Nutrition in Inflammatory Bowel Disease and the Role

Nutrition in Inflammatory Bowel Disease and the Role of Parenteral Nutrition Heather Janicki, RD, CNSC

Objectives • Nutritional considerations of the patient with inflammatory bowel disease (IBD) • Oral and nutrition support for the IBD patient • Complications of IBD patients

Inflammatory Bowel Disease (IBD) • Inflammatory disorder of the gastrointestinal tract – Symptoms include: • • Pain Nausea Fever Diarrhea – Symptoms can lead to impaired nutritional status through • Loss of appetite • Reduced nutrient intake • Altered nutrient metabolism
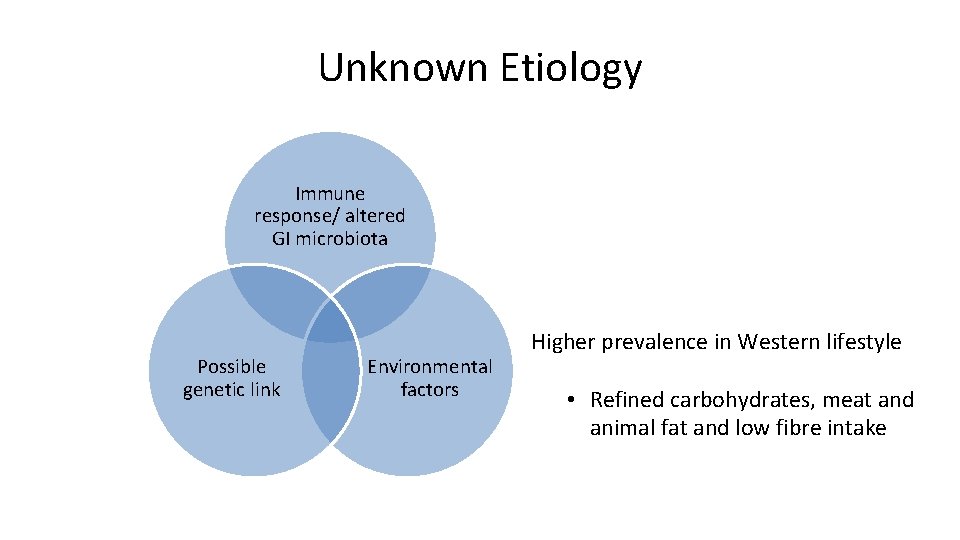
Unknown Etiology Immune response/ altered GI microbiota Possible genetic link Environmental factors Higher prevalence in Western lifestyle • Refined carbohydrates, meat and animal fat and low fibre intake
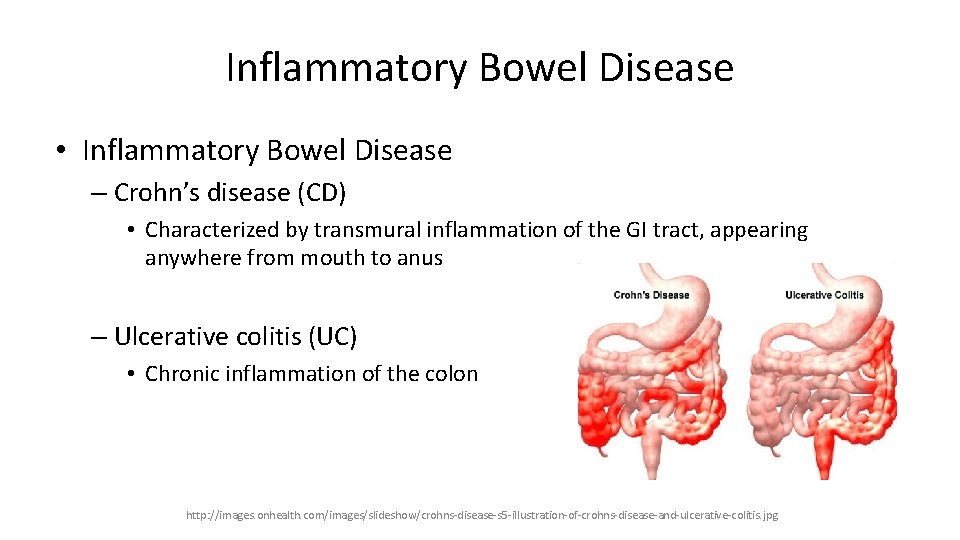
Inflammatory Bowel Disease • Inflammatory Bowel Disease – Crohn’s disease (CD) • Characterized by transmural inflammation of the GI tract, appearing anywhere from mouth to anus – Ulcerative colitis (UC) • Chronic inflammation of the colon http: //images. onhealth. com/images/slideshow/crohns-disease-s 5 -illustration-of-crohns-disease-and-ulcerative-colitis. jpg

