Nutrient Removal Objective To understand the fundamental principles

Nutrient Removal Objective: • To understand the fundamental principles of nutrient removal using chemical and biological methods • To know examples of the major wastewater treatment processes for nutrient removal. –Reference: “Operation of municipal wastewater treatment plants. Manual of Practice 11, Vol 2 (1996). Water Environment Federation “ –http: //www. staff. ncl. ac. uk/p. j. sallis/teach. html • see section ‘CIV 912’; user and password both cassie

Nutrient Removal Introduction Chemical Methods Principle of Biological Nitrogen Removal Processes Principle of Biological Phosphorus Removal Processes Combined Biological N & P Removal Processes
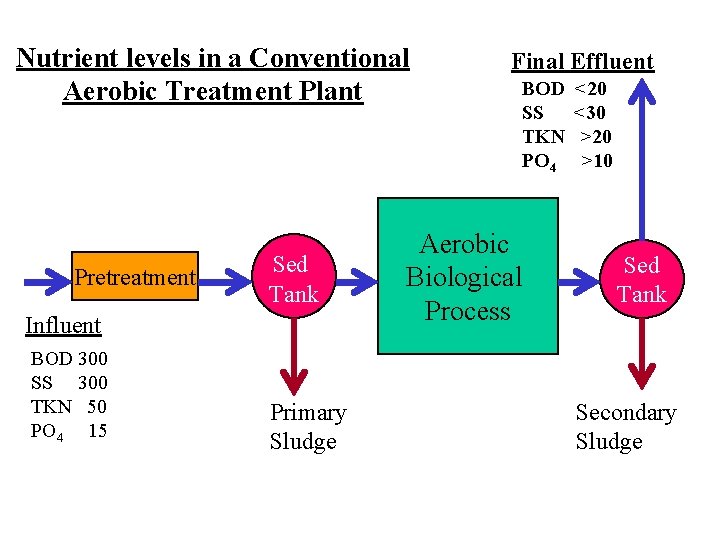
Nutrient levels in a Conventional Aerobic Treatment Plant Pretreatment Sed Tank Influent BOD 300 SS 300 TKN 50 PO 4 15 Primary Sludge Final Effluent BOD SS TKN PO 4 Aerobic Biological Process <20 <30 >20 >10 Sed Tank Secondary Sludge

Nutrient Cycles • Eutrophication potential – Nutrient balance C: N: P (100: 5: 1) 10, 000 pe x 200 l/d x 15 mg. N/l algae/d 10, 000 pe x 200 l/d x 5 mg. P/l algae/d 500 kg 1200 kg

Nutrient Removal - Standards UWWT Directive (1991): Pop >10, 000 N<15 mg/l P<2 mg/l Pop >100, 000 N<10 mg/l P<1 mg/l or 80% removal of Total P 70 - 80% removal of Total N (The above applies to “sensitive waters”)
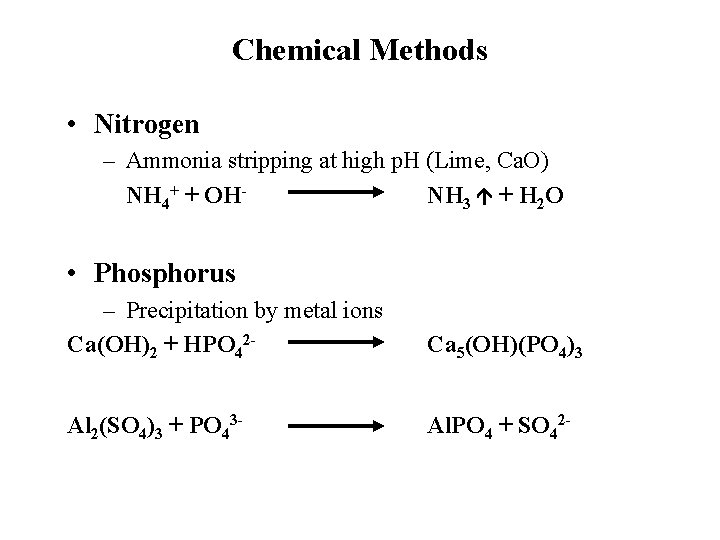
Chemical Methods • Nitrogen – Ammonia stripping at high p. H (Lime, Ca. O) NH 4+ + OHNH 3 + H 2 O • Phosphorus – Precipitation by metal ions Ca(OH)2 + HPO 42 - Ca 5(OH)(PO 4)3 Al 2(SO 4)3 + PO 43 - Al. PO 4 + SO 42 -

