NUTRIEN NITROGEN Dep Manajemen Sumberdaya Perairan Fakultas Perikanan

NUTRIEN: NITROGEN Dep. Manajemen Sumberdaya Perairan Fakultas Perikanan dan Ilmu Kelautan - IPB

ESSENTIAL NUTRIENT Tucker, MR. 1999. Essential Plant Nutrients: their presence in North Carolina soils and role


C Ø biomass-limiting nutrients: membatasi produksi biomass Ø rate-limiting nutrients: membatasi laju produktivitas primer

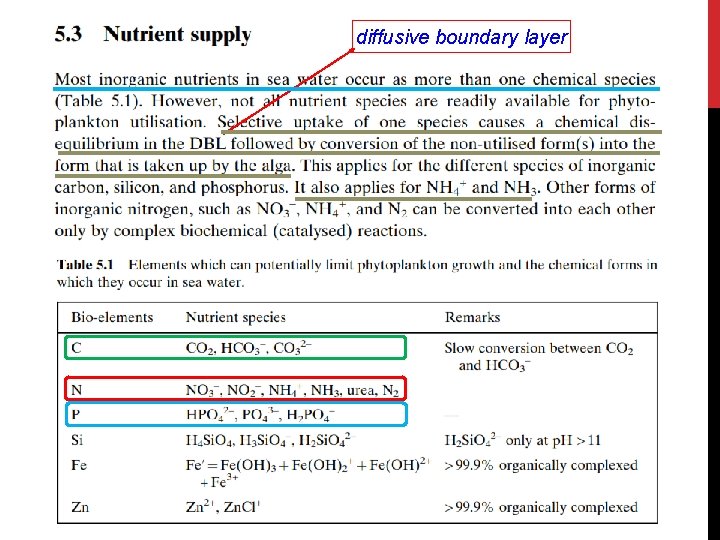
diffusive boundary layer


urea
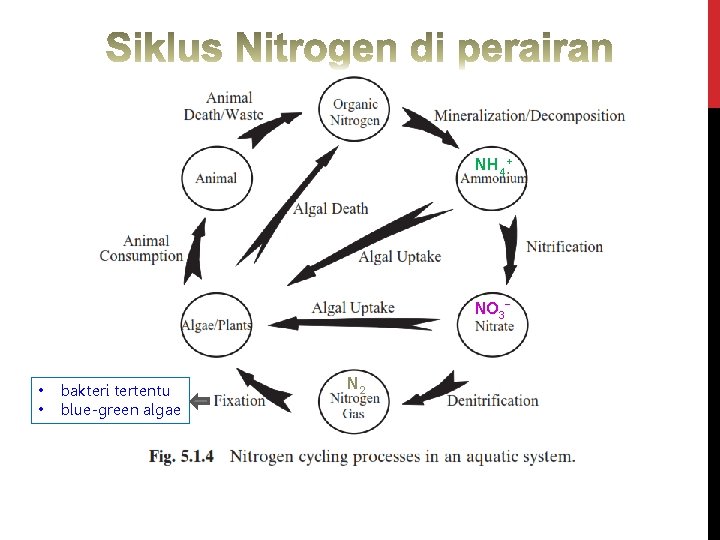
NH 4+ NO 3 • • bakteri tertentu blue-green algae N 2

Ø nitrogen also plays important roles in other water quality processes and may constitute a problem: 1. The oxidation of NH 4 to NO 3 during the nitrification process consumes oxygen and may contribute significantly to the oxygen depletion of a waterbody. 2. High concentrations of un-ionized NH 3 can be toxic to aquatic life. 3. The common form of nitrogen in water is NO 3, which itself is not toxic. However, bacteria in the intestinal tract of infants can convert nitrates to highly reactive NO 2, which may cause the so-called “ blue baby ” syndrome and death from suffocation. There are strict regulations governing the amount of nitrate that can be present in drinking water. nitrogen is not as often limiting to plant growth as phosphorus in many natural waters, especially in freshwaters. Thus, most of the eutrophication management efforts are focused on phosphorus control.