Crohn’s Disease • Nutritional deficiencies and malnutrition have been well documented – Up to 80% of patients may have some degree of malnutrition • 65 -75% of inpatients and over 50% of outpatients with CD experience significant weight loss – Result of: • • • Decreased oral intake/ fear of eating Abdominal pain Diarrhea/ nausea Strictures Malabsorption (previous resections)

Crohn’s Disease • Further complications affecting nutrition therapy – Fistulas – Bowel perforations or abscess – Bowel stricture or obstruction – Previous resections or surgeries leading to short bowel syndrome

Crohn’s Disease • Nutrition needs – Energy requirement • Elevated resting energy expenditure during active inflammation – Protein requirement: 1 -1. 5 g/ kg, up to 2 g/ kg with malnutrition • Increased with active inflammation or losses due to fistulas etc. • Increased in post operative period – Fibre • No proven regimen for fibre intake although common for CD patients to follow fibre diet • No controlled trials show a benefit in a low fibre diet for symptom improvement, decreased admissions or incidence of small bowel obstructions

Crohn’s Disease • Micronutrients – Iron deficiency anemia- secondary to blood loss and malabsorption – 35 -40% estimated incidence of osteopenia • Corticosteroids, malabsorption • Up to 1500 mg calcium per day, 800 -1000 IU of vitamin D per day, vitamin K – Vitamin B 12, fat soluble vitamins (A, D, E, K) if terminal ileum disease – Folate- methotrexate use – Zinc deficiency- high output enterocutaneous fistulas or diarrhea – Ileostomy patients • May require additional fluid and sodium
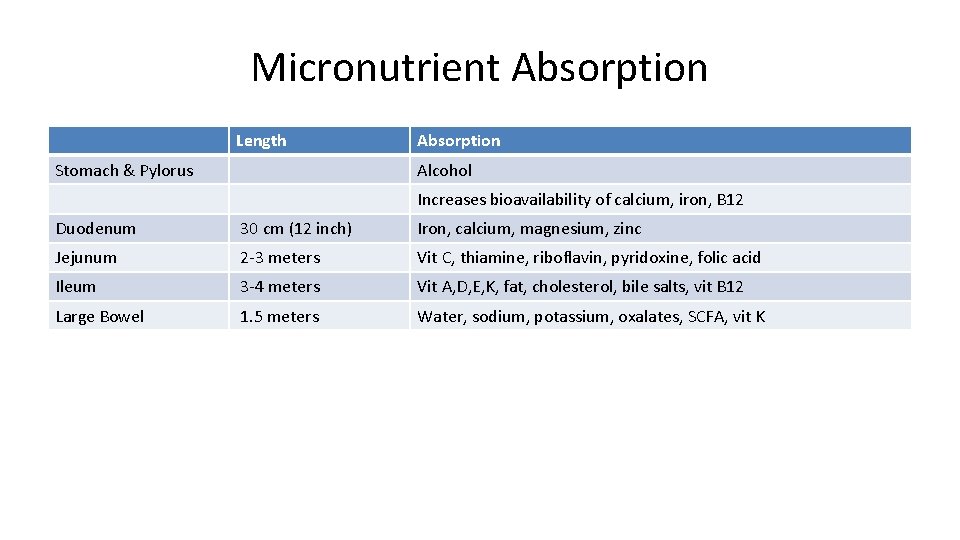
Micronutrient Absorption Length Stomach & Pylorus Absorption Alcohol Increases bioavailability of calcium, iron, B 12 Duodenum 30 cm (12 inch) Iron, calcium, magnesium, zinc Jejunum 2 -3 meters Vit C, thiamine, riboflavin, pyridoxine, folic acid Ileum 3 -4 meters Vit A, D, E, K, fat, cholesterol, bile salts, vit B 12 Large Bowel 1. 5 meters Water, sodium, potassium, oxalates, SCFA, vit K

