Nutrition for Preterm and Low Birth Weight Infants

Nutrition for Preterm and Low Birth Weight Infants
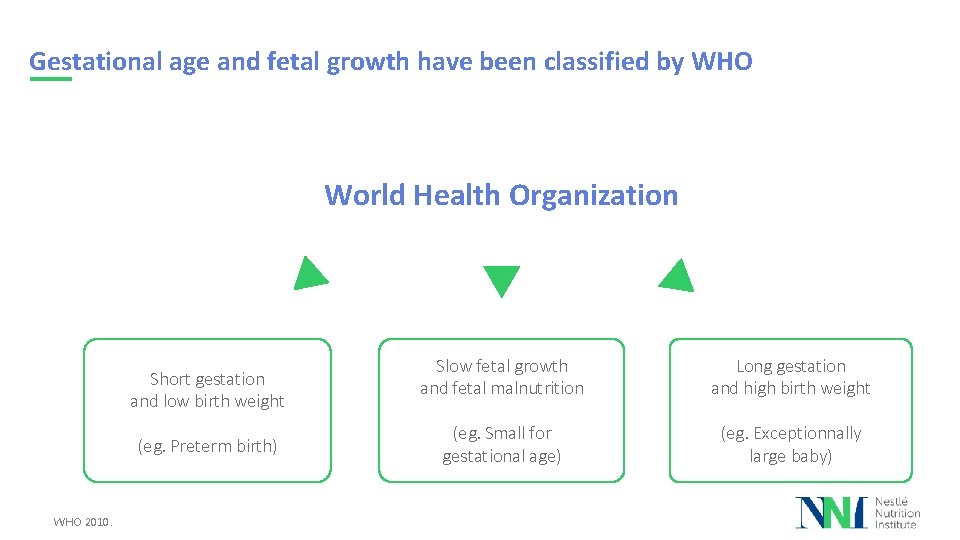
Gestational age and fetal growth have been classified by WHO World Health Organization Short gestation and low birth weight (eg. Preterm birth) WHO 2010. Slow fetal growth and fetal malnutrition Long gestation and high birth weight (eg. Small for gestational age) (eg. Exceptionnally large baby)

Low birth weight infants • Regardless of the gestational age, infants are classified based on their birth weight: Terminology Body weight at birth Low birth weight (LBW) <2, 500 g Very low birth weight (VLBW) <1, 500 g Extremely low birth (weight ELBW) <1, 000 g Note: Low birth weight can be a consequence of preterm birth (before 37 completed weeks of gestation), small size for gestational age (SGA, defined as weight for gestational age less than 10 th percentile), or a combination of both. WHO 2010; Optimal feeding of low-birth-weight infants, WHO 2006.

