Nutrition Metabolism and Body Temperature Regulation Copyright 2004

Nutrition, Metabolism, and Body Temperature Regulation Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Nutrition § Nutrient – a substance that promotes normal growth, maintenance, and repair § Major nutrients – carbohydrates, lipids, and proteins § Other nutrients – vitamins and minerals (and technically speaking, water) Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings
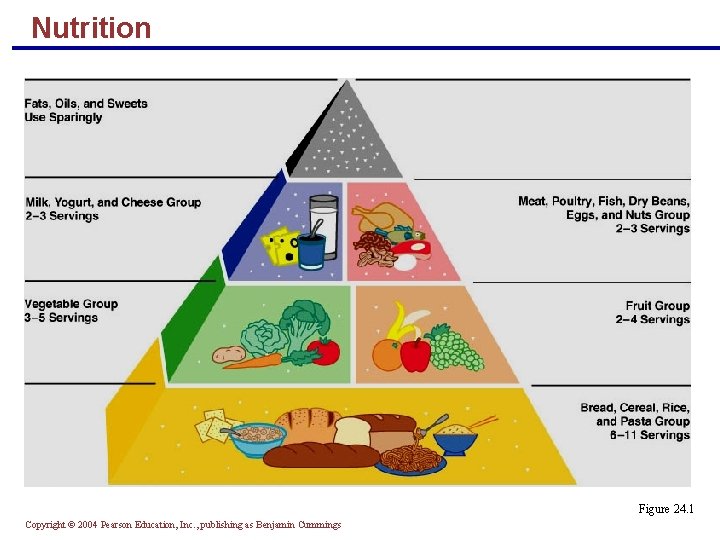
Nutrition Figure 24. 1 Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Carbohydrates § Complex carbohydrates (starches) are found in bread, cereal, flour, pasta, nuts, and potatoes § Simple carbohydrates (sugars) are found in soft drinks, candy, fruit, and ice cream § Glucose is the molecule ultimately used by body cells to make ATP § Neurons and RBCs rely almost entirely upon glucose to supply their energy needs § Excess glucose is converted to glycogen or fat and stored Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Carbohydrates § The minimum amount of carbohydrates needed to maintain adequate blood glucose levels is 100 grams per day § Starchy foods and milk have nutrients such as vitamins and minerals in addition to complex carbohydrates § Refined carbohydrate foods (candy and soft drinks) provide energy sources only and are referred to as “empty calories” Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Lipids § The most abundant dietary lipids, triglycerides, are found in both animal and plant foods § Essential fatty acids – linoleic and linolenic acid, found in most vegetables, must be ingested § Dietary fats: § Help the body to absorb vitamins § Are a major energy fuel of hepatocytes and skeletal muscle § Are a component of myelin sheaths and all cell membranes Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Lipids § Fatty deposits in adipose tissue provide: § A protective cushion around body organs § An insulating layer beneath the skin § An easy-to-store concentrated source of energy Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Lipids § Prostaglandins function in: § Smooth muscle contraction § Control of blood pressure § Inflammation § Cholesterol stabilizes membranes and is a precursor of bile salts and steroid hormones Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Lipids: Dietary Requirements § Higher for infants and children than for adults § The American Heart Association suggests that: § Fats should represent less than 30% of one’s total caloric intake § Saturated fats should be limited to 10% or less of one’s total fat intake § Daily cholesterol intake should not exceed 200 mg Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Proteins § Complete proteins that meet all the body’s amino acid needs are found in eggs, milk products, meat, and fish § Incomplete proteins are found in legumes, nuts, seeds, grains, and vegetables Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Proteins § Proteins supply: § Essential amino acids, the building blocks for nonessential amino acids § Nitrogen for nonprotein nitrogen-containing substances § Daily intake should be approximately 0. 8 g/kg of body weight Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Proteins: Synthesis and Hydrolysis § All-or-none rule § All amino acids needed must be present at the same time for protein synthesis to occur § Adequacy of caloric intake § Protein will be used as fuel if there is insufficient carbohydrate or fat available Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Proteins: Synthesis and Hydrolysis § Nitrogen balance § The rate of protein synthesis equals the rate of breakdown and loss § Positive – synthesis exceeds breakdown (normal in children and tissue repair) § Negative – breakdown exceeds synthesis (e. g. , stress, burns, infection, or injury) § Hormonal control § Anabolic hormones accelerate protein synthesis Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Vitamins § Organic compounds needed for growth and good health § They are crucial in helping the body use nutrients and often function as coenzymes § Only vitamins D, K, and B are synthesized in the body; all others must be ingested § Water-soluble vitamins (B-complex and C) are absorbed in the gastrointestinal tract § B 12 additionally requires gastric intrinsic factor to be absorbed Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Vitamins § Fat-soluble vitamins (A, D, E, and