Ulcerative Colitis (UC) • Protein-energy malnutrition is less common than CD as small bowel is not affected • Nutrition needs – Energy requirements • Elevated resting energy expenditure during active inflammation – Protein requirements: 1 -1. 5 g/ kg, up to 2 g/ kg with malnutrition • Increased with active inflammation and in post operative period • Potential role of increased fibre diet – Short chain fatty acids (butyrate) produced in the colon from fermentation of dietary fibre by colonic microorganisms – Serve as fuel for the colonocytes and promote water and sodium absorption

Ulcerative Colitis • Micronutrients – Up to 80% may develop iron deficiency anemia secondary to blood loss and malabsorption • Higher incidence than CD – 25 -32% estimated incidence of osteopenia • Corticosteroids, malabsorption • Up to 1500 mg calcium per day, 800 -1000 IU of vitamin D per day – Recurrent pouchitis or management of UC, probiotic VSL -3 (3 -6 g/ d) may offer some benefit in preventing relapse • Research is still in the early stages

Nutrition Assessment • Diagnosis • History of presenting illness including nutrition and weight loss history • Past medical and surgical history • Medications • Nutritionally relevant blood work • Anthropometrics- height/ weight, significant changes • Diet history, food intolerances and allergies, access to food

Refeeding Syndrome • Screen for patients at moderate to severe malnutrition • Severe weight loss • Aggressive feeding of dextrose can cause electrolyte abnormalities and multiple serious complications • If electrolyte levels are low (K, Mg, PO 4) replete before starting PN • Monitor closely • Start with 100 grams dextrose/ day, lower calories • Low sodium, may need lower volume to start • Monitor K, Mg, PO 4 closely. May need higher end of range in PN or additional boluses.

Nutrition Therapy • Determine appropriate route of feeding (oral, enteral and/ or parenteral), what to feed and how aggressive to feed • Supplementation required (vitamins, minerals, protein) • Any diet restrictions required Monitoring • Amount of prescribed nutrition received • Tolerance to nutrition regime, bowel care • Changing clinical picture and requirements • Education needs

Benefits of Nutrition Therapy • • • Prevents or treats malnutrition and micronutrient deficiencies Helps prevent/ reduce loss of lean body mass Improves GI tract structure and functions Improves wound healing Decreased hospital admission and length of stay • Consequences of Malnutrition – – Growth failure in pediatrics Weight loss Bone disease Micronutrient deficiencies

Oral Diet in IBD • No diet restrictions, especially when in disease remission • Based on individual tolerance • During flare up – May have a temporary intolerance to lactose – Some may prefer low fibre foods • Crohn’s patients with strictures – Avoid high fibre foods- nuts, seeds, tough fruit/ vegetable skins, corn, popcorn, celery • Patients with ileostomies – May require additional fluid and sodium

Enteral Nutrition (EN) • Commonly given via a nasogastric tube or can be provided orally • Polymeric formulas: – Contains whole proteins, carbohydrates etc and are palatable • Semi-elemental or elemental formulas – Contains nutrients that are partially or fully broken down – Require little to no digestion prior to absorption • Studies have not shown any difference between formulas