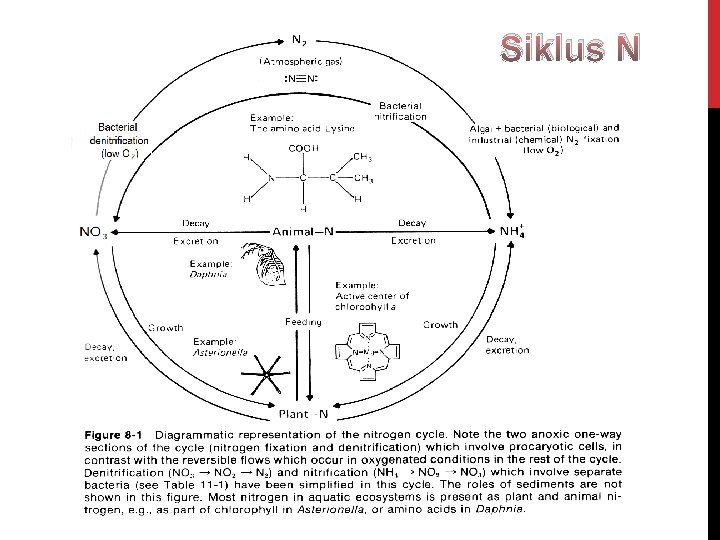
NITROGEN (N) Nitrogen di perairan terdapat dalam berbagai bentuk seperti: 1) 2) 3) 4) 5) 6) NO 3 - (Nitrat) NO 2 - (Nitrit) gas N 2 terlarut NH 3 (Ammonia) – gas terlarut NH 4+ (ion Ammonium), dan N organik kompleks – dlm jumlah besar (org N) 1. N 2 (g) + O 2 (g) 2. 2 NO (g) + O 2 (g) ----> 2 NO 2 (g) 3. 3 NO 2 + H 2 O ----> 2 H+ + 2 NO 3 - + NO Reaksi kesetimbangan amonia dan amonium: 4. NH 3 + H 2 O NH 4 OH NH 4+ + OH-
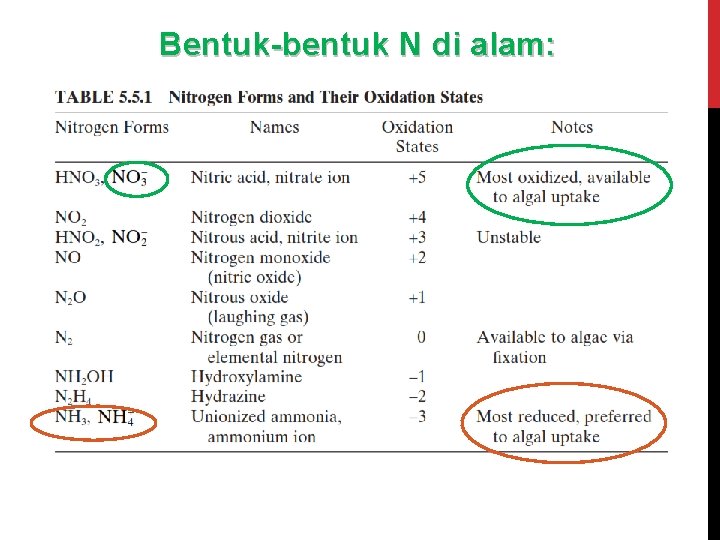
Bentuk-bentuk N di alam:
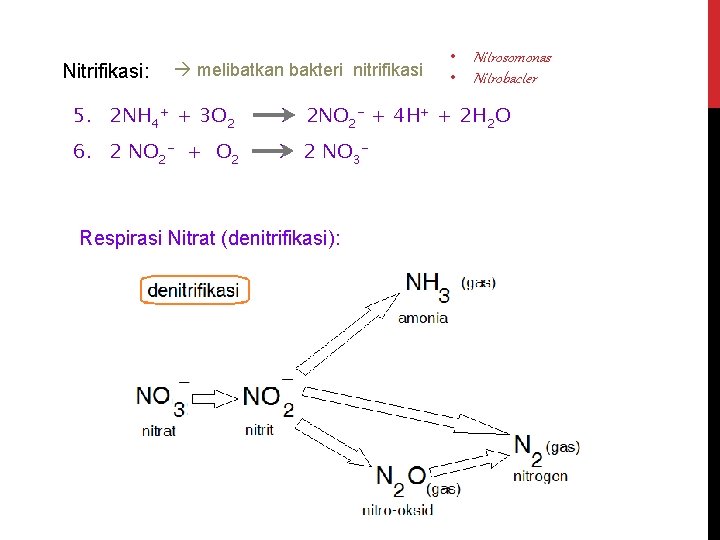
Nitrifikasi: melibatkan bakteri nitrifikasi • Nitrosomonas • Nitrobacter 5. 2 NH 4+ + 3 O 2 2 NO 2 - + 4 H+ + 2 H 2 O 6. 2 NO 2 - + O 2 2 NO 3 - Respirasi Nitrat (denitrifikasi):