K) bind to ingested lipids and are absorbed with their digestion products § Vitamins A, C, and E also act in an antioxidant cascade Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Minerals § Seven minerals are required in moderate amounts § Calcium, phosphorus, potassium, sulfur, sodium, chloride, and magnesium § Dozens are required in trace amounts § Minerals work with nutrients to ensure proper body functioning § Calcium, phosphorus, and magnesium salts harden bone Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Minerals § Sodium and chloride help maintain normal osmolarity, water balance, and are essential in nerve and muscle function § Uptake and excretion must be balanced to prevent toxic overload Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Metabolism § Metabolism – all chemical reactions necessary to maintain life § Cellular respiration – food fuels are broken down within cells and some of the energy is captured to produce ATP § Anabolic reactions – synthesis of larger molecules from smaller ones § Catabolic reactions – hydrolysis of complex structures into simpler ones Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Metabolism § Enzymes shift the high-energy phosphate groups of ATP to other molecules § These phosphorylated molecules are activated to perform cellular functions Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Stages of Metabolism § Energy-containing nutrients are processed in three major stages § Digestion – breakdown of food; nutrients are transported to tissues § Anabolism and formation of catabolic intermediates where nutrients are: § Built into lipids, proteins, and glycogen § Broken down by catabolic pathways to pyruvic acid and acetyl Co. A § Oxidative breakdown – nutrients are catabolized to carbon dioxide, water, and ATP Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings
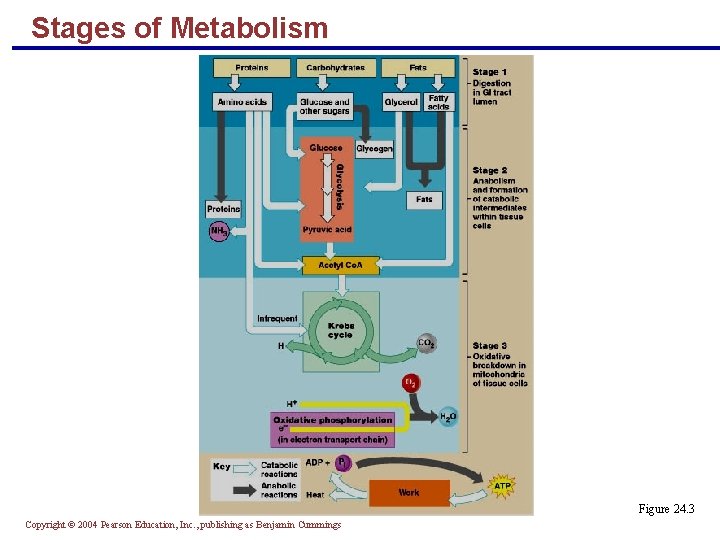
Stages of Metabolism Figure 24. 3 Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Oxidation-Reduction (Redox) Reactions § Oxidation occurs via the gain of oxygen or the loss of hydrogen § Whenever one substance is oxidized, another substance is reduced § Oxidized substances lose energy § Reduced substances gain energy § Coenzymes act as hydrogen (or electron) acceptors § Two important coenzymes are nicotinamide adenine dinucleotide (NAD+) and flavin adenine dinucleotide (FAD) Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings
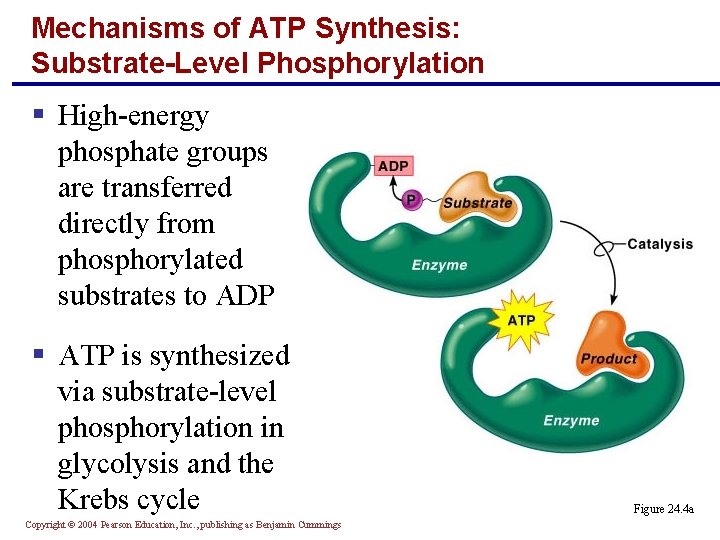
Mechanisms of ATP Synthesis: Substrate-Level Phosphorylation § High-energy phosphate groups are transferred directly from phosphorylated substrates to ADP § ATP is synthesized via substrate-level phosphorylation in glycolysis and the Krebs cycle Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings Figure 24. 4 a

Mechanisms of ATP Synthesis: Oxidative Phosphorylation § Uses the chemiosmotic process whereby the movement of substances across a membrane is coupled to chemical reactions Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Mechanisms of ATP Synthesis: Oxidative Phosphorylation § Is carried out by the electron transport proteins in the cristae of the mitochondria § Nutrient energy is used to pump hydrogen ions into the intermembrane space § A steep diffusion gradient across the membrane results § When hydrogen ions flow back across the membrane through ATP synthase, energy is captured and attaches phosphate groups to ADP (to make ATP) Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings
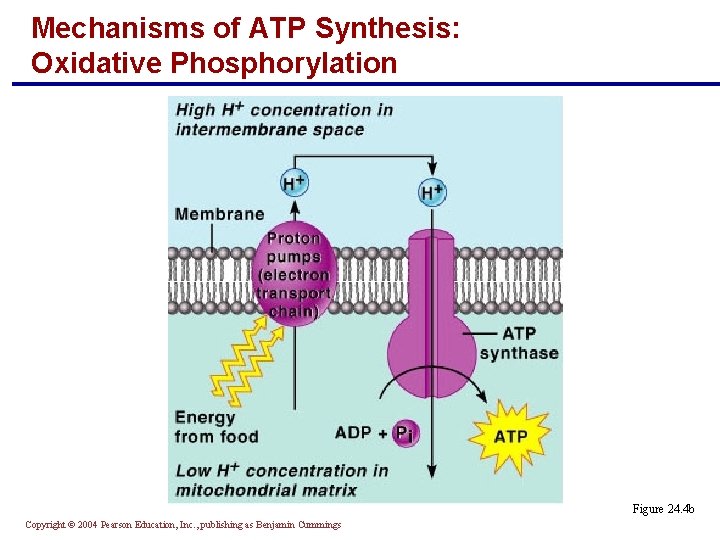
Mechanisms of ATP Synthesis: Oxidative Phosphorylation Figure 24. 4 b Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings
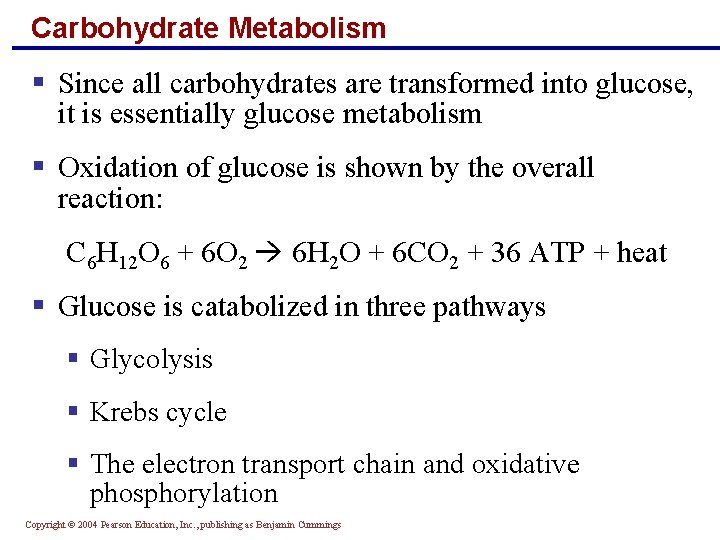
Carbohydrate Metabolism § Since all carbohydrates are transformed into glucose, it is essentially glucose metabolism § Oxidation of glucose is shown by the overall reaction: C 6 H 12 O 6 + 6 O 2 6 H 2 O + 6 CO 2 + 36 ATP + heat § Glucose is catabolized in three pathways § Glycolysis § Krebs cycle § The electron transport chain and oxidative phosphorylation Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings
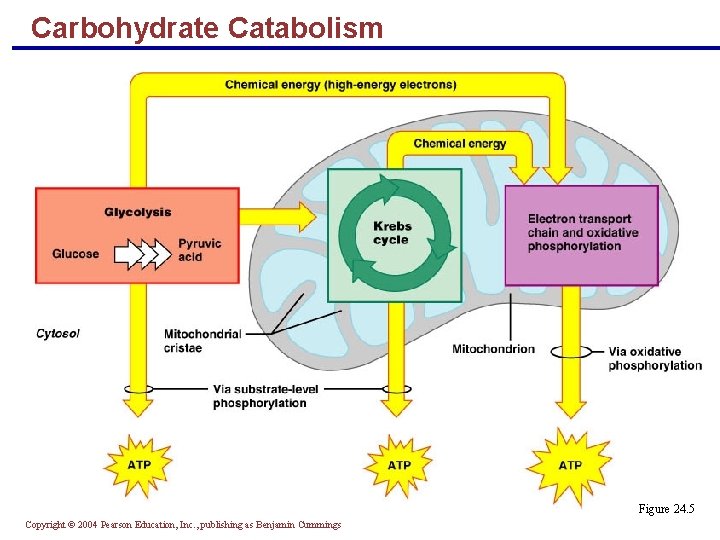
Carbohydrate Catabolism Figure 24. 5 Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Glycolysis § A three-phase pathway in which: § Glucose is oxidized into pyruvic acid § NAD+ is reduced to NADH + H+ § ATP is synthesized by substrate-level phosphorylation § Pyruvic acid: § Moves on to the Krebs cycle in an aerobic pathway § Is reduced to lactic acid in an anaerobic environment Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings
