NUTRIENT CYCLES READINGS FREEMAN Chapter 54 NUTRIENT CYCLES




























- Slides: 38

NUTRIENT CYCLES READINGS: FREEMAN Chapter 54

NUTRIENT CYCLES: ECOSYSTEM TO ECOSPHERE • Nutrient cycling occurs at the local level through the action of the biota. • Nutrient cycling occurs at the global level through geological processes, such as, atmospheric circulation, erosion and weathering.

NUTRIENT CYCLES • The atoms of earth and life are the same; they just find themselves in different places at different times. • Most of the calcium in your bones came from cows, who got it from corn, which took it from rocks that were once formed in the sea. • The path atoms take from the living (biotic) to the non-living (abiotic) world and back again is called a biogeochemical cycle.

Nutrients: The Elements of Life • Of the 50 to 70 atoms (elements) that are found in living things, only 15 or so account for the major portion of living biomass. • Only around half of these 15 have been studied extensively as they travel through ecosystems or circulate on a global scale.

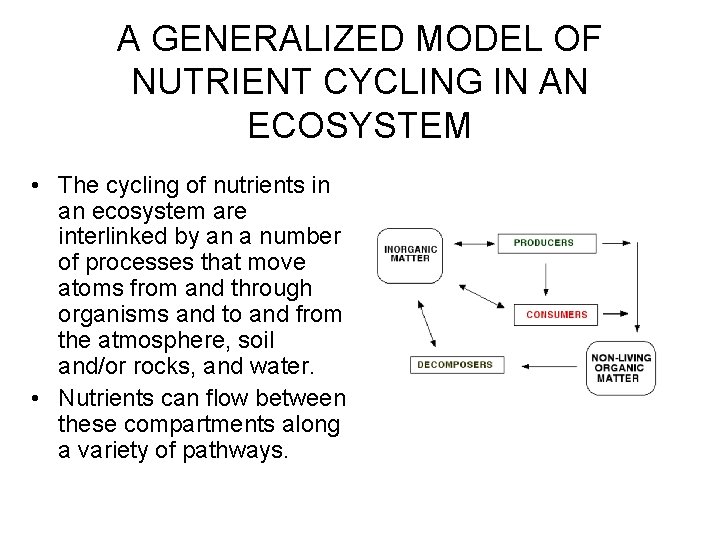
A GENERALIZED MODEL OF NUTRIENT CYCLING IN AN ECOSYSTEM • The cycling of nutrients in an ecosystem are interlinked by an a number of processes that move atoms from and through organisms and to and from the atmosphere, soil and/or rocks, and water. • Nutrients can flow between these compartments along a variety of pathways.

Nutrient Compartments in a Terrestrial Ecosystem • The organic compartment consists of the living organisms and their detritus. • The available-nutrient compartment consists of nutrients held to surface of soil particles or in solution. • The third compartment consists of nutrients held in soils or rocks that are unavailable to living organisms. • The fourth compartment is the air which can be found in the atmosphere or in the ground.

Uptake of Inorganic Nutrients from the Soil • With the exception of CO 2 and O 2 which enter though leaves, the main path of all other nutrients is from the soil through the roots of producers. • Even consumers which find Ca, P, S and other elements in the water they drink, obtain the majority of these nutrients either directly or indirectly from producers.

The Atmosphere Is a Source of Inorganic Nutrients • The atmosphere acts as a reservoir for carbon dioxide (CO 2), oxygen (O 2) and water (H 2 O). • These inorganic compounds can be exchanged directly with the biota through the processes of photosynthesis and respiration. • The most abundant gas in the atmosphere is nitrogen (N 2); about 80% by volume. Its entry into and exit from the biota is through bacteria.

Some Processes By Which Nutrients Are Recycled • Cycling within an ecosystem involves a number of processes. • These are best considered by focusing attention on specific nutrients.

