Objective and EQ Objective SWBAT calculate and compare

Objective and EQ �Objective: SWBAT calculate and compare the density of objects by using measured mass and volume �Essential Question: How can we measure and calculate the density of an object? What does density tell us about the composition of an object?

Which floats, Coke or diet Coke?

Apply it to real life: What makes Coke different from diet Coke? More sugar Less sugar

So what can we conclude about density? More dense = sink Less dense = float

Definition of Density: the amount of matter per unit of space of an object. How compact or packed together the matter is in the space of an object. This is a property that does NOT change depending on how much of it you have!
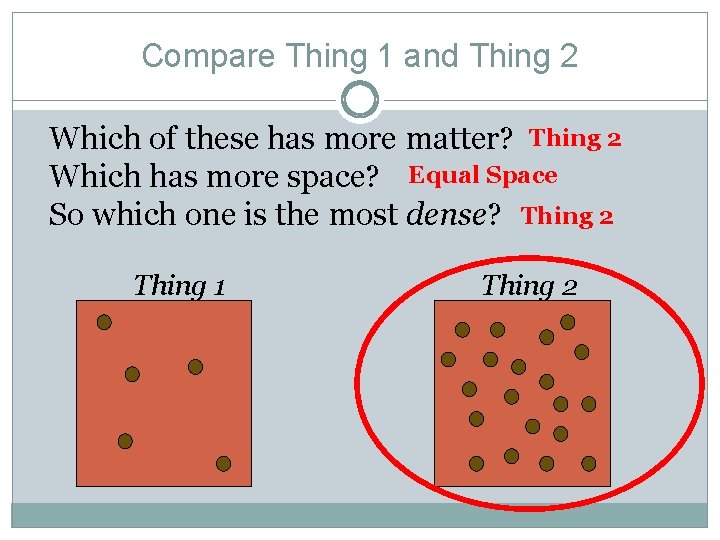
Compare Thing 1 and Thing 2 Which of these has more matter? Thing 2 Which has more space? Equal Space So which one is the most dense? Thing 2 Thing 1 Thing 2