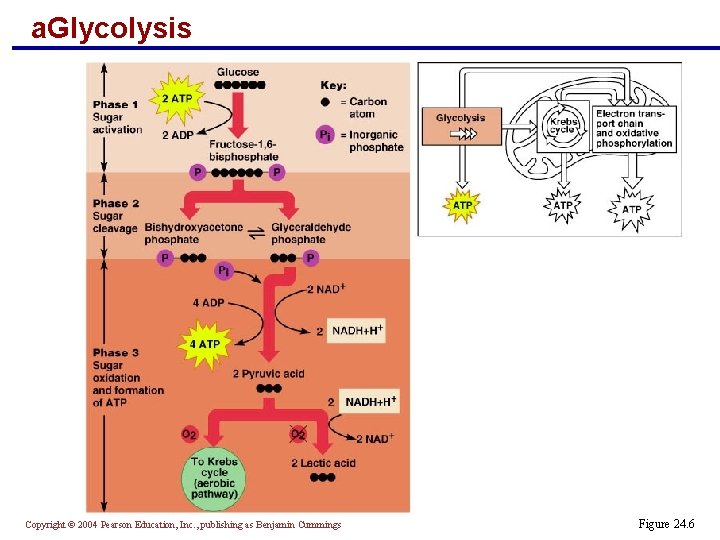
a. Glycolysis Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings Figure 24. 6

Glycolysis: Phase 1 and 2 § Phase 1: Sugar activation § Two ATP molecules activate glucose into fructose-1, 6 -diphosphate § Phase 2: Sugar cleavage § Fructose-1, 6 -bisphosphate is cleaved into two 3 -carbon isomers § Bishydroxyacetone phosphate § Glyceraldehyde 3 -phosphate Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Glycolysis: Phase 3 § Phase 3: Oxidation and ATP formation § The 3 -carbon sugars are oxidized (reducing NAD+) § Inorganic phosphate groups (Pi) are attached to each oxidized fragment § The terminal phosphates are cleaved and captured by ADP to form four ATP molecules Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Glycolysis: Phase 3 § The final products are: § Two pyruvic acid molecules § Two NADH + H+ molecules (reduced NAD+) § A net gain of two ATP molecules Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Krebs Cycle: Preparatory Step § Occurs in the mitochondrial matrix and is fueled by pyruvic acid and fatty acids Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Krebs Cycle: Preparatory Step § Pyruvic acid is converted to acetyl Co. A in three main steps: § Decarboxylation § Carbon is removed from pyruvic acid § Carbon dioxide is released Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Krebs Cycle: Preparatory Step § Oxidation § Hydrogen atoms are removed from pyruvic acid § NAD+ is reduced to NADH + H+ § Formation of acetyl Co. A – the resulting acetic acid is combined with coenzyme A, a sulfur-containing coenzyme, to form acetyl Co. A Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Krebs Cycle § An eight-step cycle in which each acetic acid is decarboxylated and oxidized, generating: § Three molecules of NADH + H+ § One molecule of FADH 2 § Two molecules of CO 2 § One molecule of ATP § For each molecule of glucose entering glycolysis, two molecules of acetyl Co. A enter the Krebs cycle PLAY Krebs Cycle Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings
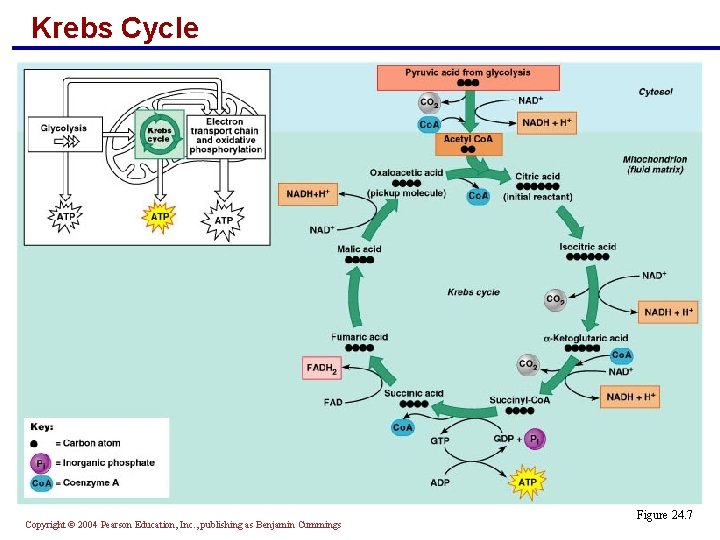
Krebs Cycle Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings Figure 24. 7

Electron Transport Chain § Food (glucose) is oxidized and the released hydrogens: § Are transported by coenzymes NADH and FADH 2 § Enter a chain of proteins bound to metal atoms (cofactors) § Combine with molecular oxygen to form water § Release energy § The energy released is harnessed to attach inorganic phosphate groups (Pi) to ADP, making ATP by oxidative phosphorylation Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Mechanism of Oxidative Phosphorylation § The hydrogens delivered to the chain are split into protons (H+) and electrons § The protons are pumped across the inner mitochondrial membrane by: § NADH dehydrogenase (FMN, Fe-S) § Cytochrome b-c 1 § Cytochrome oxidase (a-a 3) § The electrons are shuttled from one acceptor to the next Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Mechanism of Oxidative Phosphorylation § Electrons are delivered to oxygen, forming oxygen ions § Oxygen ions attract H+ to form water § H+ pumped to the intermembrane space: § Diffuses back to the matrix via ATP synthase § Releases energy to make ATP PLAY Inter. Active Physiology®: Muscular System: Muscle Metabolism Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings
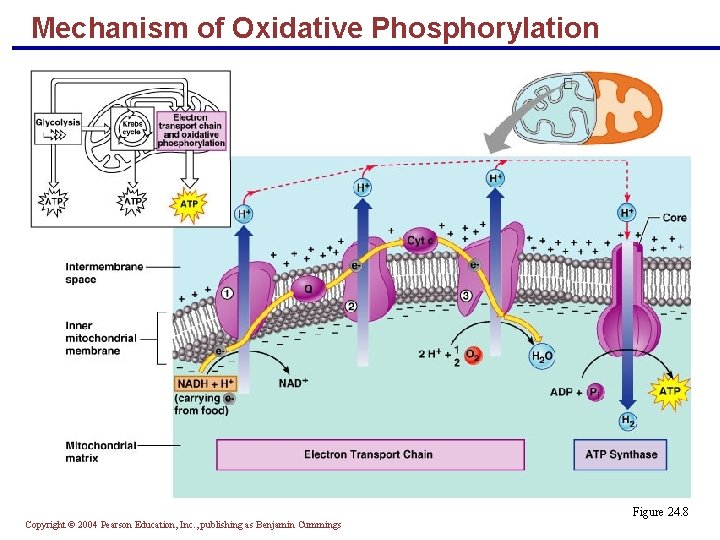
Mechanism of Oxidative Phosphorylation Figure 24. 8 Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Electronic Energy Gradient § The transfer of energy from NADH + H+ and FADH 2 to oxygen releases large amounts of energy § This energy is released in a stepwise manner through the electron transport chain Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Electronic Energy Gradient § The electrochemical proton gradient across the inner membrane: § Creates a p. H gradient § Generates a voltage gradient § These gradients cause H+ to flow back into the matrix via ATP synthase PLAY Electron Transport Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings
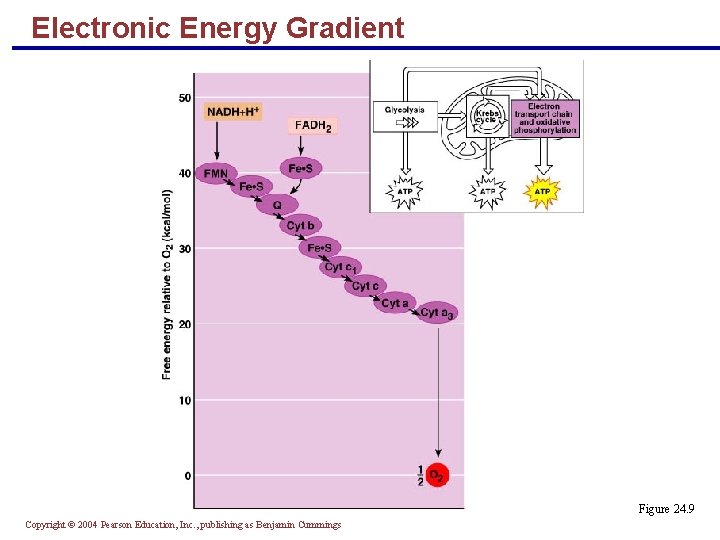
Electronic Energy Gradient Figure 24. 9 Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

