substance

ANSWER (C) EXPLANATION Ø Graphite has carbon atoms arranged in large hexagonal layers with weak van der Waals’ interactions between the layers.

Q 8. Carborundum is obtained when silica is heated at high temperature with A. Carbon B. Carbon monoxide C. Carbon dioxide D. Calcium carbonate
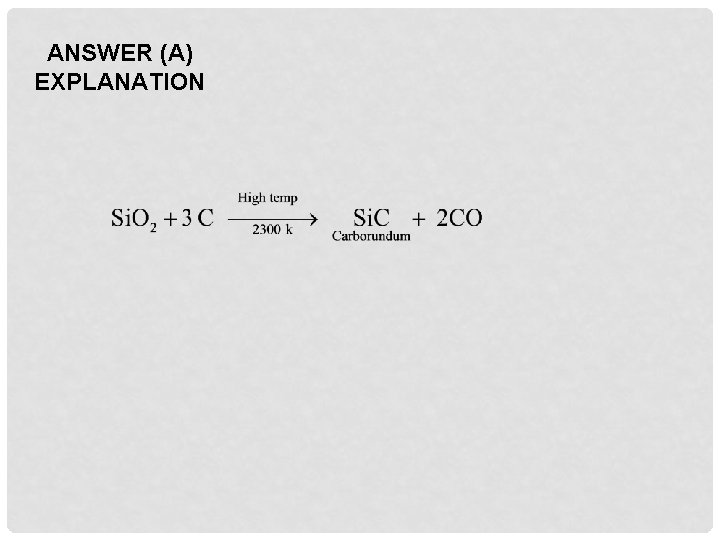
ANSWER (A) EXPLANATION

Q 9. Among the following substituted silanes the one which will give rise to cross linked silicon polymer on hydrolysis is A. R 3 Si. Cl B. R 4 Si C. R Si. Cl 3 D. R 2 Si. Cl 2
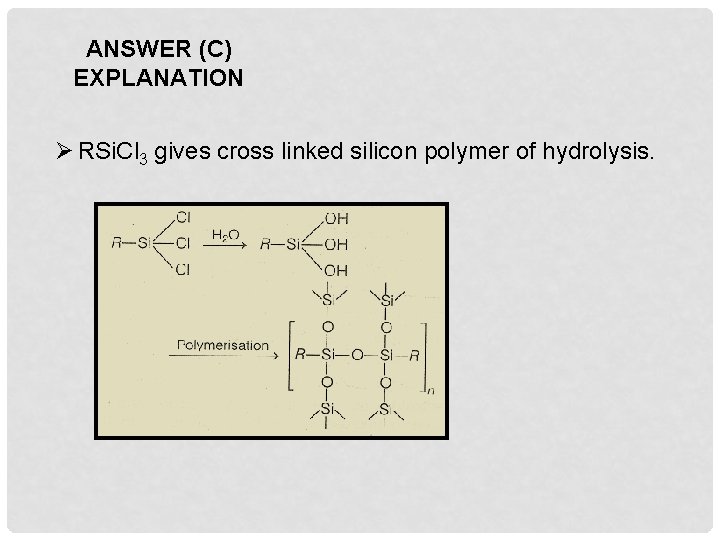
ANSWER (C) EXPLANATION Ø RSi. Cl 3 gives cross linked silicon polymer of hydrolysis.

Q 10. Name of the structure of silicates in which three oxygen atoms of [Si. O 4]4 - are shared, is A. Pyrosilicate B. Sheet silicate C. Linear chain Silicate D. Three dimensional silicate

ANSWER (B) EXPLANATION Ø The structure of silicates has been found with the help of X – ray diffraction techniques. Ø Sheet silicates are formed when three oxygen atoms (bridging O – atoms ) of each (SIO 4)4 unit are shared. Ø Hence, the general formula of sheet silicates is (SI 2 O 5)2 n

Q 11. The number of S – S bonds in sulphur trioxide trimer (S 3 O 9) is A. Three B. Two C. One D. Zero
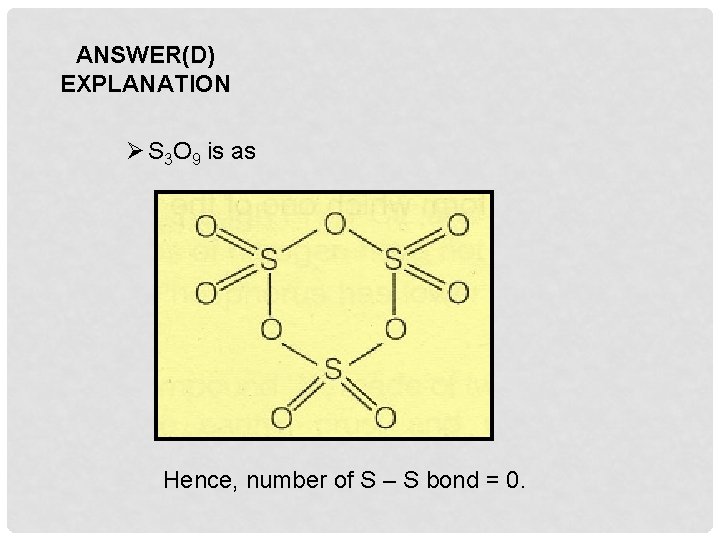
ANSWER(D) EXPLANATION Ø S 3 O 9 is as Hence, number of S – S bond = 0.

