NUTRIENTS Nutrients Macronutrients Protein Fat Carbohydrates Micronutrients Vitamins
NUTRIENTS

Nutrients • Macro-nutrients — Protein — Fat — Carbohydrates • Micro-nutrients — Vitamins — Minerals & trace-elements • Water

• macronutrients : required in relatively large amounts "big six": carbon , hydrogen , oxygen , nitrogen , phosphorous, sulfur other macronutrients: potassium , calcium , iron , magnesium • micronutrients : required in very small amounts, (but still necessary) boron (green plants) copper (some enzymes) molybdenum (nitrogen-fixing bacteria)

What are Macronutrients? Carbohydrates (CHO) Protein (PRO) Fat

Carbohydrates What are they? Two types of carbohydrates • Simple • Complex

Carbohydrates… • are made into simple sugars • simple sugars supply energy for brain activity.

Carbohydrates

Too Much/Too Little • When we eat too many carbohydrates our bodies turn them into fat & the fat becomes stored energy. • Low CHO diets stress the body causing it to break down muscle, fat and protein to make fuel for the brain.

Where do we get carbohydrates? Simple Carbohydrates: • • Hard Candy Pastries Table Sugar Honey

Sources of Carbohydrates • Complex Carbohydrates: – Grain Products • Breads • Rice • Pasta – Vegetables • Squash • Potatoes • Corn – – Dry Beans Peas Lentils Some Fruits • Banana • Plantain
Protein • Your body is made of protein. • Protein is made of amino acids, which are like building blocks. =

Amino Acids Your body arranges amino acids to build the different proteins it needs.

Protein Functions • Helps build muscles, blood, skin, hair, nails, and internal organs. • Helps the body grow & repair itself • Helps fight disease.

Too Much Protein. . . may mean too much fat. Over a long period, this can increase risk of – heart disease – diabetes – and some types of cancer.

Too Much Protein… may cause calcium loss. No calcium in the diet? =

Too Much Protein. . . • Means less carbohydrate intake to fuel muscles. • Your brain and eyes need a minimum of 100 grams of carbohydrates per day to work. • may overwork kidneys & lead to poor kidney function.

Protein Sources • Animal Sources: (Complete Proteins) – – – Meat Poultry Fish Eggs Dairy Products • Plant Sources: (Incomplete Proteins) – – Dry Beans Peas Nuts Tofu • Grain Products

Fats give food… flavor, texture and makes a person feel full.

Fat… • Promotes healthy skin and normal growth. • A part of cellular membranes.

Functions of Fat • • • Protects vital organs Provides kcal/energy to the body Aids in absorption of fat-soluble vitamins Recommend limiting 30% of kcal from fat Fat has 9 kcal per gram Provides a source of essential fatty acid
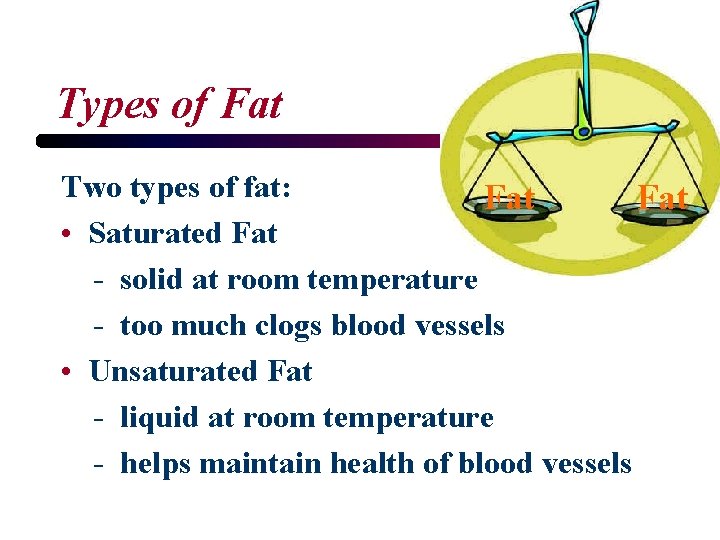
Types of Fat Two types of fat: Fat • Saturated Fat - solid at room temperature - too much clogs blood vessels • Unsaturated Fat - liquid at room temperature - helps maintain health of blood vessels

Too Much Fat. . . • Risk of… – Heart Disease – Diabetes – Hypertension (HTN) – Obesity – Various forms of cancer – Stroke

Fat 3, 500 calories equals one pound of body fat 3, 500 cal. = 1 lb.