Preterm infants • Infants are classified based on the number of weeks in the uterus (gestational age)*: Terminology Gestational age Preterm <37 weeks Moderate/late preterm 32 -37 weeks Very preterm 28 -32 weeks Extremely preterm <28 weeks • Subcategories of preterm birth Very preterm Extremely preterm 1… 28 Moderate/late preterm 32 Term 37 40 Gestational age of pregnancy (weeks) *the number of weeks that a baby has been in the uterus, which is calculated from the first day of the mother's last menstrual period until the day of delivery WHO Fact sheet Preterm Birth, 19 February 2018 (Accessed on 23 oct 2018: http: //www. who. int/news-room/fact-sheets/detail/preterm-birth). WHO 2010
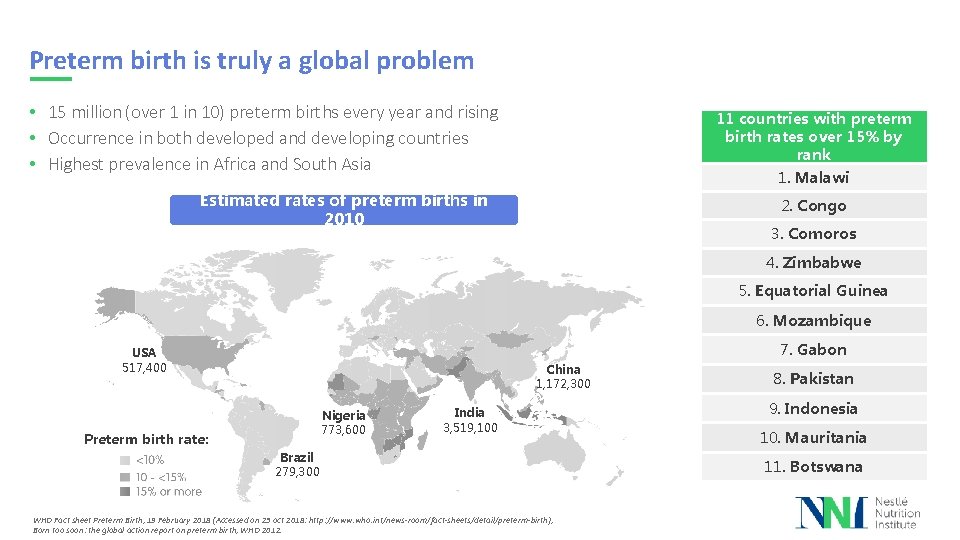
Preterm birth is truly a global problem • 15 million (over 1 in 10) preterm births every year and rising • Occurrence in both developed and developing countries • Highest prevalence in Africa and South Asia 11 countries with preterm birth rates over 15% by rank 1. Malawi Estimated rates of preterm births in 2010 2. Congo 3. Comoros 4. Zimbabwe 5. Equatorial Guinea 6. Mozambique 7. Gabon USA 517, 400 China 1, 172, 300 Nigeria 773, 600 Preterm birth rate: India 3, 519, 100 Brazil 279, 300 WHO Fact sheet Preterm Birth, 19 February 2018 (Accessed on 23 oct 2018: http: //www. who. int/news-room/fact-sheets/detail/preterm-birth), Born too soon: the global action report on preterm birth, WHO 2012. 8. Pakistan 9. Indonesia 10. Mauritania 11. Botswana
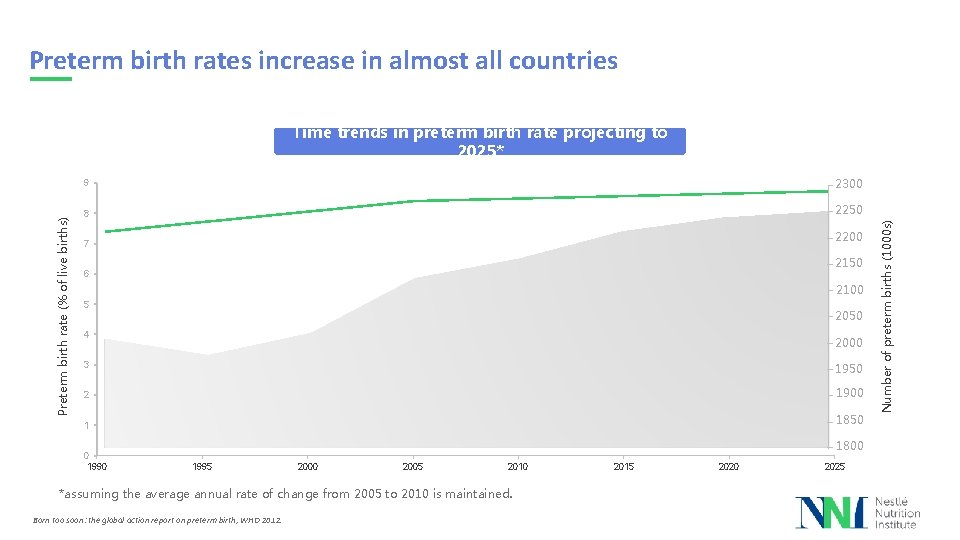
Preterm birth rates increase in almost all countries 9 2300 8 2250 2200 7 2150 6 2100 5 2050 4 2000 3 1950 2 1900 1 1850 0 1990 1800 1995 2000 2005 2010 *assuming the average annual rate of change from 2005 to 2010 is maintained. Born too soon: the global action report on preterm birth, WHO 2012. 2015 2020 2025 Number of preterm births (1000 s) Preterm birth rate (% of live births) Time trends in preterm birth rate projecting to 2025*

Prematurity is characterized by physiological immaturity • Gastro-intestinal (GI) system – The mucosal barrier function and immune responses are immature and inefficient – Many of the enzymes, gastric and bile acids and hormones needed for protein, lipid and carbohydrate digestion and absorption are insufficiently mature – The esophageal sphincter is inefficient, the gastric emptying is slower and the intestinal motility is disorganized • Immune system – Reduced innate and acquired immunity – Limited maternal antibodies placental transfer • Renal system – Incomplete nephrons (functional units of the kidney) development and functionality – Limited water excretion and urine concentration capacities • Respiratory system – Significant alterations in lung functions and physiology – Incomplete lung development and limited amounts of pulmonary surfactant that serves for oxygen utilization Neu 2007 and 2008, Premji 1998, Harding 2004, Colin 2010, Fraser 2004, Tissières 2012, Melville 2013, Carmody 2013. • Skin barrier – Water/nutrient losses and thermal instability