Biological Nutrient Removal • Assimilation – C, N, P, S etc uptake for synthesis of new cells • Dissimilation – C, N, S, oxidized/reduced to provide energy • Aerobic (oxic) – in the presence of molecular oxygen (O 2) • Anoxic – very low concentration of molecular oxygen (O 2) – significant levels of electron acceptors (NO 3 -, SO 4 -) • Anaerobic – no oxygen, lack of electron acceptors (only CO 2)

Biological Nitrogen Removal • Wastewaters contain: Org-N, ammonia, (nitrate) • Dissimilatory metabolism • Nitrification 1. 2. NH 4+ + 1. 5 O 2 NO 2 - + 0. 5 O 2 Nitrosomonas Nitrobacter NO 2 - + 2 H+ + H 2 O NO 3 - (nitrified effluent) • Denitrification NO 3 - + CH 2 + H+ N 2 + CO 2 + H 2 O denitrifying bacteria (many)
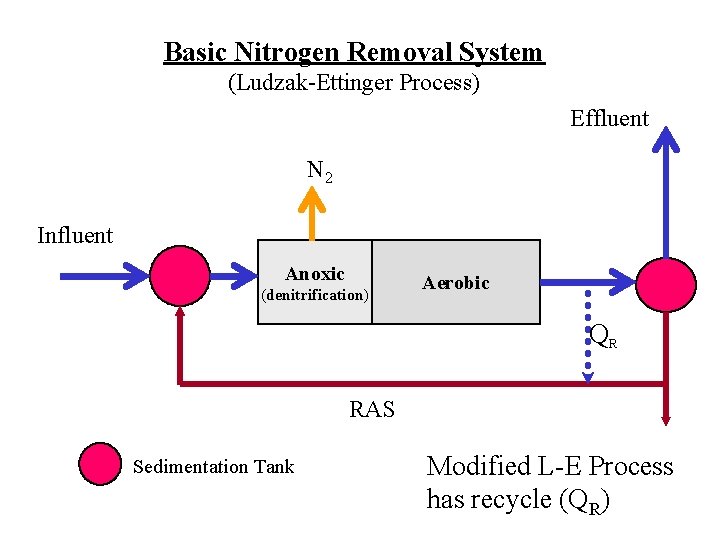
Basic Nitrogen Removal System (Ludzak-Ettinger Process) Effluent N 2 Influent Anoxic (denitrification) Aerobic QR RAS Sedimentation Tank Modified L-E Process has recycle (QR)
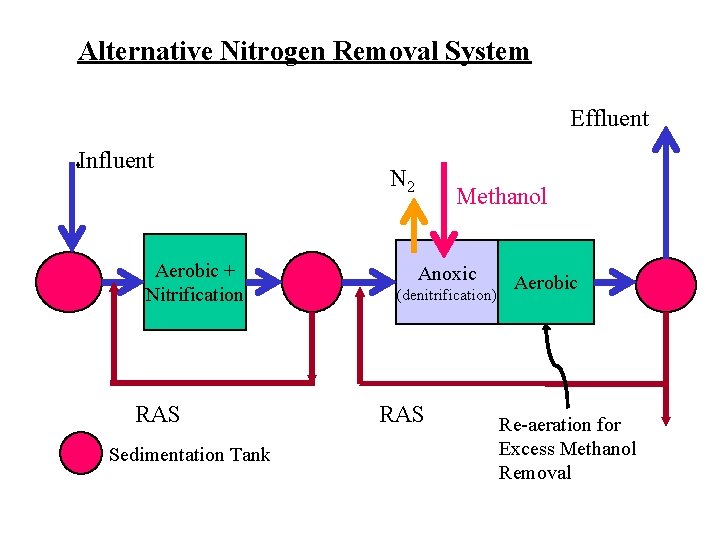
Alternative Nitrogen Removal System Effluent Influent Aerobic + Nitrification RAS Sedimentation Tank N 2 Methanol Anoxic (denitrification) RAS Aerobic Re-aeration for Excess Methanol Removal