ATP Synthase § The enzyme consists of three parts: a rotor, a knob, and a rod § Current created by H+ causes the rotor and rod to rotate § This rotation activates catalytic sites in the knob where ADP and Pi are combined to make ATP Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings
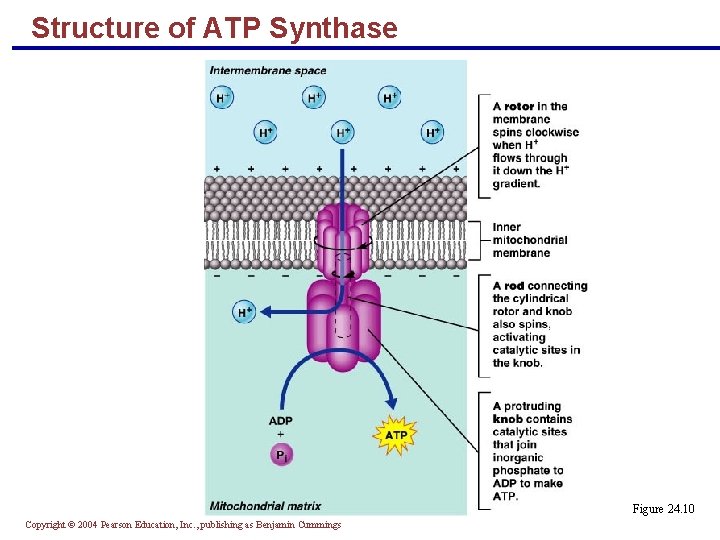
Structure of ATP Synthase Figure 24. 10 Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings
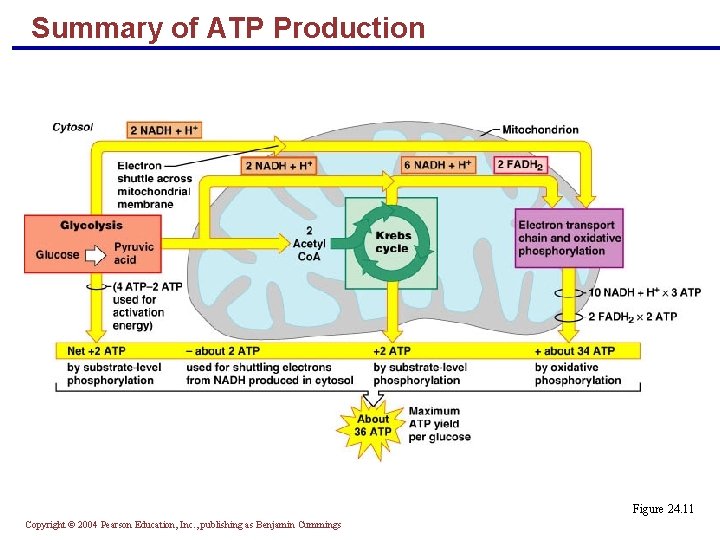
Summary of ATP Production Figure 24. 11 Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings
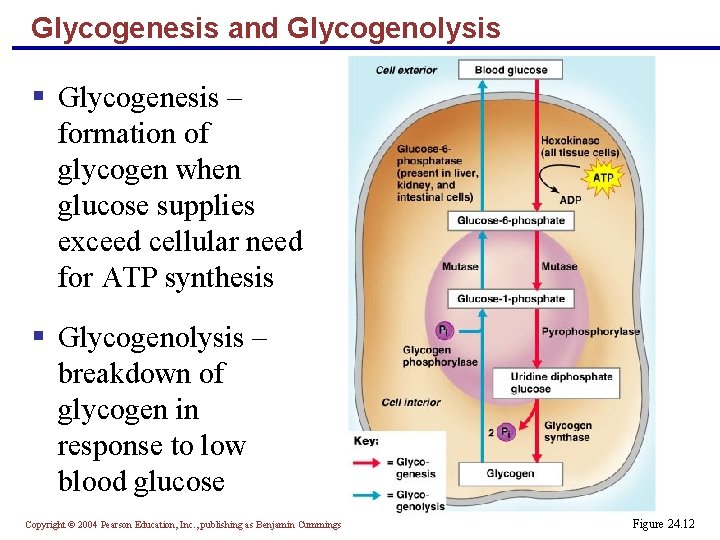
Glycogenesis and Glycogenolysis § Glycogenesis – formation of glycogen when glucose supplies exceed cellular need for ATP synthesis § Glycogenolysis – breakdown of glycogen in response to low blood glucose Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings Figure 24. 12

Gluconeogenesis § The process of forming sugar from noncarbohydrate molecules § Takes place mainly in the liver § Protects the body, especially the brain, from the damaging effects of hypoglycemia by ensuring ATP synthesis can continue Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Lipid Metabolism § Most products of fat metabolism are transported in lymph as chylomicrons § Lipids in chylomicrons are hydrolyzed by plasma enzymes and absorbed by cells § Only neutral fats are routinely oxidized for energy § Catabolism of fats involves two separate pathways § Glycerol pathway § Fatty acids pathway Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Lipid Metabolism § Glycerol is converted to glyceraldehyde phosphate § Glyceraldehyde is ultimately converted into acetyl Co. A § Acetyl Co. A enters the Krebs cycle § Fatty acids undergo beta oxidation which produces: § Two-carbon acetic acid fragments, which enter the Krebs cycle § Reduced coenzymes, which enter the electron transport chain Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings
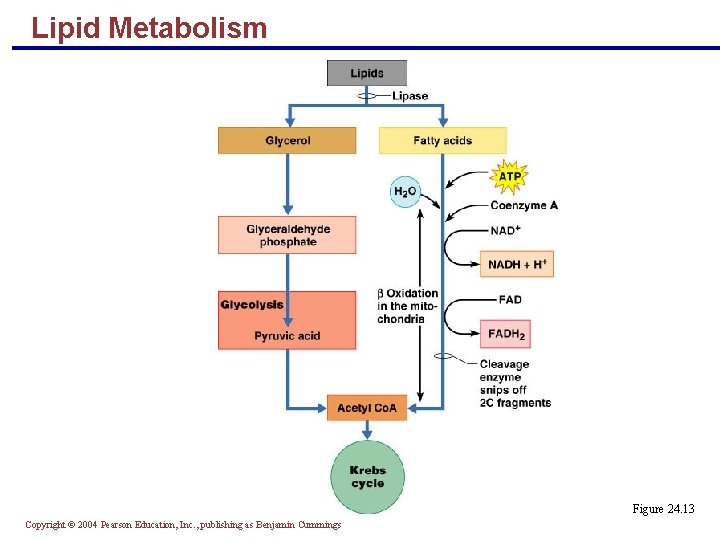
Lipid Metabolism Figure 24. 13 Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Lipogenesis and Lipolysis § Excess dietary glycerol and fatty acids undergo lipogenesis to form triglycerides § Glucose is easily converted into fat since acetyl Co. A is: § An intermediate in glucose catabolism § The starting molecule for the synthesis of fatty acids Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Lipogenesis and Lipolysis § Lipolysis, the breakdown of stored fat, is essentially lipogenesis in reverse § Oxaloacetic acid is necessary for the complete oxidation of fat § Without it, acetyl Co. A is converted into ketones (ketogenesis) Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings
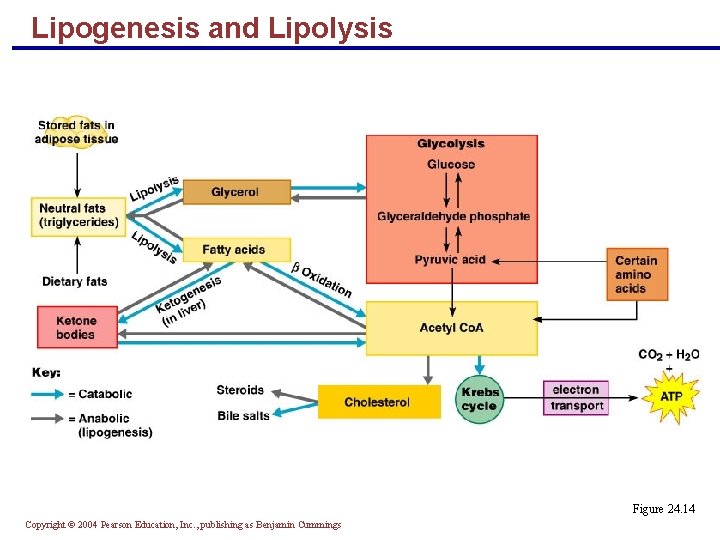
Lipogenesis and Lipolysis Figure 24. 14 Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Lipid Metabolism: Synthesis of Structural Materials § Phospholipids are important components of myelin and cell membranes Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Lipid Metabolism: Synthesis of Structural Materials § The liver: § Synthesizes lipoproteins for transport of cholesterol and fats § Makes tissue factor, a clotting factor § Synthesizes cholesterol for acetyl Co. A § Uses cholesterol to form bile salts § Certain endocrine organs use cholesterol to synthesize steroid hormones Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Protein Metabolism § Excess dietary protein results in amino acids being: § Oxidized for energy § Converted into fat for storage § Amino acids must be deaminated prior to oxidation for energy Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Protein Metabolism § Deaminated amino acids are converted into: § Pyruvic acid § One of the keto acid intermediates of the Krebs cycle § These events occur as transamination, oxidative deamination, and keto acid modification Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Oxidation of Amino Acids § Transamination – switching of an amine group from an amino acid to a keto acid (usually -ketoglutaric acid of the Krebs cycle) § Typically, glutamic acid is formed in this process Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Oxidation of Amino Acids § Oxidative deamination – the amine group of glutamic acid is: § Released as ammonia § Combined with carbon dioxide in the liver § Excreted as urea by the kidneys § Keto acid modification – keto acids from transamination are altered to produce metabolites that can enter the Krebs cycle Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Synthesis of Proteins § Amino acids are the most important anabolic nutrients, and they form: § All protein structures § The bulk of the body’s functional molecules Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Synthesis of Proteins § Amounts and types of proteins: § Are hormonally controlled § Reflect each life cycle stage § A complete set of amino acids is necessary for protein synthesis § All essential amino acids must be provided in the diet Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Summary: Carbohydrate Metabolic Reactions Table 24. 