Preterm infants face specific health risks due to physiological immaturity Infections Respiratory Distress Syndrome Jaundice and higher risk for the brain Born too soon: the global action report on preterm birth, WHO 2012. Feeding difficulties and metabolic disturbances Morbidity is inversely related to gestational age Anemia of prematurity Brain injury NEC (Necrotizing Enterocolitis) Retinopathy of prematurity

Key challenges in preterm & LBW infants: 1. High growth rate 2. Depleted nutrient stores 3. GI immaturity

Growth is of key importance in preterm infants • Optimal growth – is not precisely defined in preterm infants, yet the most commonly applied and accepted standard for postnatal growth is that of intra-uterine growth • Growth faltering – refers to suboptimal growth or failure to thrive • Catch-up growth – refers to the acceleration of the growth rate following a period of growth retardation Poindexter 2014, Mc. Leod 2007.
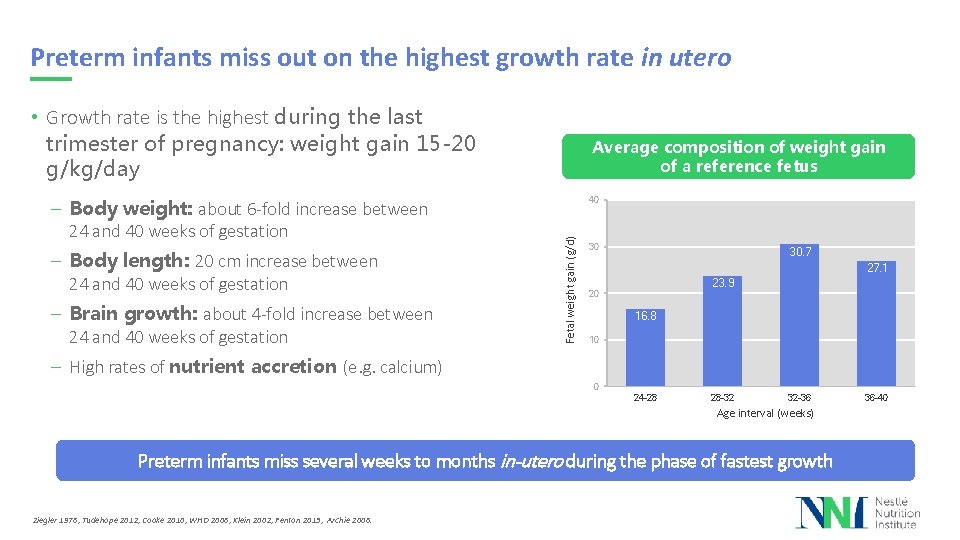
Preterm infants miss out on the highest growth rate in utero • Growth rate is the highest during the last trimester of pregnancy: weight gain 15 -20 g/kg/day – Body length: 20 cm increase between 24 and 40 weeks of gestation – Brain growth: about 4 -fold increase between 24 and 40 weeks of gestation 40 Fetal weight gain (g/d) – Body weight: about 6 -fold increase between 24 and 40 weeks of gestation Average composition of weight gain of a reference fetus 30 30. 7 27. 1 23. 9 20 16. 8 10 – High rates of nutrient accretion (e. g. calcium) 0 24 -28 28 -32 32 -36 Age interval (weeks) Preterm infants miss several weeks to months in-utero during the phase of fastest growth Ziegler 1976, Tudehope 2012, Cooke 2010, WHO 2006, Klein 2002, Fenton 2013, Archie 2006. 36 -40

Healthy catch-up growth is desired in preterm infants • Risks of slow or incomplete catch-up growth: – Poorer cognitive and educational outcomes – Higher risk of neurodevelopmental impairment – Prolonged growth restriction – Organ growth failure • Risks of a too rapid catch-up growth, later in life: – Altered fat distribution – Increased risk of obesity and insulin resistance – Increased risk of cardiovascular disease Young 2012, Hack 1991, Cooke 2003 & 2010, Hay 2008, Singhal 2003.