Sources of Fat • Saturated Fats: – – – Butter Stick Margarine Meat fat Poultry fat Dairy Products • Unsaturated Fats: – – Vegetable oils Nuts Olives Avocados
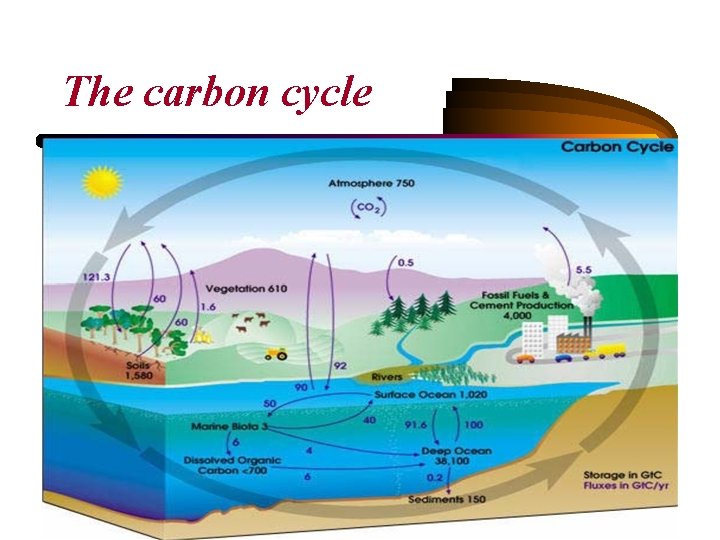
The carbon cycle
• Usually thought of as four major reservoirs of carbon (the atmosphere, the terrestrial biosphere - which includes freshwater systems and non-living organic material, such as soil carbon -, the oceans with dissolved inorganic carbon and living and non-living marine biota, and the sediments which includes fossil fuels) interconnected by pathways of exchange. • The exchanges between reservoirs, occur because of various chemical, physical, geological, and biological processes. The ocean contains the largest active pool of carbon near the surface of the Earth, but the deep ocean part of this pool does not rapidly exchange with the atmosphere.

• The global carbon budget is the balance of the exchanges (incomes and losses) of carbon between the carbon reservoirs or between one specific loop (e. g. , atmosphere biosphere) of the carbon cycle. IN THE OCEAN: • The seas contain around 36000 Gt of carbon, mostly in the form of bicarbonate ion. Inorganic carbon, that is carbon compounds with no carbon-carbon or carbonhydrogen bonds, is important in its reactions within water. This carbon exchange becomes important in controlling p. H in the ocean and can also vary as a source or sink for carbon.

• Carbon is readily exchanged between the atmosphere and ocean. In regions of oceanic upwelling, carbon is released to the atmosphere. Conversely, regions of downwelling transfer carbon (CO 2) from the atmosphere to the ocean. When CO 2 enters the ocean, carbonic acid is formed: CO 2 + H 2 O ⇌ H 2 CO 3 • This reaction achieves a chemical equilibrium. Another reaction important in controlling oceanic p. H levels is the release of hydrogen ions and bicarbonate. This reaction controls large changes in p. H: H 2 CO 3 ⇌ H+ + HCO 3−
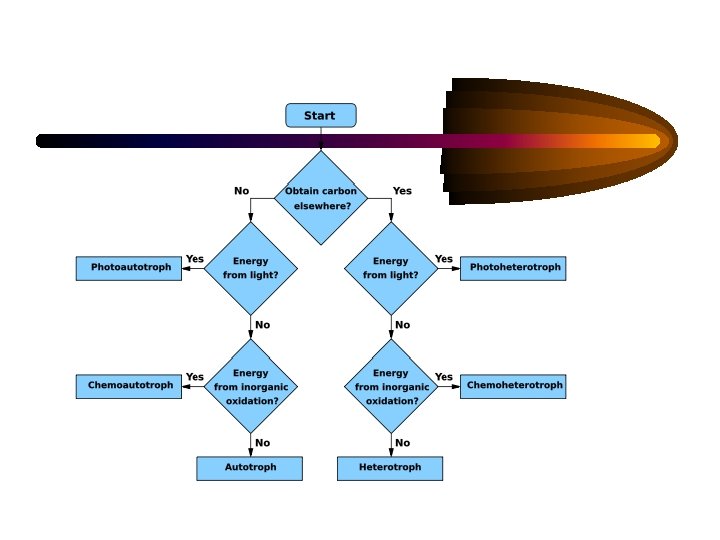