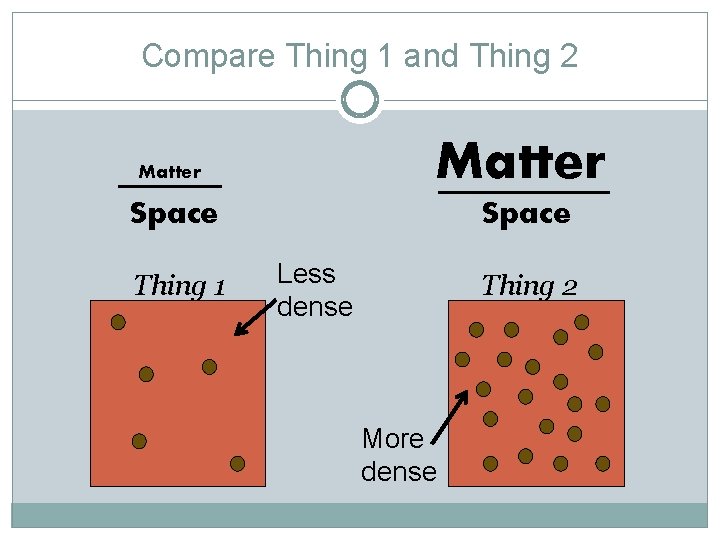
Compare Thing 1 and Thing 2 Matter Space Thing 1 Less dense Thing 2 More dense
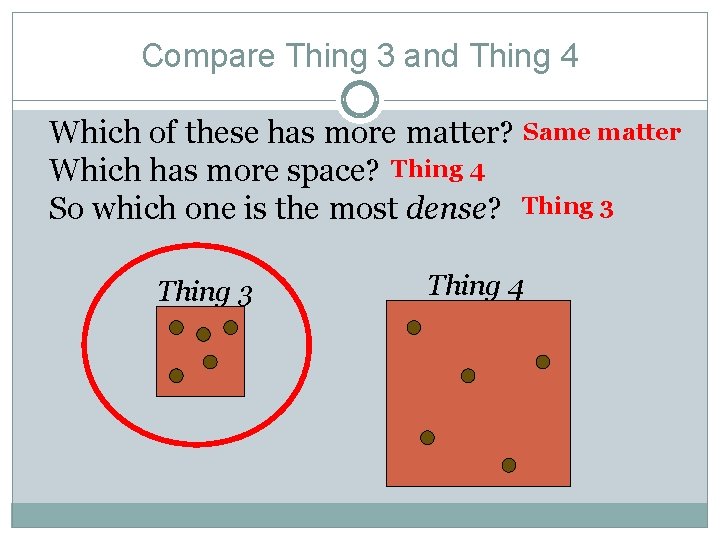
Compare Thing 3 and Thing 4 Which of these has more matter? Same matter Which has more space? Thing 4 So which one is the most dense? Thing 3 Thing 4
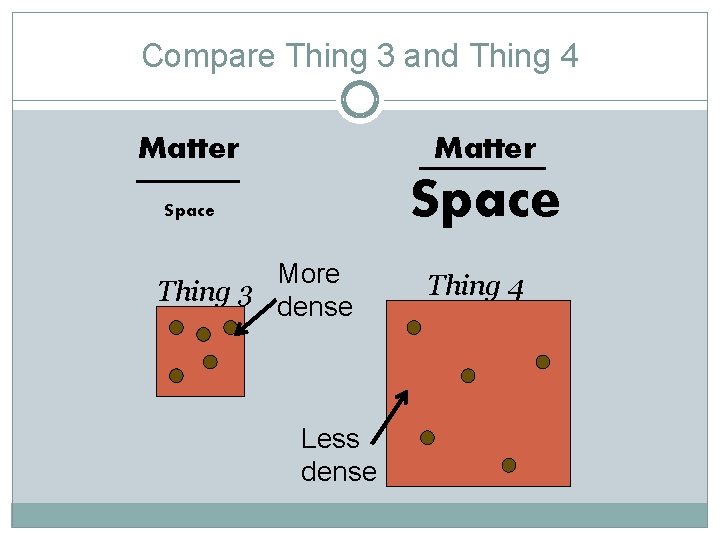
Compare Thing 3 and Thing 4 Matter Space More Thing 3 dense Less dense Thing 4
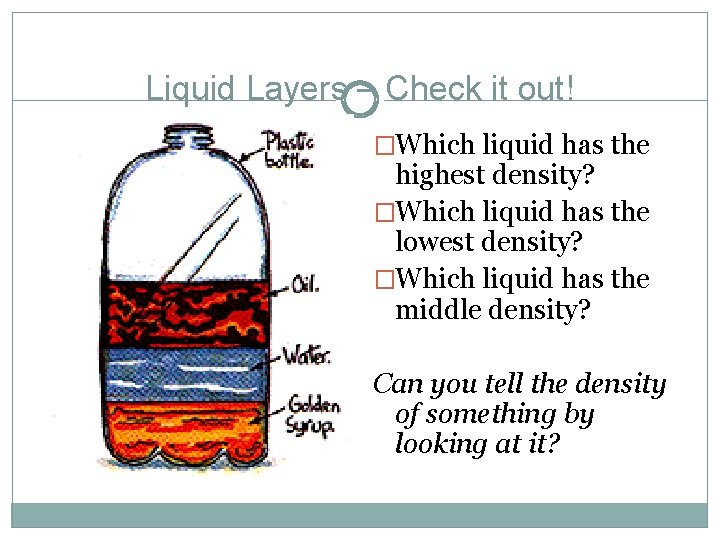
Liquid Layers – Check it out! �Which liquid has the highest density? �Which liquid has the lowest density? �Which liquid has the middle density? Can you tell the density of something by looking at it?
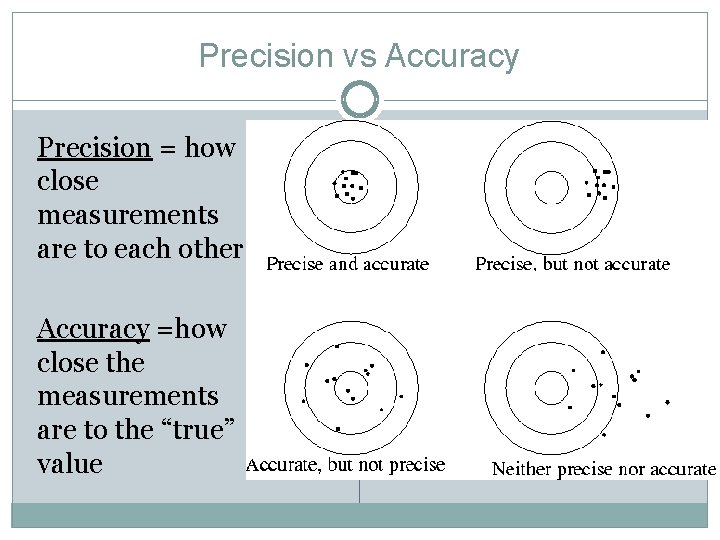
Precision vs Accuracy Precision = how close measurements are to each other Accuracy =how close the measurements are to the “true” value