2. 1 Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Summary: Lipid and Protein Metabolic Reactions Table 24. 2. 2 Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

State of the Body § The body exists in a dynamic catabolic-anabolic state § Organic molecules (except DNA) are continuously broken down and rebuilt § The body’s total supply of nutrients constitutes its nutrient pool Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

State of the Body § Amino acid pool – body’s total supply of free amino acids is the source for: § Resynthesizing body proteins § Forming amino acid derivatives § Gluconeogenesis Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings
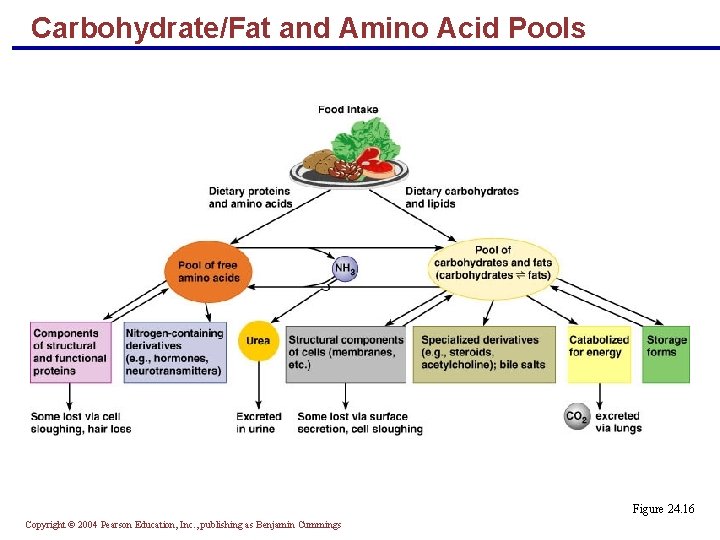
Carbohydrate/Fat and Amino Acid Pools Figure 24. 16 Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Interconversion Pathways of Nutrients § Carbohydrates are easily and frequently converted into fats § Their pools are linked by key intermediates § They differ from the amino acid pool in that: § Fats and carbohydrates are oxidized directly to produce energy § Excess carbohydrate and fat can be stored Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings
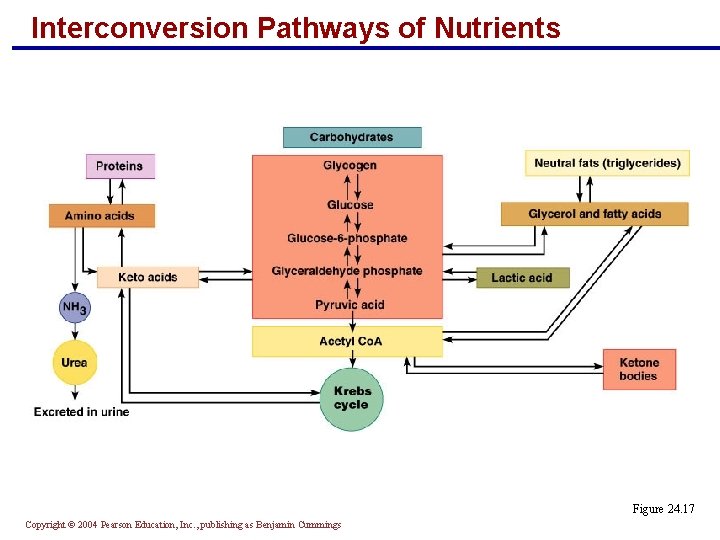
Interconversion Pathways of Nutrients Figure 24. 17 Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Absoprtive and Postabsorptive States § Metabolic controls equalize blood concentrations of nutrients between two states § Absorptive § The time during and shortly after nutrient intake § Postabsorptive § The time when the GI tract is empty § Energy sources are supplied by the breakdown of body reserves Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Absoprtive State § The major metabolic thrust is anabolism and energy storage § Amino acids become proteins § Glycerol and fatty acids are converted to triglycerides § Glucose is stored as glycogen § Dietary glucose is the major energy fuel § Excess amino acids are deaminated and used for energy or stored as fat in the liver Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings
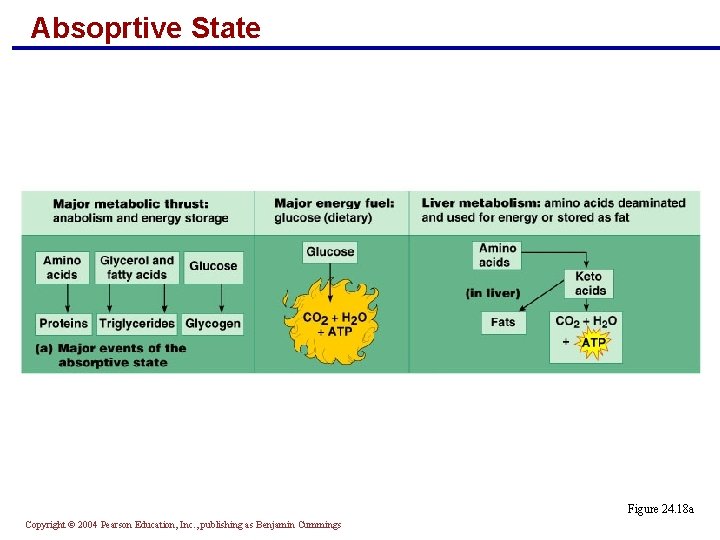
Absoprtive State Figure 24. 18 a Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Principal Pathways of the Absorptive State § In muscle: § Amino acids become protein § Glucose is converted to glycogen § In the liver: § Amino acids become protein or are deaminated to keto acids § Glucose is stored as glycogen or converted to fat Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Principal Pathways of the Absorptive State § In adipose tissue: § Glucose and fats are converted and stored as fat § All tissues use glucose to synthesize ATP Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings
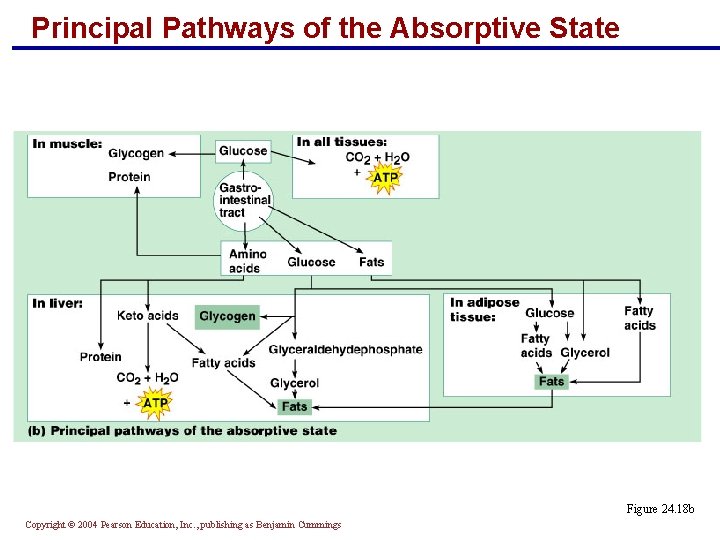
Principal Pathways of the Absorptive State Figure 24. 18 b Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Insulin Effects on Metabolism § Insulin controls the absorptive state and its secretion is stimulated by: § Increased blood glucose § Elevated amino acid levels in the blood § Gastrin, CCK, and secretin § Insulin enhances: § Active transport of amino acids into tissue cells § Facilitated diffusion of glucose into tissue Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings
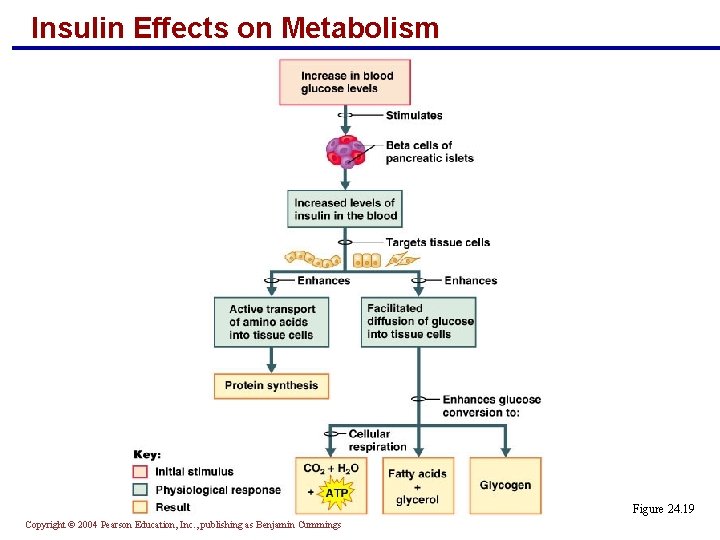
Insulin Effects on Metabolism Figure 24. 19 Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Diabetes Mellitus § A consequence of inadequate insulin production or abnormal insulin receptors § Glucose becomes unavailable to most body cells § Metabolic acidosis, protein wasting, and weight loss result as fats and tissue proteins are used for energy Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Postabsorptive State § The major metabolic thrust is catabolism and replacement of fuels in the blood § Proteins are broken down to amino acids § Triglycerides are turned into glycerol and fatty acids § Glycogen becomes glucose § Glucose is provided by glycogenolysis and gluconeogenesis § Fatty acids and ketones are the major energy fuels § Amino acids are converted to glucose in the liver Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings
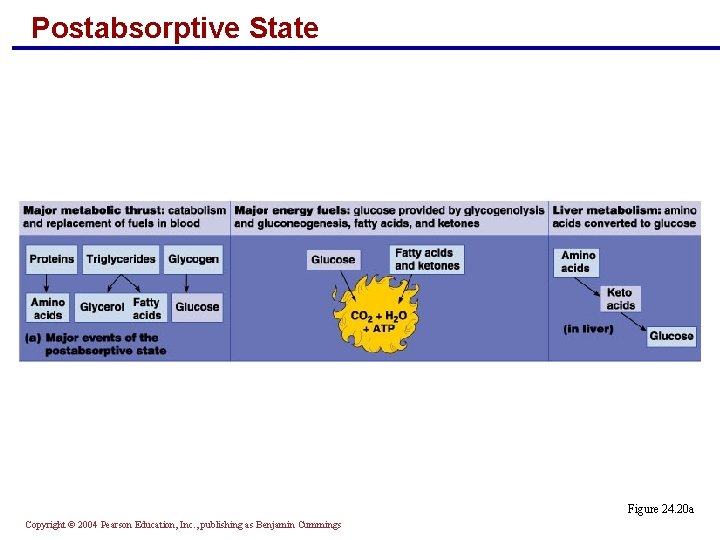
Postabsorptive State Figure 24. 20 a Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Principle Pathways in the Postabsorptive State § In muscle: § Protein is broken down to amino acids § Glycogen is converted to ATP and pyruvic acid (lactic acid in anaerobic states) Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Principle Pathways in the Postabsorptive State § In the liver: § Amino acids, pyruvic acid, stored glycogen, and fat are converted into glucose § Fat is converted into keto acids that are used to make ATP § Fatty acids (from adipose tissue) and ketone bodies (from the liver) are used in most tissue to make ATP § Glucose from the liver is used by the nervous system to generate ATP Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings
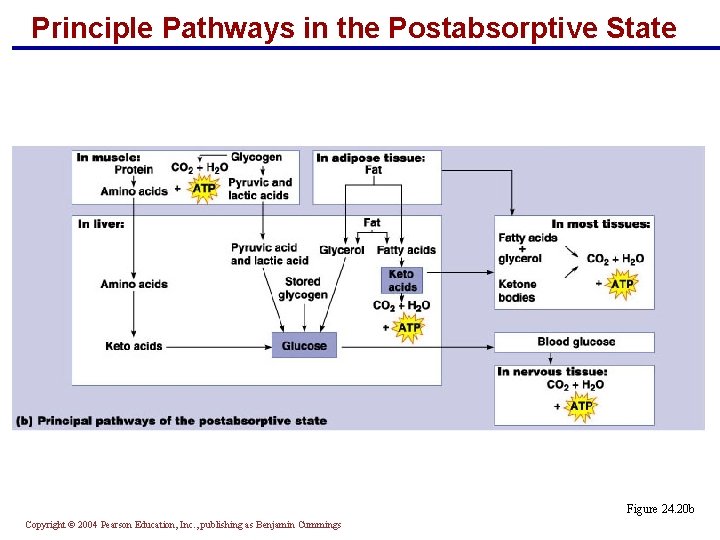
Principle Pathways in the Postabsorptive State Figure 24. 20 b Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Hormonal and Neural Controls of the Postabsorptive State § Decreased plasma glucose concentration and rising amino acid levels stimulate alpha cells of the pancreas to secrete glucagon (the antagonist of insulin) § Glucagon stimulates: § Glycogenolysis and gluconeogenesis § Fat breakdown in adipose tissue § Glucose sparing Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Hormonal and Neural Controls of the Postabsorptive State § In response to low plasma glucose, the sympathetic nervous system releases epinephrine, which acts on the liver, skeletal muscle, and adipose tissue to mobilize fat and promote glycogenolysis Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Liver Metabolism § Hepatocytes carry out over 500 intricate metabolic functions Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Liver Metabolism § A brief summary of liver functions § Packages fatty acids to be stored and transported § Synthesizes plasma proteins § Forms nonessential amino acids § Converts ammonia from deamination to urea § Stores glucose as glycogen, and regulates blood glucose homeostasis § Stores vitamins, conserves iron, degrades hormones, and detoxifies substances Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Cholesterol § Is the structural basis of bile salts, steroid hormones, and vitamin D § Makes up part of the hedgehog molecule that directs embryonic development § Is transported to and from tissues via lipoproteins Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Cholesterol § Lipoproteins are classified as: § HDLs – high-density lipoproteins have more protein content § LDLs – low-density lipoproteins have a considerable cholesterol component § VLDLs – very low density lipoproteins are mostly triglycerides Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings
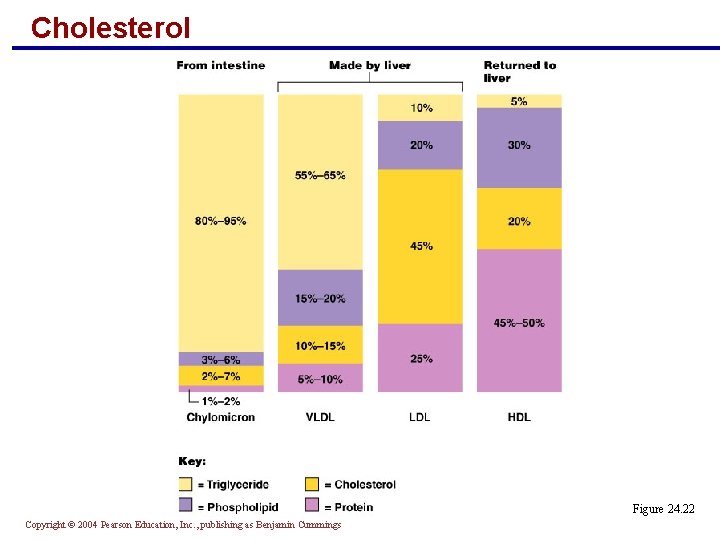
Cholesterol Figure 24. 22 Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Lipoproteins § The liver is the main source of VLDLs, which transport triglycerides to peripheral tissues (especially adipose) § LDLs transport cholesterol to the peripheral tissues and regulate cholesterol synthesis § HDLs transport excess cholesterol from peripheral tissues to the liver § Also serve the needs of steroid-producing organs (ovaries and adrenal glands) Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Lipoproteins § High levels of HDL are thought to protect against heart attack § High levels of LDL, especially lipoprotein (a), increase the risk of heart attack Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Plasma Cholesterol Levels § The liver produces cholesterol: § At a basal level of cholesterol regardless of dietary intake § Via a negative feedback loop involving serum cholesterol levels § In response to saturated fatty acids Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Plasma Cholesterol Levels § Fatty acids regulate excretion of cholesterol § Unsaturated fatty acids enhance excretion § Saturated fatty acids inhibit excretion § Certain unsaturated fatty acids (omega-3 fatty acids, found in cold-water fish) lower the proportions of saturated fats and cholesterol Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Non-Dietary