Obeticholic acid By Mohamed Fathy Bakosh Demonstrator of

Obeticholic acid By Mohamed Fathy Bakosh Demonstrator of hepatology
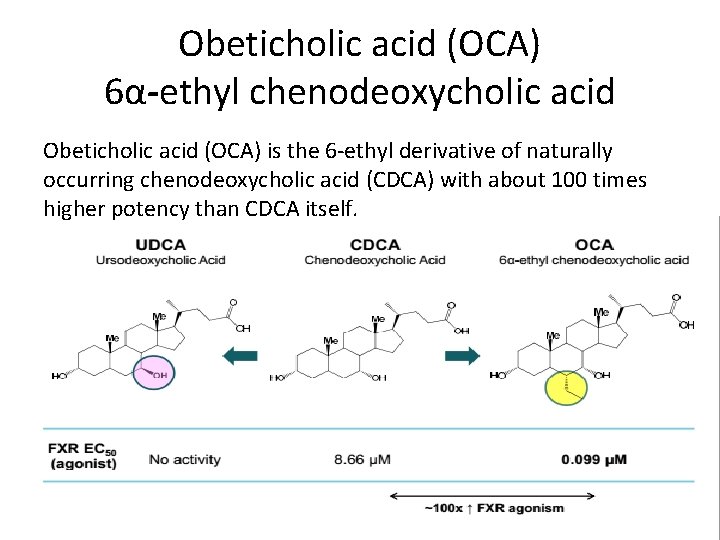
Obeticholic acid (OCA) 6α-ethyl chenodeoxycholic acid Obeticholic acid (OCA) is the 6 -ethyl derivative of naturally occurring chenodeoxycholic acid (CDCA) with about 100 times higher potency than CDCA itself.

Mechanism of actions • Obeticholic acid is a farnesoid x receptor (FXR) agonist , and reduces hepatic exposure to bile acids by reducing the amount of circulating bile acid. • FXR is expressed in the liver and intestine, and is reponsable for regulating bile acid, inflammatory, fibrotic, and metabolic pathways.
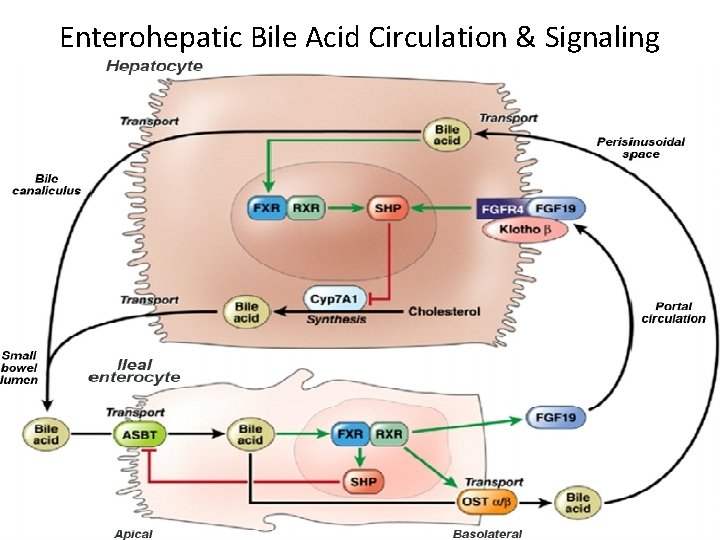
Enterohepatic Bile Acid Circulation & Signaling