• • Carbon is major source of cellular C and energy. Microorganisms are classified in two categories on the basis of C source: i-) Heterotrophs use organic compounds such as carbohydrates, lipids, and hydrocarbons as a C and energy source. ii-) Autotrophs use carbon dioxide as a C source. Mixotrophs contaminantly grow under both autotrophic and heterotrophic conditions; however, autotrophic growth is stimulated by certain organic compounds. Facultative autotrophs grow under autotrophic conditions; however, they can grow under heterotrophic conditions in the absence of CO 2 and inorganic energy sources. Chemoautotrophs utilize CO 2 as a C source and obtain energy from the oxidation of inorganic compounds. Photoautotrophs use CO 2 as a C source and utilize light as an energy source.

Important Metabolic Terms Oxygen tolerance/usage: aerobic – requires or can use oxygen (O 2) anaerobic – does not require or cannot tolerate O 2 Energy usage: autotroph – uses CO 2 as a carbon source • photoautotroph – uses light as an energy source heterotroph – requires an organic carbon source • chemoheterotroph – gets energy & carbon from organic molecules • chemoautotroph – gets energy from inorganic mol.

. . . More important terms Facultative vs Obligate: facultative – “able to, but not requiring” e. g. : facultative anaerobes – can survive w/ or w/o O 2 obligate – “absolutely requires” e. g. : obligate anaerobes – cannot tolerate O 2 obligate intracellular parasite – can only survive within a host cell

Carbon sources • • • • The most common C sources in industrial fermentations Molasses (sucrose) Starch (glucose, dextrin) Corn syrup Waste sulfite liquor (glucose) In laboratory fermentations Glucose Sucrose Fructose Cheap C sources Methanol Ethanol Methane

• In aerobic fermentations, about 50% of substrate C is incorporated into cells and about 50% of it is used as en energy source. • In anaerobic fermentations, a large fraction of substrate C is converted to products and a smaller fraction is converted to cell mass (less than 30%).
The N cycle
The N cycle over land

Nitrogen • • • It constitutes about 10% to 14% of cell dry weight. The most widely used Nitrogen sources: NH 3 or ammonium salts [NH 4 Cl, (NH 4)2 SO 4, NH 4 NO 3] Proteins Peptides Amino acids Urea Yeast extract and peptone (organic nitrogen sources, expensive) Nitrogen is incorporated into cell mass in the form of proteins and nucleic acids. • Some microorganisms such as Azotobacter sp and cyanobacteria fix nitrogen from the atmosphere to form ammonium