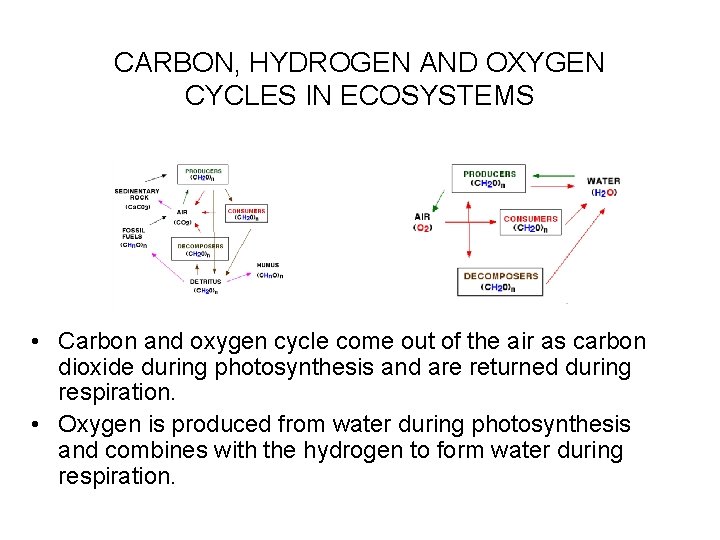
CARBON, HYDROGEN AND OXYGEN CYCLES IN ECOSYSTEMS • C, H & O basic elements of life; making up from about 98% of plant biomass. • CO 2 and O 2 enter biota from the atmosphere. • Producers convert CO 2 and H 2 O into carbohydrates (CH 2 O compounds) and release O 2 from water. • Producers, consumers and decomposers convert CH 2 O compounds, using O 2, back into CO 2 and H 2 O.

CARBON, HYDROGEN AND OXYGEN CYCLES IN ECOSYSTEMS • Carbon and oxygen cycle come out of the air as carbon dioxide during photosynthesis and are returned during respiration. • Oxygen is produced from water during photosynthesis and combines with the hydrogen to form water during respiration.

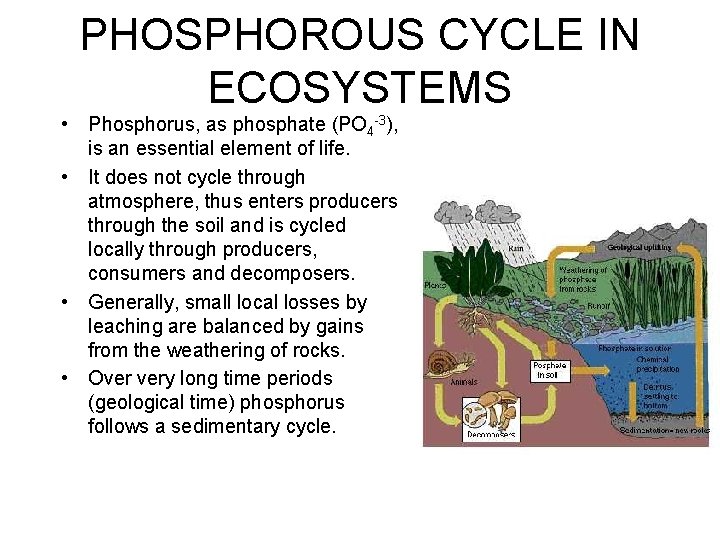
PHOSPHOROUS CYCLE IN ECOSYSTEMS • Phosphorus, as phosphate (PO 4 -3), is an essential element of life. • It does not cycle through atmosphere, thus enters producers through the soil and is cycled locally through producers, consumers and decomposers. • Generally, small local losses by leaching are balanced by gains from the weathering of rocks. • Over very long time periods (geological time) phosphorus follows a sedimentary cycle.