Factors Affecting Cholesterol § Stress, cigarette smoking, and coffee drinking increase LDL levels § Aerobic exercise increases HDL levels § Body shape is correlated with cholesterol levels § Fat carried on the upper body is correlated with high cholesterol levels § Fat carried on the hips and thighs is correlated with lower levels Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Body Energy Balance § Bond energy released from catabolized food must equal the total energy output § Energy intake – equal to the energy liberated during the oxidation of food § Energy output includes the energy: § Immediately lost as heat (about 60% of the total) § Used to do work (driven by ATP) § Stored in the form of fat and glycogen Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Body Energy Balance § Nearly all energy derived from food is eventually converted to heat § Cells cannot use this energy to do work, but the heat: § Warms the tissues and blood § Helps maintain the homeostatic body temperature § Allows metabolic reactions to occur efficiently Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Regulation of Food Intake § When energy intake and energy outflow are balanced, body weight remains stable § The hypothalamus releases peptides that influence feeding behavior § Orexins are powerful appetite enhancers § Neuropeptide Y causes a craving for carbohydrates § Galanin produces a craving for fats § GLP-1 and serotonin make us feel full and satisfied Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Feeding Behaviors § Feeding behavior and hunger depend on one or more of five factors § Neural signals from the digestive tract § Bloodborne signals related to the body energy stores § Hormones, body temperature, and psychological factors Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Nutrient Signals Related to Energy Stores § High plasma levels of nutrients that signal depressed eating § Plasma glucose levels § Amino acids in the plasma § Fatty acids and leptin Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Hormones, Temperature, and Psychological Factors § Glucagon and epinephrine stimulate hunger § Insulin and cholecystokinin depress hunger § Increased body temperature may inhibit eating behavior § Psychological factors that have little to do with caloric balance can also influence eating behaviors Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Control of Feeding Behavior and Satiety § Leptin, secreted by fat tissue, appears to be the overall satiety signal § Acts on the ventromedial hypothalamus § Controls appetite and energy output § Suppresses the secretion of neuropeptide Y, a potent appetite stimulant § Blood levels of insulin and glucocorticoids play a role in regulating leptin release Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings
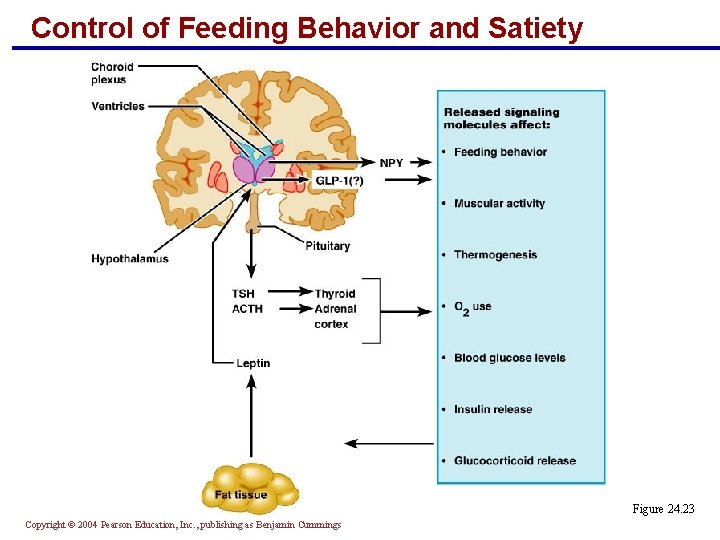
Control of Feeding Behavior and Satiety Figure 24. 23 Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Metabolic Rate § Rate of energy output (expressed per hour) equal to the total heat produced by: § All the chemical reactions in the body § The mechanical work of the body § Measured directly with a calorimeter or indirectly with a respirometer Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Metabolic Rate § Basal metabolic rate (BMR) § Reflects the energy the body needs to perform its most essential activities § Total metabolic rate (TMR) § Total rate of kilocalorie consumption to fuel all ongoing activities Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Factors that Influence BMR § Surface area, age, gender, stress, and hormones § As the ratio of surface area to volume increases, BMR increases § Males have a disproportionately high BMR § Stress increases BMR § Thyroxine increases oxygen consumption, cellular respiration, and BMR Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings
Regulation of Body Temperature § Body temperature – balance between heat production and heat loss § At rest, the liver, heart, brain, and endocrine organs account for most heat production § During vigorous exercise, heat production from skeletal muscles can increase 30– 40 times § Normal body temperature is 36. 2 C (98. 2 F); optimal enzyme activity occurs at this temperature § Temperature spikes above this range denature proteins and depress neurons Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings
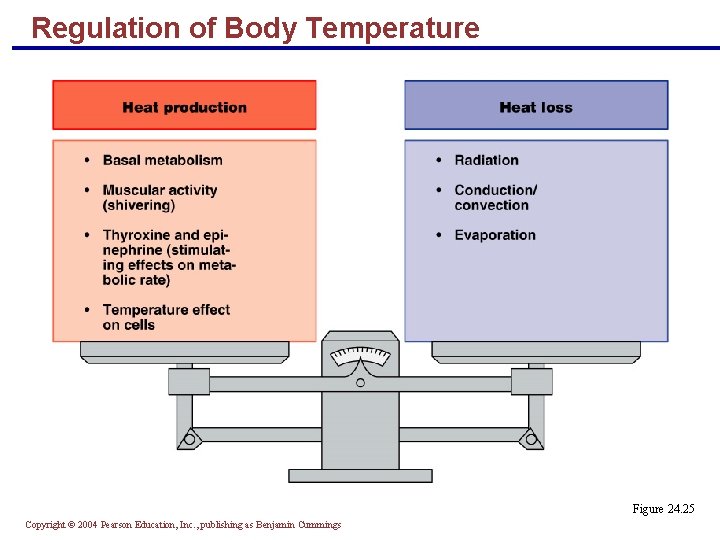