The challenge of growth in preterm and LBW infants • Preterm infants should grow at the same rate as the healthy unborn fetus • This growth rate, considerably faster than in term infants, requires a large amount of nutrients (early growth velocity rates exert a significant effect on neurodevelopment and growth patterns in later life) • However, due to feeding difficulties and medical instability, this can be challenging to achieve Fenton 2013, Koletzko 2014.

Depleted nutrient stores affect growth in preterm and LBW infants • Preterm and LBW infants are often born with depleted nutrient stores – Depletion in iron, calcium, vitamin A and lipids Lapillonne 2014, Young 2012.

Depleted nutrient stores in preterm and LBW infants • Infants normally constitute their nutrient stores during the third trimester of pregnancy • Preterm infants, however, are born before this critical time • This renders them especially vulnerable to poor growth and nutritional deficiencies Agostoni 2010, Koletzko 2014.

The burden of GI immaturity • In preterm and LBW infants, a number of functional aspects of the gastrointestinal (GI) system remain immature at birth • These include, most importantly poor digestion and absorption, as well as disorganized intestinal motility Neu 2007, Stratiki 2007, Gritz 2015.
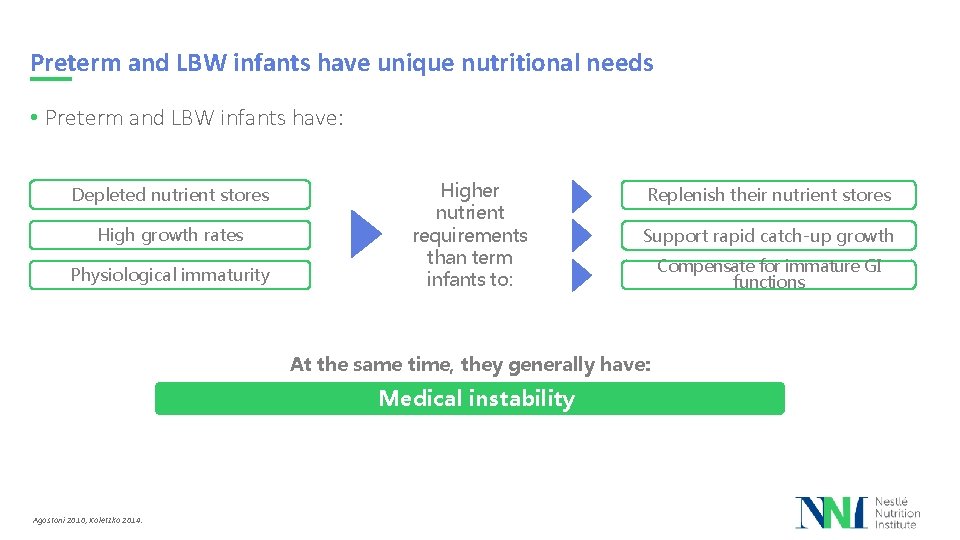
Preterm and LBW infants have unique nutritional needs • Preterm and LBW infants have: Depleted nutrient stores High growth rates Physiological immaturity Higher nutrient requirements than term infants to: Replenish their nutrient stores Support rapid catch-up growth At the same time, they generally have: Medical instability Agostoni 2010, Koletzko 2014. Compensate for immature GI functions