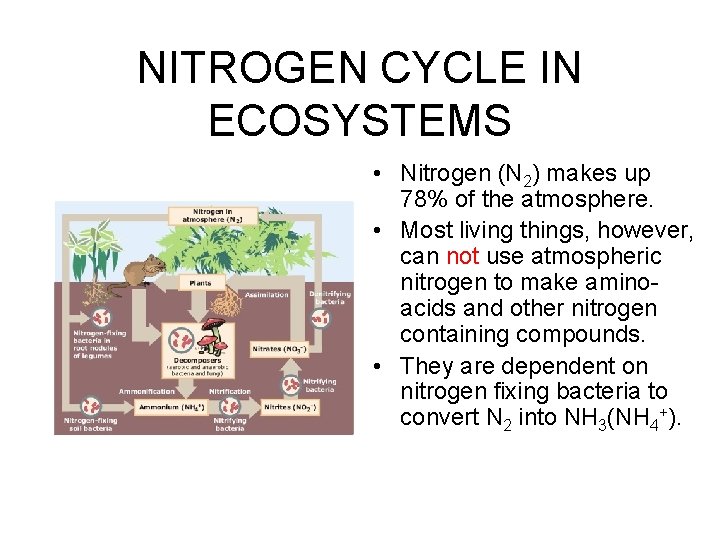
NITROGEN CYCLE IN ECOSYSTEMS • Nitrogen (N 2) makes up 78% of the atmosphere. • Most living things, however, can not use atmospheric nitrogen to make aminoacids and other nitrogen containing compounds. • They are dependent on nitrogen fixing bacteria to convert N 2 into NH 3(NH 4+).

Sources of Nitrogen to the Soil • Natural ecosystems receive their soil nitrogen through biological fixation and atmospheric deposition. • Agricultural ecosystems receive additional nitrogen through fertilizer addition.

Biological Sources of Soil Nitrogen • Only a few species of bacteria and cyanobacteria are capable of nitrogen fixation. • Some are fee-living and others form mutualistic associations with plants. • A few are lichens.

Atmospheric Sources of Soil Nitrogen • Lightning was the major source of soil nitrogen until recent times when the burning of fossil fuels became a major source of atmospheric deposition. • Nitrogen oxides come from a variety of combustion sources that use fossil fuels. In urban areas, at least half of these pollutants come cars and other vehicles.

Agricultural Supplements to Soil Nitrogen • Various forms of commercial fertilizer are added to agricultural fields to supplement the nitrogen lost through plant harvest. • Crop rotation with legumes such as soybeans or alfalfa is also practiced to supplement soil nitrogen.

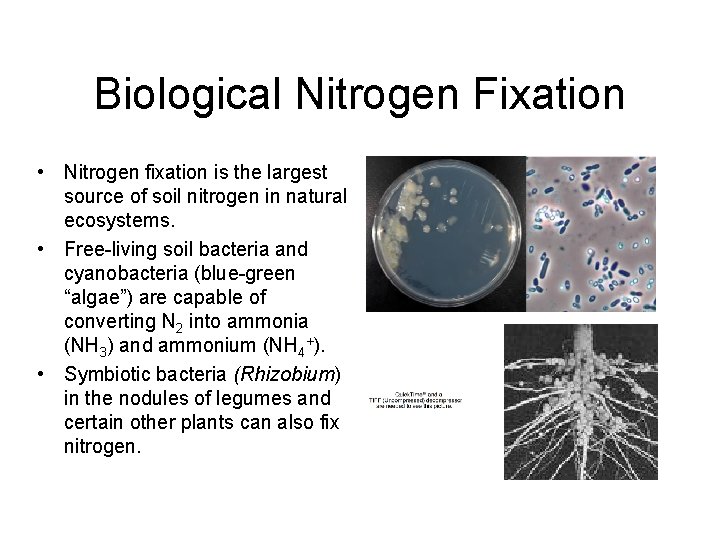
Biological Nitrogen Fixation • Nitrogen fixation is the largest source of soil nitrogen in natural ecosystems. • Free-living soil bacteria and cyanobacteria (blue-green “algae”) are capable of converting N 2 into ammonia (NH 3) and ammonium (NH 4+). • Symbiotic bacteria (Rhizobium) in the nodules of legumes and certain other plants can also fix nitrogen.