Biological Phosphorus Removal • Selection of Bacteria in Sludge – Luxury uptake of Phosphorus • (Acinetobacter, Pseudomonas) – Cyclic Environmental Conditions • High BOD when anaerobic • Low BOD when aerobic • Sidestream – P is stripped from sludge in separate unit process • Mainstream – P is concentrated to high levels in the sludge (biomass)
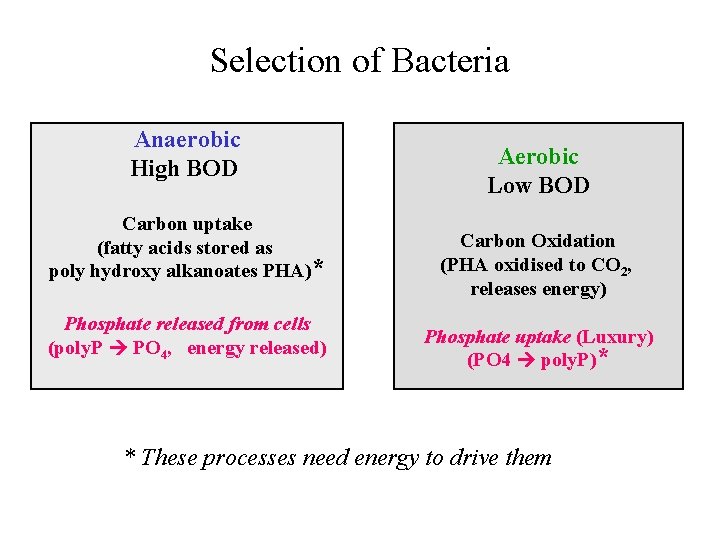
Selection of Bacteria Anaerobic High BOD Carbon uptake (fatty acids stored as poly hydroxy alkanoates PHA)* Phosphate released from cells (poly. P PO 4, energy released) Aerobic Low BOD Carbon Oxidation (PHA oxidised to CO 2, releases energy) Phosphate uptake (Luxury) (PO 4 poly. P)* * These processes need energy to drive them
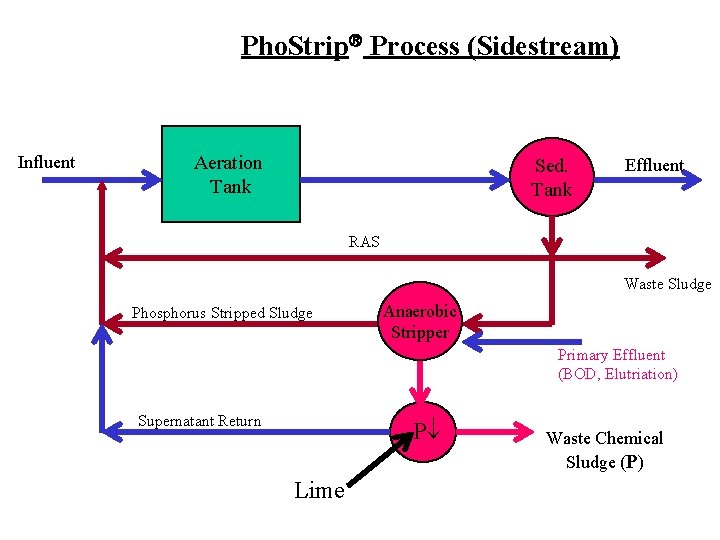
Pho. Strip Process (Sidestream) Influent Aeration Tank Sed. Tank Effluent RAS Waste Sludge Phosphorus Stripped Sludge Anaerobic Stripper Primary Effluent (BOD, Elutriation) Supernatant Return P Lime Waste Chemical Sludge (P)