Total Nitrogen (TN): Total Kjeldhal Nitrogen (TKN): Total Amonia Nitrogen (TAN) atau TA: total amonia [ ] = konsentrasi (mol/L atau mg/L) N-organik

Ammonia

Ammonia - N § metoda Indophenol (=metoda phenate) ( Utk sampel air dgn kesadahan < 400 mg/L dan berkadar NO 2 -N < 5 mg Phenol dan hypochlorite (chlorox) bereaksi dalam kondisi larutan basa membentuk phenylquinone-monoimine yang selanjutnya akan bereaksi dengan ammonia membentuk indophenol yang berwarna biru. Kepekatan warna biru sebanding dengan kadar ammonia yang ada. Phenol + Hypochlorite ⇨ phenylquinone-monoimine + NH 3 ⇨Indophenol (biru) kondisi alkalin/basa Semua amonia (NH 3 & NH 4+) NH 3 Phenol + NH 3 + 3 Cl. O- ‑‑‑‑> Indophenol + 2 H 2 O + OH- + 3 Cl(Hypochlorite) (biru) Sigid Hariyadi setiap set penentuan ammonia, harus selalu disertai dengan penentuan standar ammonia dan blanko ¤Kadar ammonia yang terukur pada metoda ini adalah ammonia total yaitu terdiri dari NH 3 dan NH 4 -, karena pada larutan bersuasana basa kuat semua ammonia berada dlm bentuk NH 3 TAN

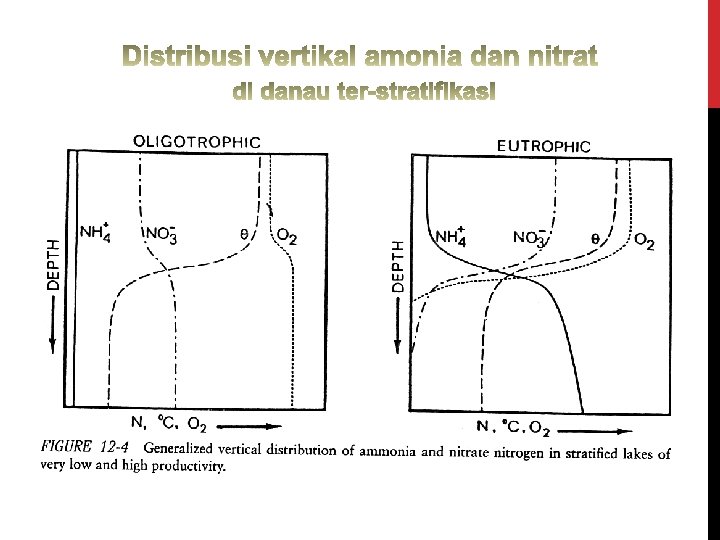
Prosentase NH 3 thd ammonia total (TAN) sangat dipengaruhi oleh: • p. H • temperatur • salinitas air payau & air laut Sigid. Hariyadi Total Ammonia Nitrogen Sumber: Trussel (1972) in Boyd (1979)