Nitrification • Several species of bacteria can convert ammonium (NH 4+) into nitrites (NO 2 -). • Other bacterial species convert nitrites (NO 2 -) to nitrates (NO 3 -).

Uptake of Nitrogen by Plants • Plants can take in either ammonium (NH 4+) or nitrates (NO 3 -) and make amino acids or nucleic acids. • These molecules are the building blocks of proteins and DNA, RNA, ATP, NADP, respectively. • These building blocks of life are passed on to other trophic levels through consumption and decomposition.

Ammonification • Decomposers convert organic nitrogen (CHON) into ammonia (NH 3) and ammonium (NH 4+). • A large number of species of bacteria and fungi are capable of converting organic molecules into ammonia.

Denitrification • A broad range of bacterial species can convert nitrites, nitrates and nitrous oxides into molecular nitrogen (N 2). • They do this under anaerobic conditions as a means of obtaining oxygen (O 2). • Thus, the recycling of N is complete.

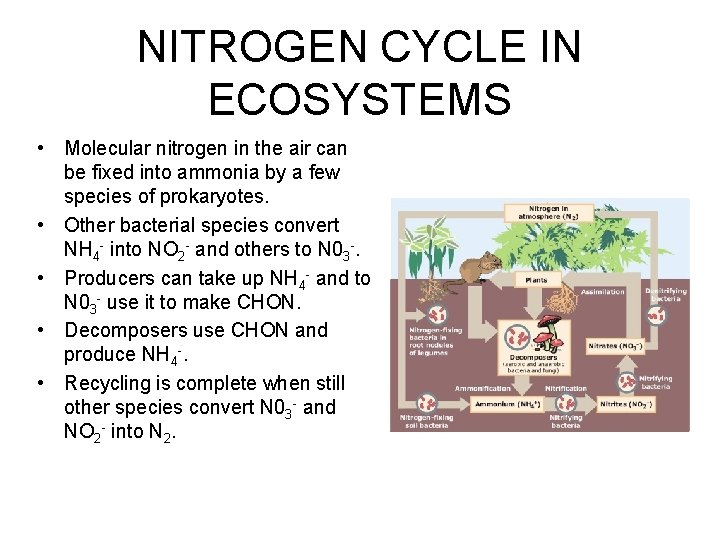
NITROGEN CYCLE IN ECOSYSTEMS • Molecular nitrogen in the air can be fixed into ammonia by a few species of prokaryotes. • Other bacterial species convert NH 4 - into NO 2 - and others to N 03 -. • Producers can take up NH 4 - and to N 03 - use it to make CHON. • Decomposers use CHON and produce NH 4 -. • Recycling is complete when still other species convert N 03 - and NO 2 - into N 2.

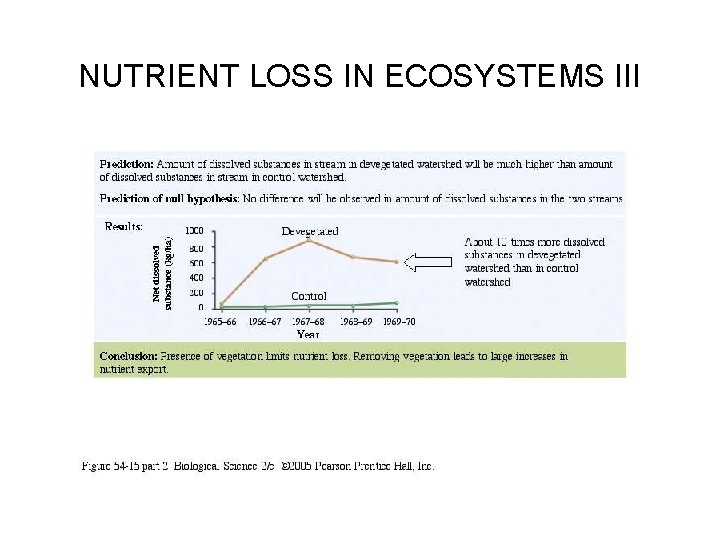
NUTRIENT LOSS IN ECOSYSTEMS I • The role of vegetation in nutrient cycles is clearly seen in clear cut experiments at Hubbard Brook. • When all vegetation was cut from a 38 -acre watershed, the output of water and loss of nutrients increased; 60 fold for nitrates, and at least 10 fold for other nutrients. • Freeman describes the experiments on page 1254 and in Figure 54. 15.