More energy is required in preterm and LBW infants • Compared to term infants, preterm and LBW infants have: • Increased energy requirements – Due to higher basal metabolic rate and high growth rate Ziegler 2007, Thureen 2007, Nzegwu 2014, Koletzko 2014, Agostoni 2010. Main recommendations: Energy Koletzko 2014 ESPGHAN 2010 kcal/kg/d 110 -130 110 -135
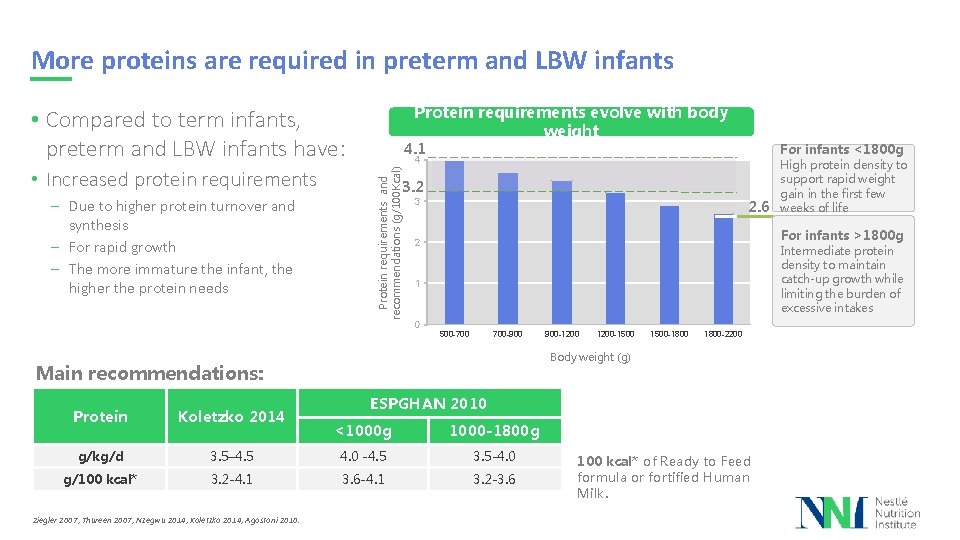
More proteins are required in preterm and LBW infants Protein requirements evolve with body weight • Compared to term infants, preterm and LBW infants have: – Due to higher protein turnover and synthesis – For rapid growth – The more immature the infant, the higher the protein needs 4 Protein requirements and recommendations (g/100 Kcal) • Increased protein requirements 4. 1 High protein density to support rapid weight gain in the first few 2. 6 weeks of life 3. 2 3 For infants >1800 g 2 Intermediate protein density to maintain catch-up growth while limiting the burden of excessive intakes 1 0 500 -700 700 -900 Koletzko 2014 g/kg/d g/100 kcal* 1200 -1500 -1800 -2200 ESPGHAN 2010 <1000 g 1000 -1800 g 3. 5– 4. 5 4. 0 -4. 5 3. 5 -4. 0 3. 2 -4. 1 3. 6 -4. 1 3. 2 -3. 6 Ziegler 2007, Thureen 2007, Nzegwu 2014, Koletzko 2014, Agostoni 2010. 900 -1200 Body weight (g) Main recommendations: Protein For infants <1800 g 100 kcal* of Ready to Feed formula or fortified Human Milk.
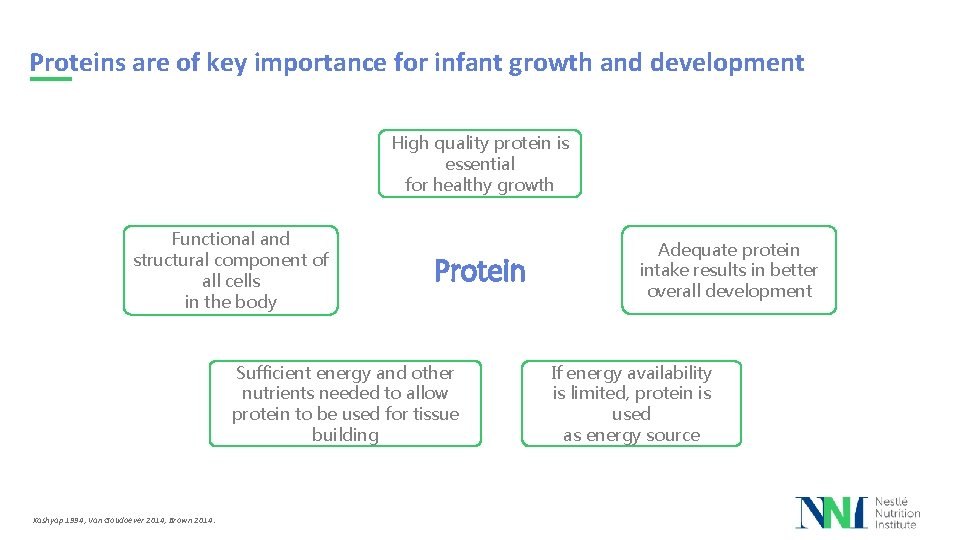
Proteins are of key importance for infant growth and development High quality protein is essential for healthy growth Functional and structural component of all cells in the body Protein Sufficient energy and other nutrients needed to allow protein to be used for tissue building Kashyap 1994, Van Goudoever 2014, Brown 2014. Adequate protein intake results in better overall development If energy availability is limited, protein is used as energy source

More LC-PUFAs, vitamins and minerals are needed in preterm & LBW infants • Compared to term infants, preterm and LBW infants have: – Increased calcium and phosphorus requirements for bone formation – Higher needs for vitamins and minerals due to limited body stores and high needs to support growth – Higher fluid needs (15 -25%) due to higher insensible water loss through immature skin and higher ratio of surface area to body weight – Slightly higher needs of PUFAs for growth and development Aggarwal 2001, Koletzko 2014, Fleith 2005.