% un-ionized ammonia: % NH 3 = 100 1 + antilog (p. Ka – p. H) Temp (o. C) 15 20 22 24 25 26 27 28 29 30 31 32 p. Ka 9, 56 9, 40 9, 33 9, 27 9, 24 9, 21 9, 18 9, 15 9, 12 9, 09 9, 06 9, 03 p. Ka = -log (Ka) Ka : konstanta ionisasi
![Amonia dan Amonium: [ ] = konsentrasi (mol/L) Konstanta kesetimbangan amonia Ke dipengaruhi oleh Amonia dan Amonium: [ ] = konsentrasi (mol/L) Konstanta kesetimbangan amonia Ke dipengaruhi oleh](http://slidetodoc.com/presentation_image_h/0c5dab879d1afc0adbb4748fbd834330/image-20.jpg)
Amonia dan Amonium: [ ] = konsentrasi (mol/L) Konstanta kesetimbangan amonia Ke dipengaruhi oleh temperatur (T): T dalam C pada 25 C
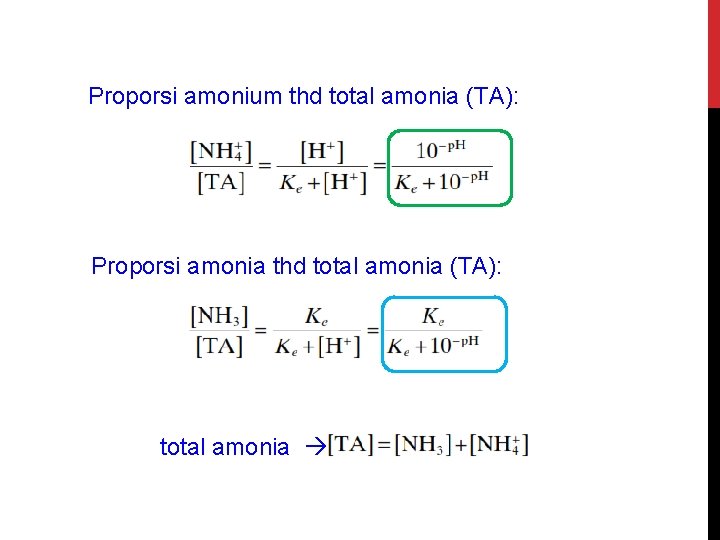
Proporsi amonium thd total amonia (TA): Proporsi amonia thd total amonia (TA): total amonia
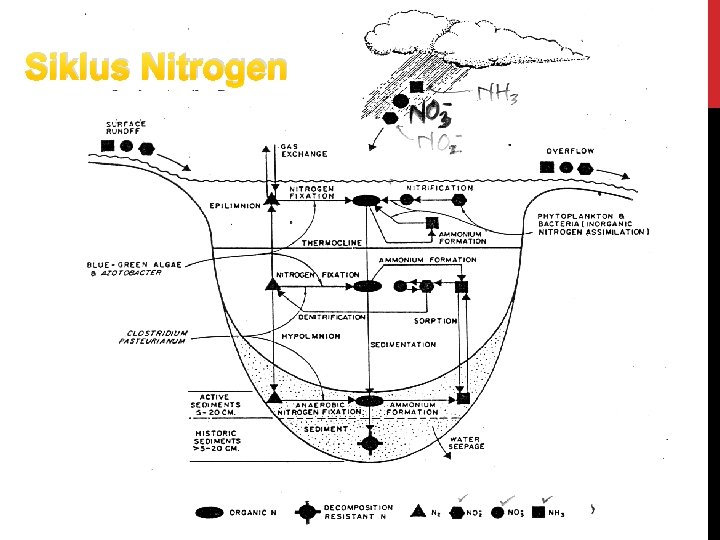
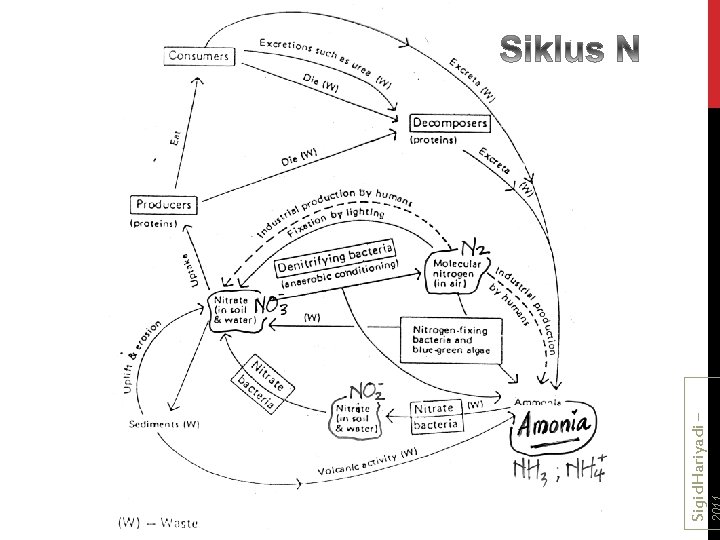
Siklus Nitrogen
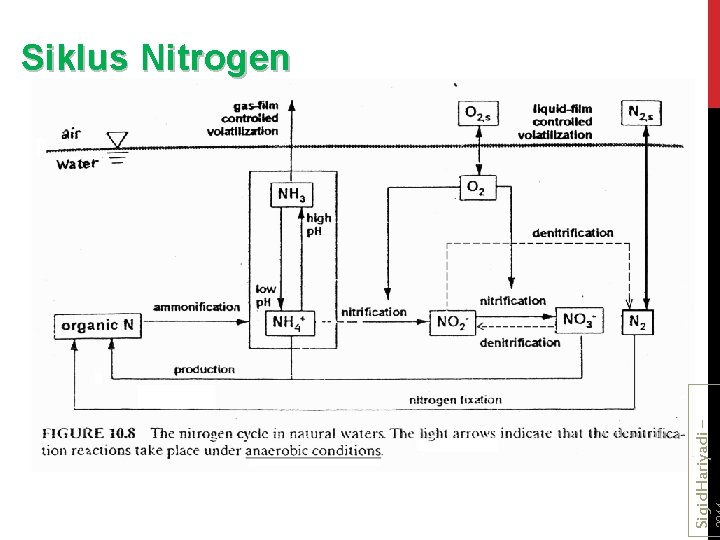
Sigid. Hariyadi – Siklus Nitrogen
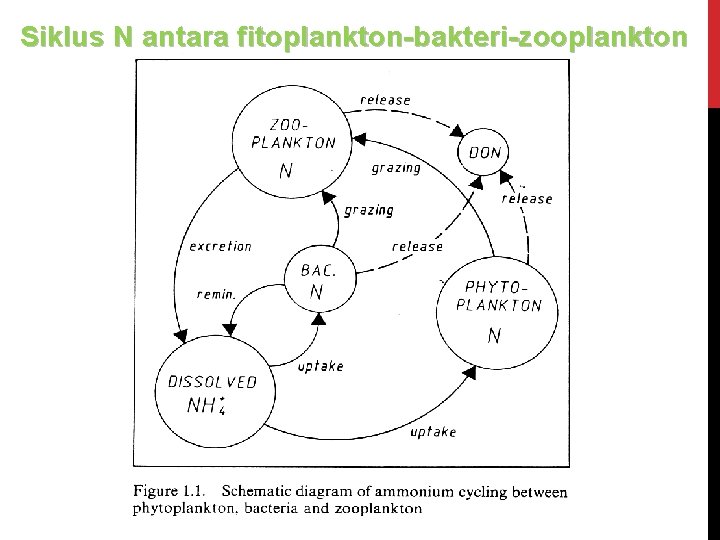
Siklus N antara fitoplankton-bakteri-zooplankton