Q 12. In curing cement plasters water is sprinkled from time to time. This helps in A. Keeping it cool B. Developing interlocking needle like crystals of hydrated silicates C. Hydrating sand gravel mixed with cement D. Converting sand into silicic acid

ANSWER (B) EXPLANATION Ø The hydration of calcium aluminates and silicates involved in the setting of cement is an endothermic process, hence cement structures have to be cooled during setting by sprinkling water.

Q 13. Nitrogen shows different oxidation states in the range A. 0 to +5 B. 3 to +5 C. 5 to +3 D. 3 to +3

ANSWER (B) EXPLANATION Ø Nitrogen shows all the oxidation states ranging from – 3 to + 5 in its compounds.

Q 14. A hydride of nitrogen which is acidic, is A. NH 3 B. N 2 H 4 C. N 2 H 2 D. N 3 H
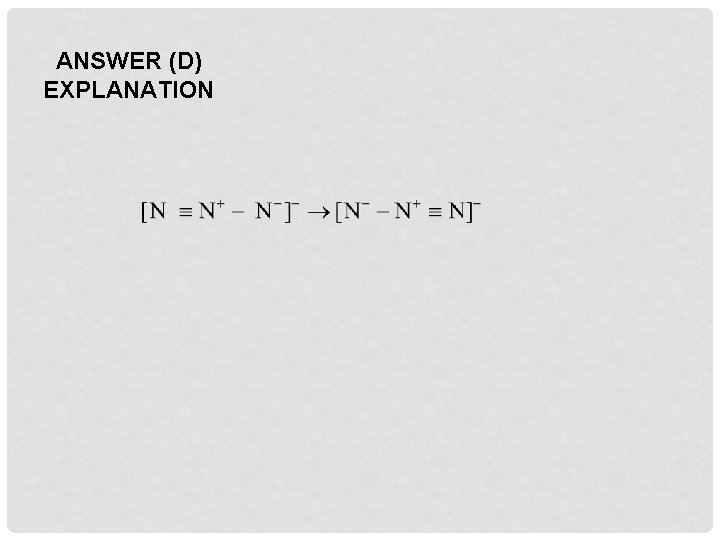
ANSWER (D) EXPLANATION

Q 15. Extra pure N 2 can be obtained by heating A. NH 3 with Cu. O B. NH 4 NO 3 C. (NH 4)2 Cr 2 O 7 D. Ba(N 3) 2

ANSWER (D) EXPLANATION Ø Azide salt of barium can be obtained in purest from as well as the decomposition product contain solid Ba as by – product along with gaseous nitrogen.

A. 6 B. 16 C. 20 D. 7
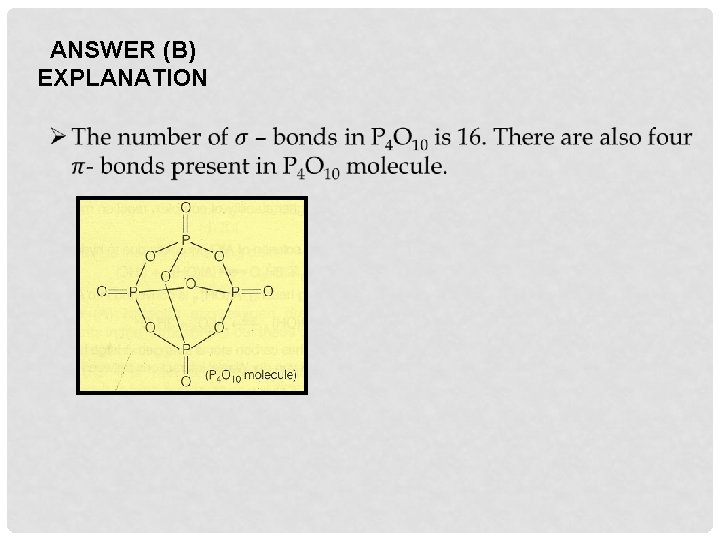
ANSWER (B) EXPLANATION

Q 17. PH 3 the hydride of phosphorus is A. Metallic B. Ionic C. Non – metallic D. Covalent

ANSWER (D) EXPLANATION Ø PH 3 is a covalent hydride.

Q 18. When concentrated HNO 3 is heated with P 2 O 5, it forms A. N 2 O B. NO C. NO 2 D. N 2 O 5

ANSWER(D) EXPLANATION

Q 19. Among the following, the number of compounds that can react with PCl 5 to give POCl 3 is O 2, CO 2, SO 2, H 2 OH 2 SO 4, P 4 O 10 A. 1 B. 2 C. 3 D. 4

ANSWER (D) EXPLANATION

Q 20. When conc. H 2 SO 4 is heated with P 2 O 5, the acid is converted to A. Sulphur trioxide B. Sulphur dioxide C. Sulphur D. A mixture of sulphur dioxide and sulphure trioxide

ANSWER (A) EXPLANATION Ø when conc. H 2 SO 4 is heated with P 2 O 5, the acid is converted into sulphur trioxide.

MORE MCQ’S FOR ALL SUBJECT PLEASE LOG ON TO WWW. WISEDANE. COM EXCLUSIVE PORTAL FOR ONLINE COACHING NEET AIIMS JIPMER
- Slides: 43