Human milk: preferred nutrition for preterm & LBW infants • Preterm milk: The best feeding option for the preterm infant – Rich in bioactive, immunomodulatory factors and highquality nutrients supporting immunity, intestinal microbiota development, neurodevelopment and feeding tolerance Benefits of providing human milk for premature infants Immunological benefits • Lower incidence of infections • Decreased risk of NEC • Decreased rates of sepsis • • Decreased rates of urinary tract infections Decreased risk of diarrhoea • =Decreased risk of NEC necrotizing enterocolitis. media Menon 2013, Arslanoglu 2013, Adamkin 2014, Bertino 2013. otitis Gastrointestinal (GI) benefits • Improved maturation of the GI tract • More rapid gastric emptying • Improved lactase activity • Improved digestion • Improved nutrient absorption

Human milk: preferred nutrition for preterm & LBW infants • Donor milk: The second best alternative when mother’s milk is not sufficiently available – Pasteurized (to reduce the risk of infection) term human milk – Limitations: • Pasteurization inactivates some of the nutritional elements of human milk (but preserves its beneficial effects) • Less rich in nutrients than preterm mother’s milk • Processing reduces fat and protein amounts Menon 2013, Arslanoglu 2013, Adamkin 2014, Bertino 2013.
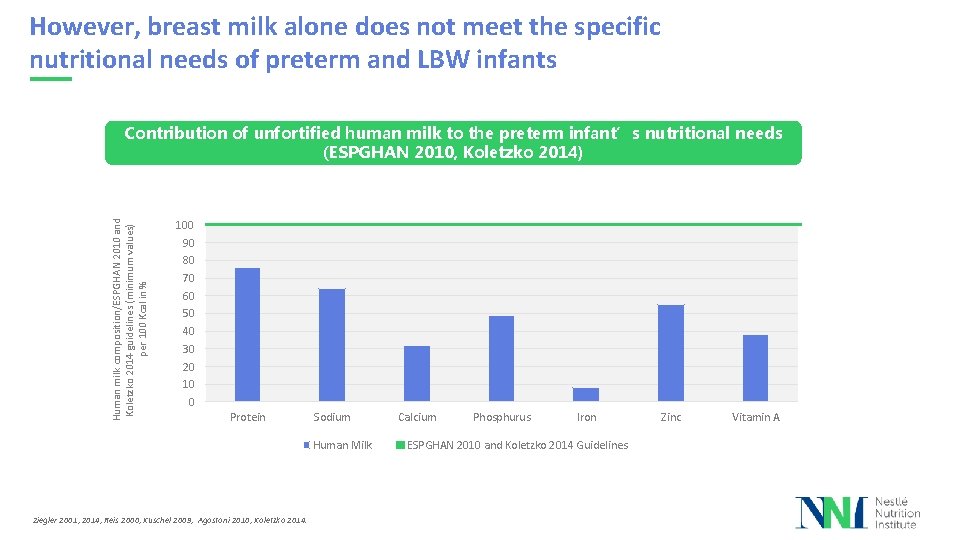
However, breast milk alone does not meet the specific nutritional needs of preterm and LBW infants Human milk composition/ESPGHAN 2010 and Koletzko 2014 guidelines (minimum values) per 100 Kcal in % Contribution of unfortified human milk to the preterm infant’s nutritional needs (ESPGHAN 2010, Koletzko 2014) 100 90 80 70 60 50 40 30 20 10 0 Protein Sodium Human Milk Ziegler 2001, 2014, Reis 2000, Kuschel 2009, Agostoni 2010, Koletzko 2014. Calcium Phosphurus Iron ESPGHAN 2010 and Koletzko 2014 Guidelines Zinc Vitamin A