Enteral Nutrition • Indicated in both CD and UC to meet nutritional needs • EN may be the first line of therapy in some children to promote remission of CD – Given exclusively for 6 -8 weeks, mostly as polymeric – Lower recurrence rates in patients treated with EN versus normal diet • EN provided as 50% of calories • Adult patients: may be less effective as a sole treatment compared to corticosteroids in CD • Evidence is limited to support the use of EN as primary therapy for treatment of UC • Can be used in active flares, similar rates of remission and need for surgery compared to PN

Enteral Nutrition • Mechanism of how EN improves outcomes in CD is unclear. Theories include: – Altered or reduced gut microbiota – Avoidance of food additives- emulsifiers, colourings etc – Prevention/ correction of malnutrition- improved wound healing and gut permeability • Benefits: – – Maintain gut integrity No long term adverse effects Cost efficient Fewer complications than parenteral nutrition

Enteral Nutrition • Long-term challenges: – Access- Naso gastric tube versus G- tube – Palatability, length of time without solid food – Cost of EN formulas – High relapse rate when patient returns to normal diet • ? Weaning period of 2 -4 weeks may help relapse – EN provided as 50% of calories

Parenteral Nutrition • Infusion of dextrose, amino acids, IV fat emulsion (IVFE), electrolytes, vitamins and trace elements through an IV • Goal: prevent or correct nutritional deficiencies and malnutrition when the function of the GI tract is inadequate or bowel rest indicated • 2 types – Central Parenteral Nutrition (CPN) often referred to as Total Parenteral Nutrition (TPN) – Peripheral Parenteral Nutrition (PPN)

Parenteral Nutrition Objectives • Maintain or replace lean body mass, macronutrient and micronutrient deficiencies when enteral/ oral feeding is contraindicated • Provide fluids with care to prevent over-hydration or dehydration • Advance to oral/ enteral feeds as soon as possible • Minimize complications

Parenteral Nutrition • Not recommended for primary or maintenance therapy in CD or UC • Historically used for patients on bowel rest – Bowel rest- limit intestinal exposure to proinflammatory agents, allow for mucosal healing and reduce risk of surgical interventions – No conclusive role for TPN or bowel rest. • May reduce bowel movement frequency and stool weight • May reduce need for surgery in CD but not UC patients • No difference at one year remission rates for PN and bowel rest, EN or supplementary PN with unrestricted food

Parenteral Nutrition Indications • Essential in very severe cases – When enteral nutrition is not tolerated • Inability to maintain weight and nutrition status on oral and/ or enteral nutrition – Short bowel syndrome – Small bowel obstruction – High output fistulas when EN is not feasible – Intractable diarrhea/vomiting – Prolonged ileus – NPO > 7 days

Fistulas • Enterocutaneous fistulas: – Abnormal communication between the intestine and the skin – Often occur 7 -10 days after surgery – Enteral nutrition indicated: • low output fistulas (<500 m. L/d) • Proximal fistula if able to feed distally • Distal ileal or colonic fistulas – Parenteral nutrition indicated: • Pancreatic fistula • Jejunal/ ileal high output fistula • Any fistula when distal access is not possible

Short Bowel Syndrome • Inadequate absorption of the small bowel resulting from loss of bowel length and/ or function • < 200 cm of small bowel remaining • >70 -90 cm of small bowel with intact colon can usually regain autonomy from PN • 130 -150 cm if colon is removed • Factors affecting adaptation of the bowel – Length of remaining bowel/ health of the bowel – Section of bowel resected

Short Bowel Syndrome • Primary goal is to regain/ maintain as much gut function possible – First phase- post operative- significant fluid loses and hypersecretion of gastric fluids – Second phase- gut adaptation period up to 2 years post operative – Must determine the length of bowel remaining, terminal ileum and amount of colon remaining – Vitamin/ mineral levels should be monitored. At baseline, 3 months post surgery and then annually • At risk for fat soluble vitamin deficiencies, vitamin B 12 and zinc

Parenteral Nutrition Contraindications • Functional GI Tract • Previously well nourished • TPN for less than 7 days in well nourished patients • Poor prognosis • Risks exceed benefit