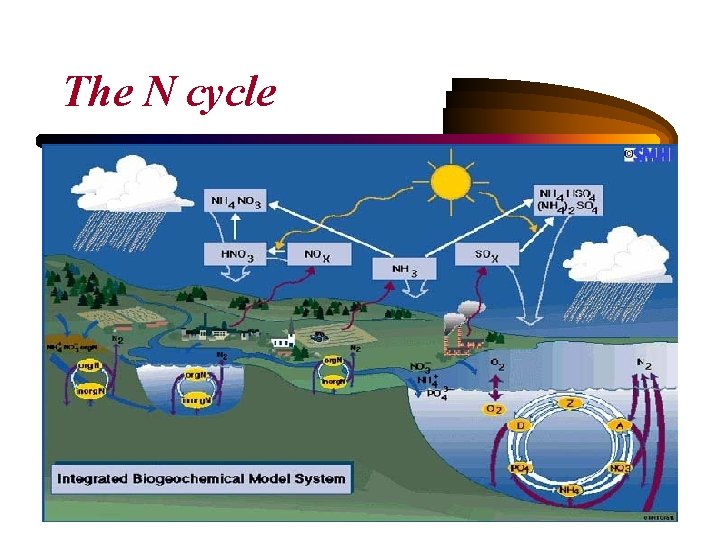
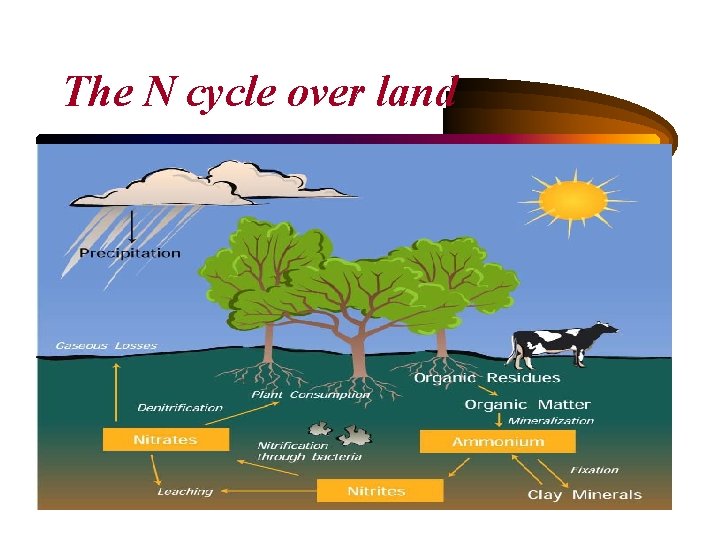
• Nitrogen is essential to all living systems: Eighty percent of Earth's atmosphere is made up of nitrogen in its gas phase. • Atmospheric nitrogen becomes part of living organisms in two ways: 1. through bacteria in the soil that form nitrates out of nitrogen in the air. 2. through lightning. During electrical storms, large amounts of nitrogen are oxidized and united with water to produce an acid that falls to Earth in rainfall and deposits nitrates in the soil. • Plants take up the nitrates and convert them to proteins that travel up the food chain through herbivores and carnivores.

• When organisms excrete waste, the nitrogen is released back into the environment. When they die and decompose, the nitrogen is broken down and converted to ammonia. • Nitrates may also be converted to gaseous nitrogen through a process called denitrification and returned to the atmosphere, continuing the cycle.

• Human impacts: 1. by artificial nitrogen fertilization (through the Haber Process, using energy from fossil fuels to convert N 2 to ammonia gas (NH 3) and planting of nitrogen fixing crops (Vitousek et al. , 1997). 2. transfer of nitrogen trace gases (N 2 O) to the atmosphere via agricultural fertilization, biomass burning, cattle and feedlots, and other industrial sources (Chapin et al. 2002). N 2 O in the stratosphere breaks down and acts as a catalyst in the destruction of atmospheric ozone. 3. NH 3 in the atmosphere has tripled as the result of human activities. It acts as an aerosol, decreasing air quality and clinging on to water droplets (acid rain).

4. Fossil fuel combustion has contributed to a 6 or 7 fold increase in NOx flux to the atmosphere. NO alters atmospheric chemistry, and is a precursor of tropospheric (lower atmosphere) ozone production, which contributes to smog, acid rain, and increases nitrogen inputs to ecosystems (Smil, 2000). 5. Ecosystem processes can increase with nitrogen fertilization, but anthropogenic input can also result in nitrogen saturation, which weakens productivity and can kill plants (Vitousek et al. , 1997) → algae blooms. 6. Decreases in biodiversity both over land in the ocean can result if higher nitrogen availability increases nitrogen-demanding species (Aerts and Berendse 1988).
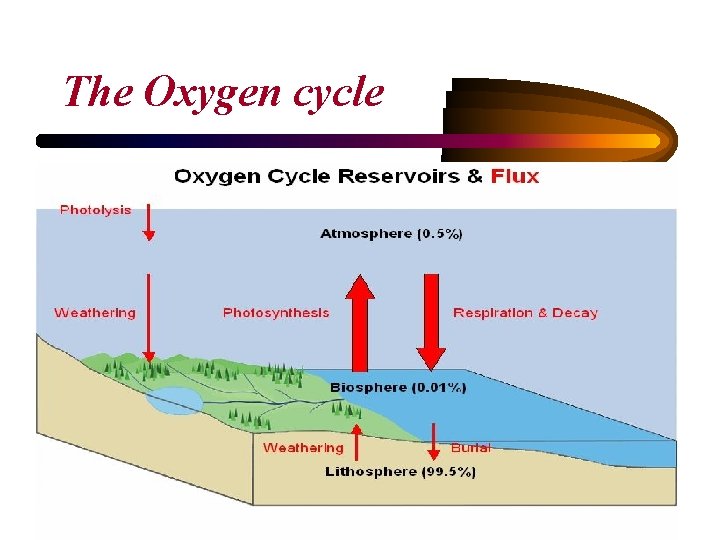
The Oxygen cycle

Oxygen • Oxygen is present all organic cell components and cellular water and constitutes 20% of dry weight of cells. • Molecular oxygen is required is a terminal electron acceptor in the aerobic metabolism of C compounds. • Gaseous oxygen is introduced into growth media by sparging air or by surface aeration.