NUTRIENT LOSS IN ECOSYSTEMS II

NUTRIENT LOSS IN ECOSYSTEMS III

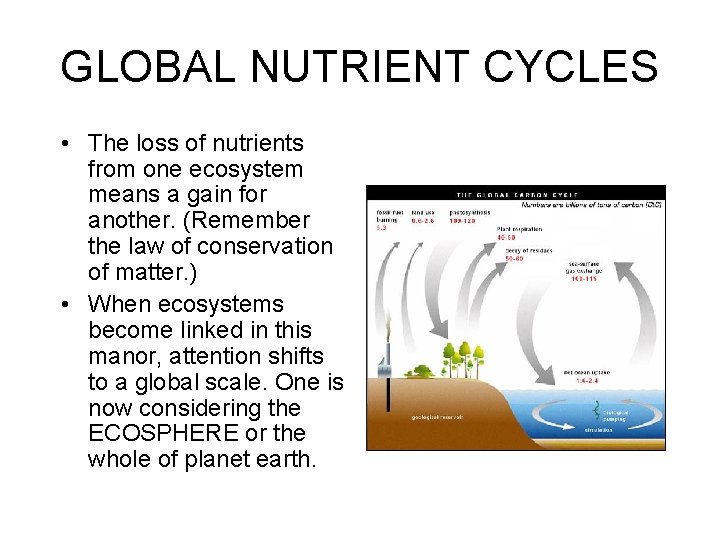
GLOBAL NUTRIENT CYCLES • The loss of nutrients from one ecosystem means a gain for another. (Remember the law of conservation of matter. ) • When ecosystems become linked in this manor, attention shifts to a global scale. One is now considering the ECOSPHERE or the whole of planet earth.

GLOBAL WATER CYCLE I • Water is the solvent in which all the chemistry of life takes place and the source of its hydrogen. • The earth’s oceans, ice caps, glaciers, lakes, rivers, soils and atmosphere contains about 1. 5 billion cubic kilometers of H 2 O. • It has been estimated that all the earth’s water is split by plant cells and reconstituted by the biota about every 2, 000 years.

GLOBAL WATER CYCLE II • Oceans contain a little less than 98% of the earth’s water. • Around 1. 8% is ice; found in the two polar ice caps and mountain glaciers. • Only 0. 5% is found in the water table and ground water. • The atmosphere contains only 0. 001% of the earth’s water, but is the major driver of weather.

GLOBAL WATER CYCLE III • The rate at which water cycles is shown in Figure 54. 16 (Freeman, 2005). • Evaporation exceeds precipitation over the oceans; thus there is a net movement of water to the land. • Nearly 60% of the precipitation that falls on land is either evaporated or transpired by plants; the remainder is runoff and ground water.