Bile acid homeostasis • The total bile acid pool in humans is tightly controlled by a coordinated regulation of expression of genes involved with synthesis, secretion, reabsorption and reuptake of bile acids by the liver. • The major components of the bile acid pool are cholic and chenodeoxycholic acid with lesser amounts deoxycholic and lithocholic acid and minor amounts of ursodeoxycholic acid • CDCA as the most potent FXR activator, while DCA and LCA activated FXR with a lower efficacy than CDCA Hydrophilic unconjugated BAs, such as CA and UDCA, could not activate FXR.
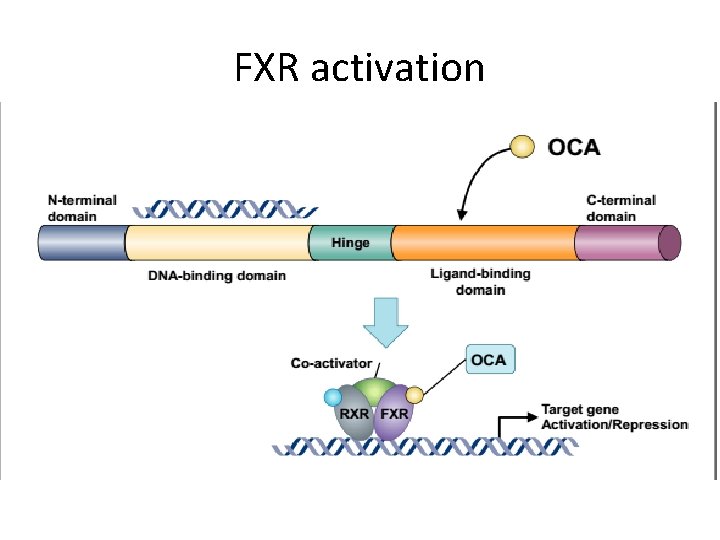
FXR activation
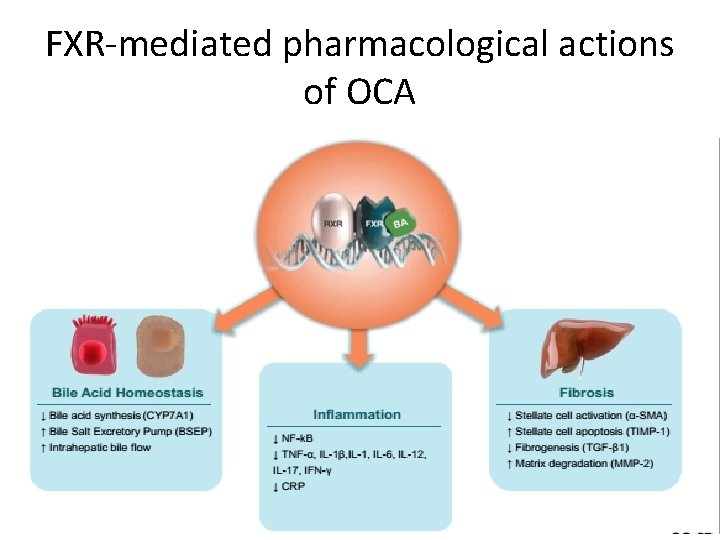
FXR-mediated pharmacological actions of OCA

UDCA and OCA properties

OCA –key properties

Indication • Treatment of primary biliary cholangitis in combination with ursodeoxycholic acid (UDCA) in adults with an inadequate response to UDCA or as monotherapy in adults unable to tolerate UDCA.

Adverse reactions • Common Dermatologic: pruritis (up to 70%). Endocrine and metabolic: dose-dependant reductions in HDL-C levels. Others: Fatigue(19% to 25%) • Serious Hepatic : hepatic decompensation, Ascites and jaundice

Dosage & adminstration

Dosage & adminstration Intolerable pruritis : 1. Consider adding an antihistamine or bile acid binding resin. 2. Consider interrupting obeticholic acid treatment for 2 weeks, then restart at reduced dosage. 3. If intolerant to 5 mg once daily, consider dosage reduction to 5 mg every other day. 4. If intolerant to 10 mg once daily, consider dosage reduction to 5 mg every other day.

Warning &precautions

Interactions

Future indication-NASH • REGENERATE, a pivotal phase 3 study of obeticholic acid (OCA) by Intercept Pharmaceuticals, Inc. , has demonstrated positive results in patients with liver fibrosis from nonalcoholic steatohepatitis (NASH). • The primary efficacy analysis indicated that treatment with once-daily OCA 25 mg met the primary endpoint of fibrosis improvement (≥ 1 stage) with no worsening of NASH after 18 months.

Future indication-PSC • The AESOP trial was a 24 -week, double-blind, placebo-controlled , phase 2 trial including 77 patients with PSC who received either 1. 5 mg to 3 mg of obeticholic acid, 5 mg to 10 mg of obeticholic acid, or placebo. Compared with a 1% increase of alkaline phosphatase in the control group, both groups who received obeticholic acid treatment had a mean reduction of 22% (P <. 05).
Thank you
- Slides: 18