• Plants use the energy of sunlight to convert carbon dioxide and water into carbohydrates and oxygen via photosynthesis. 106 CO 2 + 16 HNO 3 + H 3 PO 4 +78 H 2 O ↔ C 106 H 175 O 42 N 16 P + 150 O 2 • Photosynthesizing organisms include the plant life of the land areas as well as the phytoplankton of the oceans. • The tiny marine cyanobacteria Prochlorococcus was discovered in 1986 and accounts for more than half of the photosynthesis of the open ocean. • Animals form the other half of the oxygen cycle breathing in oxygen used to break carbohydrates down into energy in a process called respiration. O 2 + carbohydrates → CO 2 + H 2 O + energy

Hydrogen • Hydrogen constitutes 8% of cell dry weight and is derived from C campouns, such as carbohydrates. • Methanogens can utilize hydrogen as an energy source.
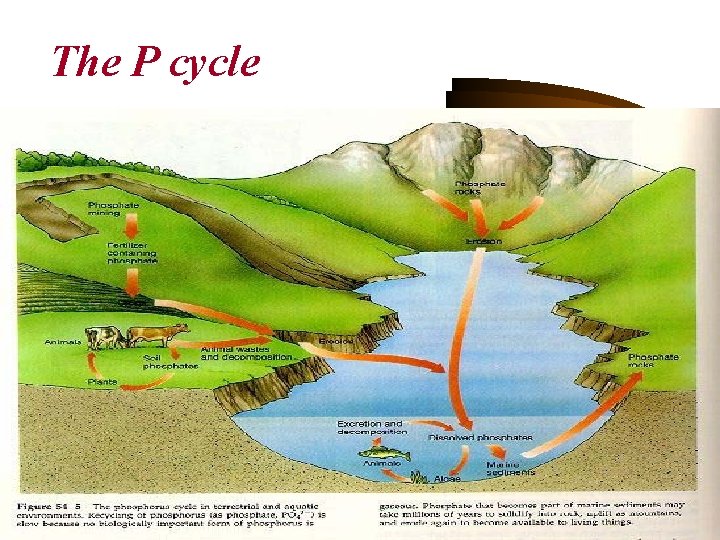
The P cycle

Phosphorus • Phosphorus constitutes 3% o dry cell weight and is present in nucleic acids and in the cell wall of some gram-positive bacteria such as teichoic acids. • The most common phosphate sources • Potassium dihydrogen phosphate (monopotassium phosphate or monobasic potassium phosphate) (KH 2 PO 4) • Dipotassium phosphate (K 2 HPO 4) (Inorganic phosphate salts) • Glycerophosphates (organic phosphate sources) • Phosphorus regulates cell metabolism. • The phosphate level in the media should be less than 1 m. M for the formation of many secondary metabolites such as antibiotics.

• The phosphorus cycle describes the movement of phosphorus through the lithosphere, hydrosphere, and biosphere. The atmosphere does not play a significant role, because phosphorus and phosphorus-based compounds are usually solids at the typical ranges of temperature and pressure found on Earth. • Phosphorus normally occurs in nature as part of a phosphate ion, consisting of a phosphorus atom and some number of oxygen atoms, the most abundant form (called orthophosphate) having four oxygens: PO 43 -.

• Most phosphates are found as salts in ocean sediments or in rocks. Over time, geologic processes can bring ocean sediments to land, and weathering will carry terrestrial phosphates back to the ocean. • Plants absorb phosphates from the soil and phosphate enters the food chain. After death, the animal or plant decays, and the phosphates are returned to the soil. Runoff may carry them back to the ocean or they may be reincorporated into rock. • Phosphates move quickly through plants and animals; however, the processes that move them through the soil or ocean are very slow, making the phosphorus cycle overall one of the slowest biogeochemical cycles.