Regulation of Body Temperature Figure 24. 25 Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Core and Shell Temperature § Organs in the core (within the skull, thoracic, and abdominal cavities) have the highest temperature § The shell, essentially the skin, has the lowest temperature § Blood serves as the major agent of heat transfer between the core and shell § Core temperature remains relatively constant, while shell temperature fluctuates substantially (20 C– 40 C) Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Mechanisms of Heat Exchange § The body uses four mechanisms of heat exchange § Radiation – loss of heat in the form of infrared rays § Conduction – transfer of heat by direct contact § Convection – transfer of heat to the surrounding air § Evaporation – heat loss due to the evaporation of water from the lungs, mouth mucosa, and skin (insensible heat loss) § Evaporative heat loss becomes sensible when body temperature rises and sweating produces increased water for vaporization Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Role of the Hypothalamus § The main thermoregulation center is the preoptic region of the hypothalamus § The heat-loss and heat-promoting centers comprise thermoregulatory centers § The hypothalamus: § Receives input from thermoreceptors in the skin and core § Responds by initiating appropriate heat-loss and heat-promoting activities Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Heat-Promoting Mechanisms § Low external temperature or low temperature of circulating blood activates heat-promoting centers of the hypothalamus to cause: § Vasoconstriction of cutaneous blood vessels § Increased metabolic rate § Shivering § Enhanced thyroxine release Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Heat-Loss Mechanisms § When the core temperature rises, the heat-loss center is activated to cause: § Vasodilation of cutaneous blood vessels § Enhanced sweating § Voluntary measures commonly taken to reduce body heat include: § Reducing activity and seeking a cooler environment § Wearing light-colored and loose-fitting clothing Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Mechanisms of Body Temperature Regulation Figure 24. 27 Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Hyperthermia § Normal heat loss processes become ineffective and elevated body temperatures depress the hypothalamus § This sets up a positive-feedback mechanism, sharply increasing body temperature and metabolic rate § This condition, called heat stroke, can be fatal if not corrected Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Heat Exhaustion § Heat-associated collapse after vigorous exercise, evidenced by elevated body temperature, mental confusion, and fainting § Due to dehydration and low blood pressure § Heat-loss mechanisms are fully functional § Can progress to heat stroke if the body is not cooled and rehydrated Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Fever § Controlled hyperthermia, often a result of infection, cancer, allergic reactions, or central nervous system injuries § White blood cells, injured tissue cells, and macrophages release pyrogens that act on the hypothalamus, causing the release of prostaglandins § Prostaglandins reset the hypothalamic thermostat § The higher set point is maintained until the natural body defenses reverse the disease process Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Developmental Aspects § Good nutrition is essential in utero as well as throughout life § Lack of proteins needed for fetal growth and in the first three years of life can lead to mental deficits and learning disorders § With the exception of insulin-dependent diabetes mellitus, children free of genetic disorders rarely exhibit metabolic problems § In later years, non-insulin-dependent diabetes mellitus becomes a major problem Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings

Developmental Aspects § Many agents prescribed for age-related medical problems influence nutrition § Diuretics can cause hypokalemia by promoting potassium loss § Antibiotics can interfere with food absorption § Mineral oil interferes with absorption of fat-soluble vitamins § Excessive alcohol consumption leads to malabsorption problems, certain vitamin and mineral deficiencies, deranged metabolism, and damage to the liver and pancreas Copyright © 2004 Pearson Education, Inc. , publishing as Benjamin Cummings
- Slides: 121