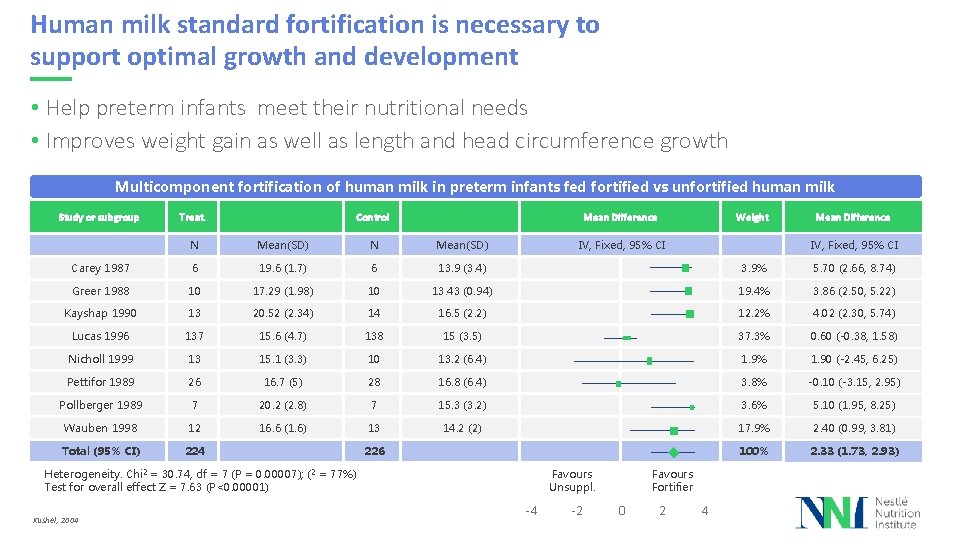
Human milk standard fortification is necessary to support optimal growth and development • Help preterm infants meet their nutritional needs • Improves weight gain as well as length and head circumference growth Multicomponent fortification of human milk in preterm infants fed fortified vs unfortified human milk Study or subgroup Treat. Control Mean Difference Weight IV, Fixed, 95% CI Mean Difference N Mean(SD) Carey 1987 6 19. 6 (1. 7) 6 13. 9 (3. 4) 3. 9% 5. 70 (2. 66, 8. 74) Greer 1988 10 17. 29 (1. 98) 10 13. 43 (0. 94) 19. 4% 3. 86 (2. 50, 5. 22) Kayshap 1990 13 20. 52 (2. 34) 14 16. 5 (2. 2) 12. 2% 4. 02 (2. 30, 5. 74) Lucas 1996 137 15. 6 (4. 7) 138 15 (3. 5) 37. 3% 0. 60 (-0. 38, 1. 58) Nicholl 1999 13 15. 1 (3. 3) 10 13. 2 (6. 4) 1. 9% 1. 90 (-2. 45, 6. 25) Pettifor 1989 26 16. 7 (5) 28 16. 8 (6. 4) 3. 8% -0. 10 (-3. 15, 2. 95) Pollberger 1989 7 20. 2 (2. 8) 7 15. 3 (3. 2) 3. 6% 5. 10 (1. 95, 8. 25) Wauben 1998 12 16. 6 (1. 6) 13 14. 2 (2) 17. 9% 2. 40 (0. 99, 3. 81) Total (95% CI) 224 100% 2. 33 (1. 73, 2. 93) 226 Heterogeneity. Chi 2 = 30. 74, df = 7 (P = 0. 00007); ( 2 = 77%) Test for overall effect Z = 7. 63 (P<0. 00001) Kushel, 2004 IV, Fixed, 95% CI Favours Unsuppl. -4 -2 Favours Fortifier 0 2 4
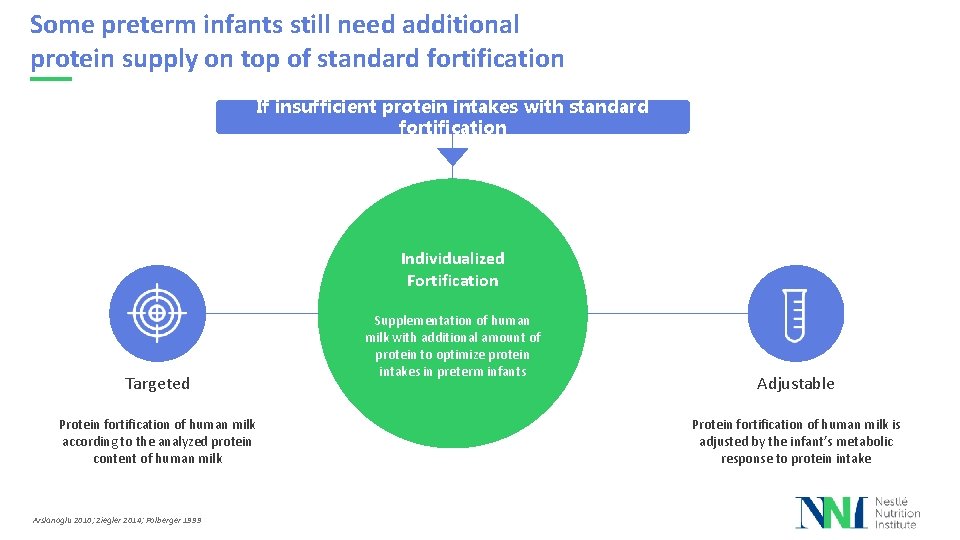
Some preterm infants still need additional protein supply on top of standard fortification If insufficient protein intakes with standard fortification Individualized Fortification Targeted Protein fortification of human milk according to the analyzed protein content of human milk Arslanoglu 2010; Ziegler 2014; Polberger 1999 Supplementation of human milk with additional amount of protein to optimize protein intakes in preterm infants Adjustable Protein fortification of human milk is adjusted by the infant’s metabolic response to protein intake