GLOBAL WATER CYCLE IV

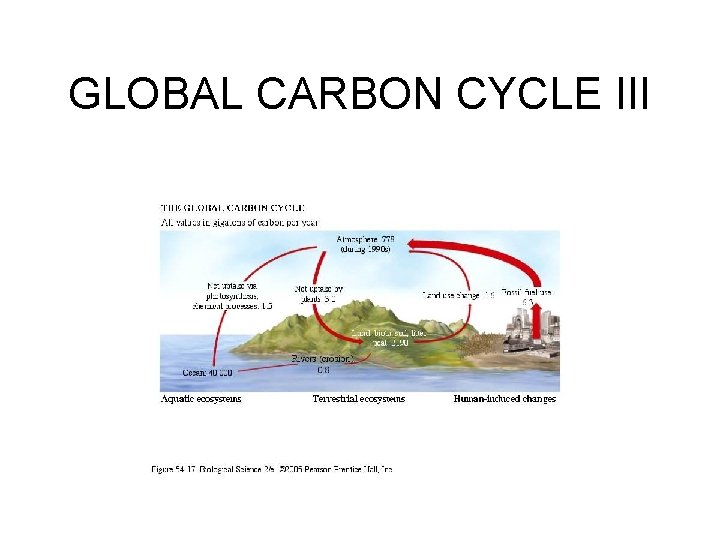
GLOBAL CARBON CYCLE I • All but a small portion of the earth’s carbon (C) is tied up in sedimentary rocks; but the portion that circulates is what sustains life. • The active pool of carbon is estimated to be around 40, 000 gigatons. • 93. 2 % found in the ocean; 3. 7% in soils; 1. 7% in atmosphere; 1. 4% in vegetation.

GLOBAL CARBON CYCLE II • The rate at which the biota exchanges CO 2 with atmosphere has been estimated to be every 300 years. • The rate at which carbon cycles through various components of the ecosphere is summarized in Figure 54. 17 in Freeman (2005). • Since the industrial revolution, a new source of stored sedimentary carbon has been added to the atmosphere from the burning of fossil fuels causing a concern with respect to climate change.

GLOBAL CARBON CYCLE III

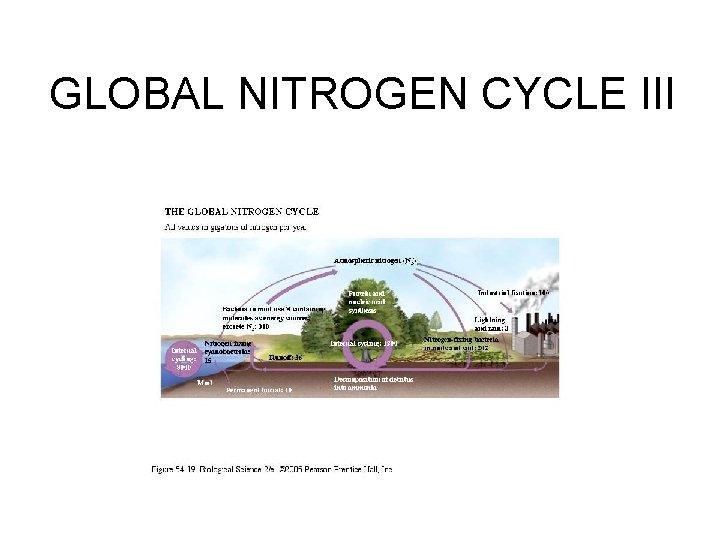
GLOBAL NITROGEN CYCLE I • 99. 4% of exchangeable N is found in the atmosphere; 0. 5% is dissolved in the ocean; 0. 04% in detritus ; 0. 006% as inorganic N sources; 0. 0004% in living biota. • Figure 54. 19 in Freeman (2005) gives major pathways and rates of exchange.

GLOBAL NITROGEN CYCLE II • Humans are adding large amounts of N to ecosystems. Some estimates of are given in Figure 54. 20 in Freeman (2005). • Among the fossil fuel sources, power plants and automobiles are important sources of atmospheric nitrogen deposition in the US. • Investigations of native plant and natural ecosystem responses to nitrogen deposition and global warming will be a focus of study.

GLOBAL NITROGEN CYCLE III

NUTRIENT CYCLES READINGS: FREEMAN Chapter 54