Parenteral Nutrition Complications • PN can be life saving/ sustaining but can also be associated with serious complications – Macronutrient (hyperglycemia, azotemia etc. ) – Fluid and electrolyte, acid base imbalance – Micronutrient ( may require extra zinc, selenium, TPN contains no iron, excess manganese, copper) – Hepatobiliary (parenteral nutrition associated liver disease) – Metabolic bone disease – Infections

Parenteral Nutrition Monitoring • Bloodwork- electrolytes, triglyceride, glucose, liver/ renal function • Weight- dehydration, fluid retention • Ins and outs • Medical condition- continually re-assess for need for PN • Line infection/ complications
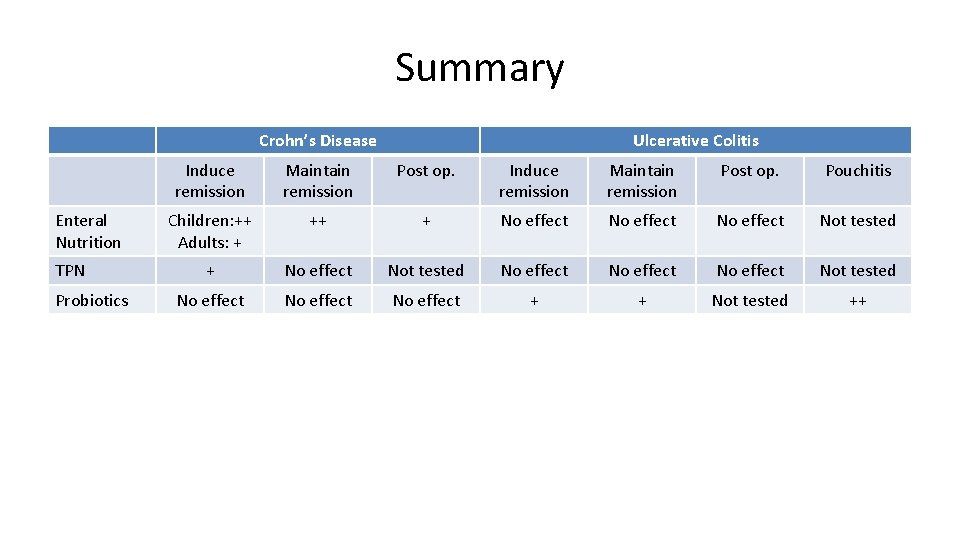
Summary Crohn’s Disease Enteral Nutrition TPN Probiotics Ulcerative Colitis Induce remission Maintain remission Post op. Pouchitis Children: ++ Adults: + ++ + No effect Not tested + No effect Not tested No effect No effect + + Not tested ++

Questions

References • Mueller, C. , Mc. Clave, S. , & Kuhn, J. M. (2012). The A. S. P. E. N. adult nutrition support core curriculum. Silver Spring, MD: American Society for Parenteral and Enteral Nutrition. • (n. d. ). Retrieved September 10, 2017, from http: //www. uptodate. com/contents/nutritionand-dietary-interventions-in-adults-with-inflammatory-bowel-disease • Shah N, Parian A, Mullin G, Limketkai B. Oral Diets and Nutrition Support for Inflammatory Bowel Disease: What Is The Evidence? Nutrition in Clinical Practice 2015; 30(4): 462 -473 • Triantafillidis J, Papalois A. The role or total parenteral nutrition in inflammatory bowel disease: current aspects. Scandinavian Journal of Gastroenterology 2014; 49: 3 -14 • Durchschein F, Petritsch W, Hammer H. Diet therapy for inflammatory bowel disease: The established and the new. World J Gastroenterol 2016; 22(7): 2179 -2194 • Altamore R, Damiano G, Abruzzo A, Palumbo V, Tomasello G, Buscemi S, Monte A. Enteral Nutrition Support to Treat Malnutrition in Inflammatory Bowel Disease. Nutrients 2015: 7: 2125 -2133
- Slides: 34