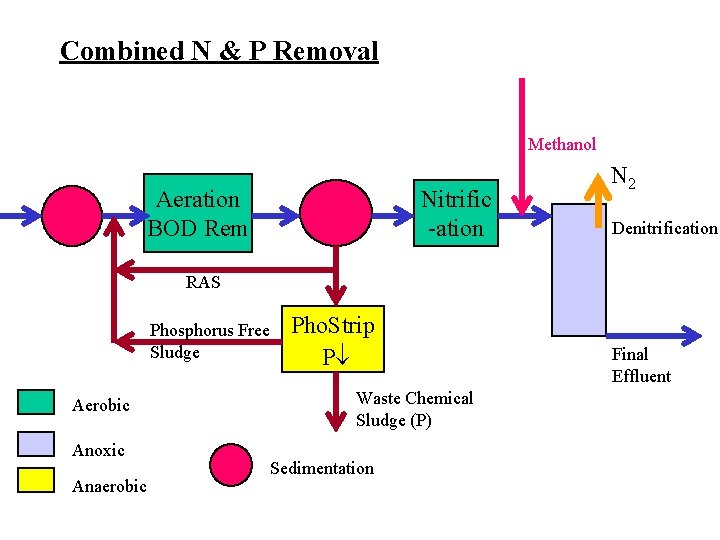
Combined N & P Removal Methanol Nitrific -ation Aeration BOD Rem N 2 Denitrification RAS Phosphorus Free Sludge Aerobic Anoxic Anaerobic Pho. Strip P Waste Chemical Sludge (P) Sedimentation Final Effluent
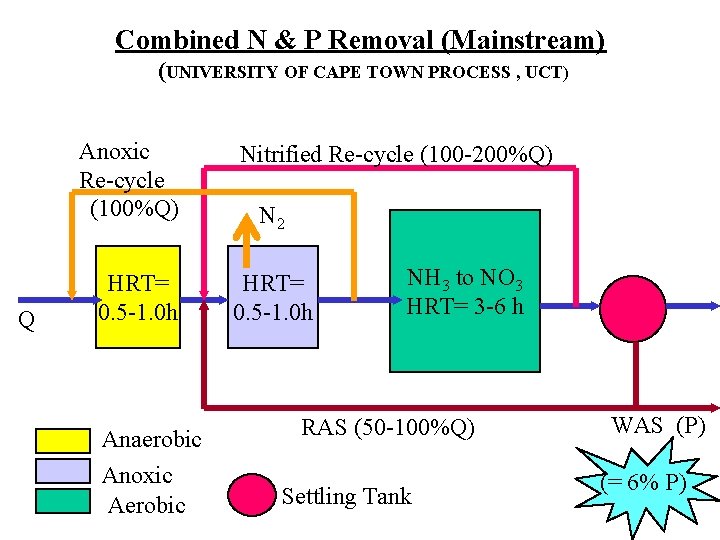
Combined N & P Removal (Mainstream) (UNIVERSITY OF CAPE TOWN PROCESS , UCT) Anoxic Re-cycle (100%Q) Q HRT= 0. 5 -1. 0 h Anaerobic Anoxic Aerobic Nitrified Re-cycle (100 -200%Q) N 2 HRT= 0. 5 -1. 0 h NH 3 to NO 3 HRT= 3 -6 h RAS (50 -100%Q) Settling Tank WAS (P) (= 6% P)

Operational Considerations • Maintain discrete environments – excess recycle rate gives completely mixed system • Limitations – Combined System optimized for N (denitrification), biological P removal non-optimized (requires chemical supplementation) • Efficiency – denitrification re-uses Oxygen bound in the nitrate • Contingency – provide P removal by chemical means (when biological process fails)
- Slides: 16