• The primary biological importance of phosphates is as a component of nucleotides, which serve as energy storage within cells (ATP) or when linked together, form the nucleic acids DNA and RNA. Phosphorus is also found in bones, and in phospholipids (found in all biological membranes).

Sulfur • • • Sulfur constitutes 1% of cell dry weight and is present in proteins and some coenzymes The most common sulfur sources: Sulfate salts such as (NH 4)2 SO 4 Sulfur containing aminoacids. Certain autotrophs utilize S 2+ and S 0 as energy sources.

Potassium • K is a cofactor for some enzymes and is required in carbohydrate metabolism. • Cells tend to actively take up K+ and Mg 2+ and take out Na+ and Ca 2+. • The most coomonly used K salts: • Dipotassium phosphate (K 2 HPO 4), • Potassium dihydrogen phosphate (monopotassium phosphate or monobasic potassium phosphate) (KH 2 PO 4), • Potassium phosphate (K 3 PO 4).

Magnesium • Mg is a cofactor for some enzymes and is present in cell walls and membranes. • Ribosomes require Mg 2+ ions. • The most common Mg 2+ sources: • Magnesium sulfate heptahydrate (Mg. SO 4. 7 H 2 O) • Mg. Cl 2

Role(s) of essential mineral “macronutrient” elements in plants. Element Role(s) in plant (not all inclusive) N Constituent of amino acids, proteins, nucleic acids (DNA and RNA), nucleotides and coenzymes. P Component of sugar phosphates, nucleic acids (DNA and RNA), nucleotides, coenzymes, phospholipids, phytic acid, ATP, ADP, AMP. K Enzyme activator for over 40 enzymes, osmotic regulator, maintains electrical neutrality. Ca Constituent of the middle lamella of cell walls. Required to activate some enzymes involved in the hydrolysis of ATP and phospholipids. Mg Constituent of the chlorophyll molecule. Indirectly involved in phosphate transfer. S Component of S-containing amino acids cysteine and methionine and thus many proteins/enzymes.

Micronutrients (trace elements) • Zinc* • Iron • Chloride* • Copper • Molybdenum* • Manganese • Boron* • Cobalt • Nickel *documented responses in inland northwest dryland field crops

Zinc (Zn) • Required for many enzymes involved in photosynthesis and reproduction of genetic material (DNA) during cell division • Deficiency symptoms: stunted plants; little leaf syndrome, yellowing on younger leaves

Zinc (Zn) • Zinc is a cofactor for some enzymes • and also regulates some fermentations such as penicillin production.

Iron (Fe) • Central role in chlorophyll production, photosynthesis, energy transfer within plant • Deficiency symptoms: general yellowing or interveinal chlorosis (green veins, yellow between veins) on younger leaves

Iron (Fe) • • Fe is an important cofactor. Also plays a regulatory role in some fermentations. It is present in ferredoxin and cytochrome. Fe deficiency is required for the excretion of riboboflavin by Ashbya gosypii • Fe concentration regulates penicillin production by Penicillium chrysogenum

Manganese (Mn) • Important for energy transfer, photosynthesis reactions • Deficiency symptoms: greenish-grey spots or flecks on lower leaves; chlorosis

Manganese (Mn) • Mn is an enzyme cofactor, • Mn plays a role in the regulation of secondary metabolism and excretion of primary metabolites.

Trace elements needed under specific growth conditions: Copper (Cu) • Important for energy transfer, photosynthesis, resistance to certain diseases • Deficiency symptoms: decreased nodulation and N fixation by legumes, “white tip” disorder in cereals

Copper (Cu) • Cu is present in certain respiratory-chain components and enzymes, • Cu deficiency stimulates penicillin and citric acid production

Cobalt (Co) • Co is present in corrinoid compounds such as vitamin B 12. • Co is required by propionic bacteria and certain methanogens.

Molybdenum (Mo) • Important in the activation of enzymes involved in nitrogen fixation by legumes and nitrate reduction in non-legumes • Deficiency symptoms: general yellowing, small plants; similar to nitrogen deficiency (more important for legumes than cereals)

Molybdenum (Mo) • Mo is a cofactor of nitrate reductase and nitrogenase, • Mo is required for growth on NO 3 and N 2 as the sole source of N 2.