Definition of Density: the amount of matter per unit of space of an object Matter: Mass per: Space: Volume

Ex 1: Osmium is a very dense metal. What is its density if 50. 00 g of the metal occupies a volume of 2. 22 cm 3? Given: m = 50. 00 g Unknown: D = ? V = 2. 22 cm 3 Equation: mass D = volume D= 50. 00 g 2. 22 cm 3 D = 22. 5 g/cm 3 Don’t forget units!!! Don’t forget units! Box answer!

Ex 2: What is the density of 48 g of a metal if the metal raises the level of water in a graduated cylinder from 25 m. L to 3314 m. L? Volume (m. L) of water displaced = Final volume – initial volume = 33 m. L -- 25 m. L = 8 m. L volume 25 m. L lecture. PLUS Timberlake 33 m. L

Ex 2: What is the density of 48 g of a metal if the metal raises the level of water in a graduated cylinder from 25 m. L to 33 m. L? Given: m = 48 g Unknown: D = ? V = 33 – 25 = 8 m. L Equation: mass D = volume D= 48 g 8 m. L D = 6. 0 g/m. L Don’t forget units!!! Don’t forget units! Box answer!

Example 3: You do 16 Gold has a density of 19. 32 g/cm 3. Find the mass of 6. 39 cm 3 of gold. D = mass = 50. 00 g = volume 2. 22 cm 3 = 22. 522522 g/cm 3 = 22. 5 g/cm 3 lecture. PLUS Timberlake

Gold has a density of 19. 32 g/cm 3. Find the mass of 6. 39 cm 3 of gold. mass x volume x Density = volume Solve for mass Mass = volume x density = 6. 39 cm 3 x 19. 32 g/cm 3 = 123 g

Whiteboard Practice An unknown liquid is discovered at a crime scene. A volume of 2. 3 m. L has a mass of 4. 1 grams, what the liquid’s density? 4. 1 g / 2. 3 m. L = 1. 8 g/m. L A metal can displaces water from 25 to 40 m. L. If the can weighs 15 g, what is the density? 15 g / 15 m. L = 1. 0 g/m. L Diamond has a density of 3. 26 g/cm 3. What is the mass of a diamond that has a volume of 0. 350 cm 3? 3. 26 g/cm 3 x 0. 350 cm 3 = 1. 14 g
- Slides: 18