Summary of different methods of human milk fortification Standard fortification Individualized fortification Targeting fortification Adjustable fortification Fortification of human milk (HM) with HM fortifier according to a standard protocol Fortification of human milk according to the analysed protein content of HM Protein fortification of HM is adjusted by the infant’s metabolic response to protein intake Does not take into account variability of human milk nutrient content Taking into account the actual protein content of HM Taking into account the infant’s individual protein needs Does not require milk analysis Requires labour and cost intensive milk analysis Requires analysis of blood urea nitrogen (BUN), a marker of protein metabolism Simple method, most commonly used Depending on the availability of milk analysis BUN is measure on a routine basis in the NICU, thus this method is easily feasible Protein intake is not individualized, thus might be too low Results in improved protein intakes Adamkin, 2014; Radmaker, 2016; Arslanoglu, 2010
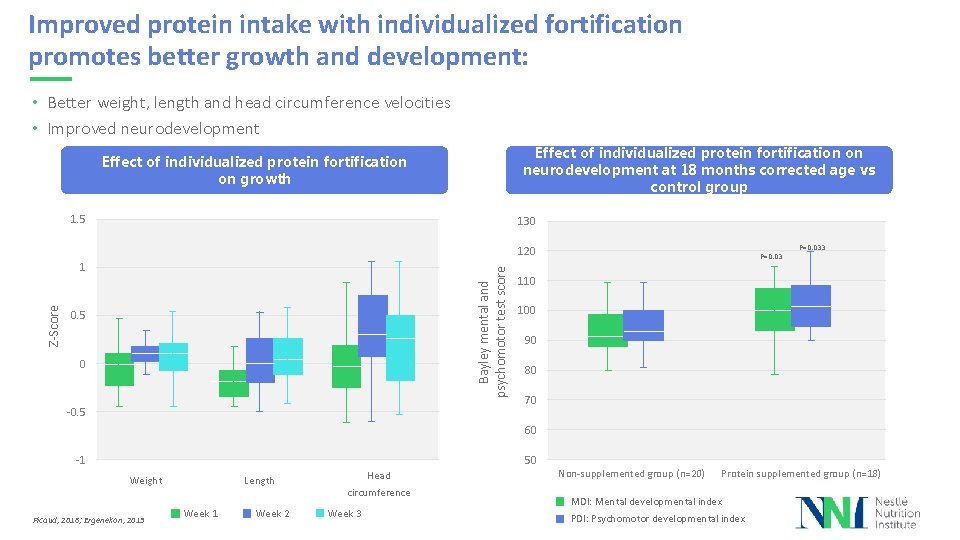
Improved protein intake with individualized fortification promotes better growth and development: • Better weight, length and head circumference velocities • Improved neurodevelopment Effect of individualized protein fortification on neurodevelopment at 18 months corrected age vs control group Effect of individualized protein fortification on growth 1. 5 130 120 Bayley mental and psychomotor test score Z-Score 1 0. 5 0 -0. 5 P=0. 033 110 100 90 80 70 60 50 -1 Weight Picaud, 2016; Ergenekon, 2013 Length Week 1 Week 2 Head circumference Week 3 Non-supplemented group (n=20) Protein supplemented group (n=18) MDI: Mental developmental index PDI: Psychomotor developmental index

Conclusions 1. Preterm birth is truly a global problem and preterm birth rates increase in almost all countries 2. Preterm and LBW infants have unique nutritional needs due to high growth rate, depleted nutrient stores & GI immaturity 3. Human Milk is the preferred nutrition for preterm and LBW infants 4. Human milk fortification is necessary to meet preterm infant’s nutritional needs and to support growth and development 5. In some cases individualized protein fortification additionally to standard fortification is required
- Slides: 29