Calcium (Ca) • Ca is a cofactor for amylases and some proteases, • Ca is present in some bacterial spores and in cell walls of some cells, such as plant cells.

Sodium (Na) • Na is needed in trace amounts by some bacteria, • Methanogens are required Na for ion balance. • Na is important in the transport of charged species in eucaryotic cells.

Chlorine/chloride (Cl) • Roles in disease resistance, stem strength, water relations/drought tolerance • Deficiency symptoms: lodging, leaf spot syndrome (more important for cereals than legumes)

Chlorine/chloride (Cl) • Some halobacteria and marine microbes require Cl -, (Na+ too) • Nickel (Ni) • Ni is required by some methanogens as a cofactor, • Selenium (Se) • Se is required in formate metabolism of some organisms.

Trace elements that are rarely required B, Al, Si, Cr, V, Sn, Be, F, Ti, Ga, Ge, Br, Zr, W, Li, I Boron (B) • Important in sugar transport, cell wall properties that influence cell growth or expansion • Deficiency symptoms: stunted plants, dead or misshapen young leaves, red coloration, sterile flowers, bud and/or fruit drop (more important for legumes than cereals)

Role(s) of essential mineral “micronutrient” elements in plants. Element Role(s) in plant (not all inclusive) Fe Component of cytochromes and non-heme iron proteins involved in photosynthesis, N 2 fixation and respiration. Mn Required for the photosynthetic evolution of O 2 (splitting of H 2 O). Required to activate many dehydrogenases, decarboxylases, kinases, oxidases, and peroxidases. Cu Essential component of ascorbic oxidase, tyrosinase, monoamine oxidase, uricase, and cytochrome oxidase. Zn Essential constituent of alcohol dehydrogenase, glutamic dehydrogenase, carbonic anhydrase, and other enzymes. B Forms complexes with some CHO’s* and there is indirect evidence for involvement in CHO transport. Mo A constituent of nitrate reductase. Essential for N 2 fixation. Ni Essential for the function of urease and N nutrition in general. Cl Required for the photosynthetic reactions involved in O 2 evolution.

Growth medium A growth medium or culture medium is a liquid or gel designed to support the growth of microorganisms or cells, or small plants like the moss Physcomitrella patens. There are different types of media for growing different types of cells. An Agar Plate -- an example of a bacterial growth medium. Specifically, it is a streak plate; the orange lines and dots are formed by bacterial colonies.

Growth medium • Defined media contain specific amounts of pure chemical compounds with known chemical compositions. • A medium containing glucose, (NH 4)2 SO 4, KH 2 PO 4 and Mg. Cl 2 is a defined medium. • Complex media contain natural compounds whose chemical composition is not exactly known. • A medium containing yeast extracts, peptone, molasses, or corn steep liquor, is a complex medium. • A complex medium can provide the necessary growth factors, vitamins, hormones, and trace elements

Comparıson of defined and complex media Defined media Complex media • Advantages • The results are more reproducible • Better control of the fermentation • Recovery and purification of a product is often easier and cheaper • Advantages • Higher cell yields, • Less expensive

Questions

References ü Michael L. Shuler and Fikret Kargı, Bioprocess Engineering: Basic Concepts (2 nd Edition), Prentice Hall, New York, 2002. ü 1. James E. Bailey and David F. Ollis, Biochemical Engineering Fundementals (2 nd Edition), Mc. Graw-Hill, New York, 1986.

• www. oluakinkugbechildnutritioncentre. com/prese ntations/nutrients. pdf • http: //plone. rockyview. ab. ca/cochrane/teacherwebpages/mcleod/biology-20/bio-20 -digestionnutrition/macro. ppt#263, 9, Polysaccharides • http: //www. foodafactoflife. org. uk/attachments/a 8 e 6 ca 04 -451 f-49843320 f 03 e. ppt#362, 22, Over consumption of fat

• http: //www. schoolnutritionandfitness. com/data/pd f/Teacher. Power. Points/Macronutrients. ppt • http: //www. mckinley. uiuc. edu/handouts/pdfs/macr onutrients. pdf • http: //extension. oregonstate. edu/umatilla/mf/sites/ default/files/Role_of_Micronutrients_2008. pdf
- Slides: 79