Siklus N
Sigid. Hariyadi –
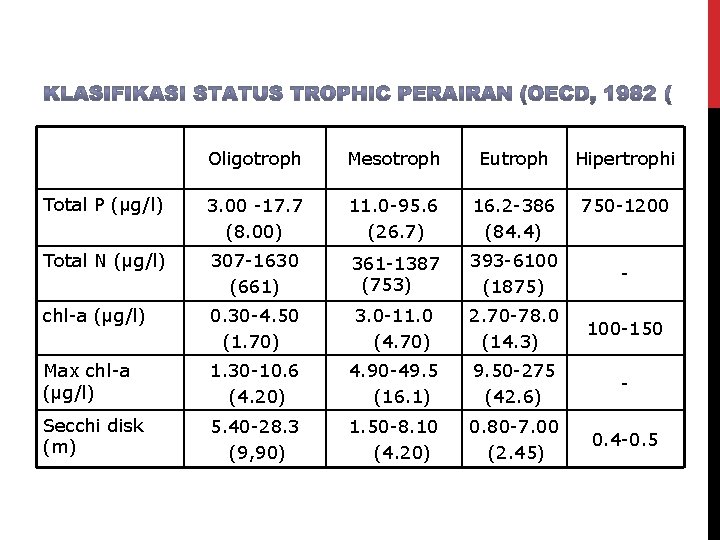
Oligotroph Mesotroph Eutroph Hipertrophi Total P (μg/l) 3. 00 -17. 7 (8. 00) 11. 0 -95. 6 (26. 7) 16. 2 -386 (84. 4) 750 -1200 Total N (μg/l) 307 -1630 (661) 361 -1387 (753) 393 -6100 (1875) - chl-a (μg/l) 0. 30 -4. 50 (1. 70) 3. 0 -11. 0 (4. 70) 2. 70 -78. 0 (14. 3) 100 -150 Max chl-a (μg/l) 1. 30 -10. 6 (4. 20) 4. 90 -49. 5 (16. 1) 9. 50 -275 (42. 6) - Secchi disk (m) 5. 40 -28. 3 (9, 90) 1. 50 -8. 10 (4. 20) 0. 80 -7. 00 (2. 45) 0. 4 -0. 5

Konsep N-P ratio: Di dalam sampel plankton, rasio atom = 106 N-P ratio C : 16 N : 1 P Redfield ratio (1963) Ø sediaan nutrien bagi perkembangan algae Normalnya ⇨ 16 atom N : 1 atom P 7. 2 mg/L N : 1 mg/L P Bila N-P ratio < 7 ⇨ N potensial sbg faktor pembatas Bila N-P ratio > 8 ⇨ P potensial sbg faktor pembatas “Biologically available” N = nitrat + nitrit + ammonia “Bio-available” P = orthophosphate Ø konsentrasi dari masing-masing N dan P tetap perlu diketahui

Referensi: ① ② ③

Terima kasih
- Slides: 31