Numbers Brain Teasers List some similarities and differences

Numbers

Brain Teasers List some similarities and differences between the coke can and the diet coke can. List some similarities and differences between the two rocks.

Data Terms Quantitative Measurements Give results in a definite form, usually values Examples 24 L, 10 cm, 14 ºC

Data Terms Qualitative Measurements Give results in a descriptive, non-numeric form. Examples The beaker was warm. The density was greater than that of water.

Data Terms Accuracy Examples How close a measurement comes to the actual value of whatever is being measured Water freezes at 0º C, and boils at 100º C. How close is the measurement to the values.

Data Terms Precision Reproducibility of the measurement Examples 9 out of 10 lab groups report the temperature of boiling water to be 95º C. A basketball player shoots 20 free throws, 18 of which bounce off the right side of the rim.

Accuracy vs. Precision Target Practice Accurate Precise Accurate & Precise

Percent error Theoretical – Experimental x 100 = % error Theoretical

Closure Give and example of a qualitative and quantitative measurement.

Significant Figures Digits in a measurement that have meaning relative to the equipment being used

Significant Figures Place What is the increment on the equipment? What you know for sure.

Significant Figures Digits with meaning Digits that can be known precisely plus a last digit that must be estimated.

Significant Figures Equipment to Evaluate To what place (tenths, hundredths, etc. ) can these measurement instruments accurately measure? What place is the estimation? Triple beam balance Analytical balance Thermometer Graduated cylinders Beakers Ruler Erlenmeyer Flasks Burette

Brain Teaser #2 What’s the message behind the pictures? + What’s the meaning? Extra pictogram: “HIJKLMNO”

Significant Figures What do you notice? Depends on type of equipment being used. Depends on size of equipment used.

Significant Figures 1. All digits 1 -9 are significant. How do you know 2. Zeros between significant digits how many sig are always significant. figs? 3. Trailing 0’s are significant only if the number contains a decimal point 4. Zeros in the beginning of a number with a decimal point are not significant. 5. Zeros following a significant number with a decimal are significant. Raw Data Rules n

Significant Figures Pacific to Atlantic Rule Examples Pacific = Decimal Present Start from the Pacific (left hand side), every digit beginning with the first 1 -9 integer is significant 20. 0 = 3 sig digits 0. 00320400 = 6 sig digits 1000. = 4 sig digits

Significant Figures Atlantic Rule to Atlantic = Decimal Absent Pacific Start from the Atlantic (right hand side), every digit beginning with the first 1 -9 integer is significant Examples 100020 = 5 sig digits 1000 = 1 sig digits

Practice 1. 2. 3. 4. How many significant figures are in 400. 0 4004 0. 004
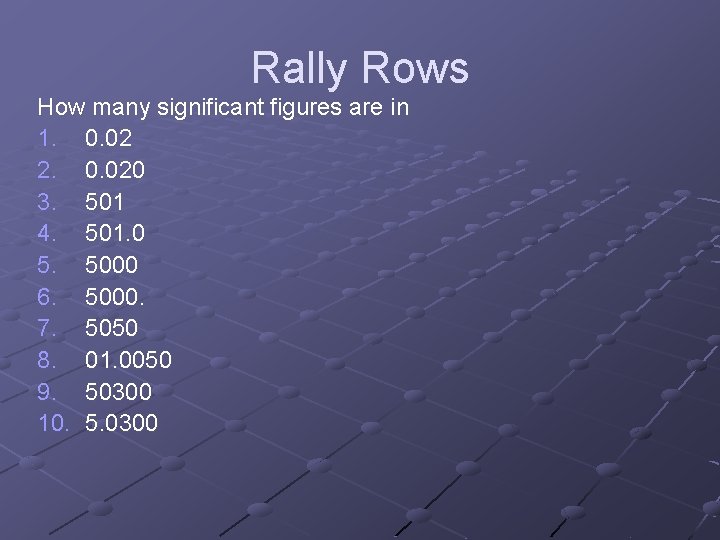
Rally Rows How many significant figures are in 1. 0. 02 2. 0. 020 3. 501 4. 501. 0 5. 5000 6. 5000. 7. 5050 8. 01. 0050 9. 50300 10. 5. 0300

Summary Things to consider What do significant figure s tell you about the measurement equipment? If you wanted to measure the mass of a whale, what scale would you want to use? Would it matter if you know its mass accurately to 1 gram? If you wanted to measure the mass a grain of sand , what scale would you want to use? Would it matter if you know its mass accurately to 1 gram?

Instrument Measure Need to make sure you are measuring and recording to the correct number of digits n Measure what you know for sure and then guess one more digit Rulers n Draw a line on your paper and measure it to the correct number of digits Beaker vs. graduated cylinder Electronic balance vs. triple beam balance

Brain Teaser You and a partner will create a puzzle. Each of you will have certain dimensions. Take a notecard, ruler and scissors. Cut your puzzle piece dimensions. Can you fit them together? Do you believe that using significant figures is important?

Scientific Notation

Scientific Notation Shorthand way of expressing numbers that make them easier to work with Example 6. 02 x 1023 2. 34 x 105 3. 78 x 10 -3

Scientific Notation Any Patterns?

Scientific Notation Rules 1. Base number 1 -9 2. Exponent = the number of times the decimal must be moved to bring the base number to 1 -9. 3. Numbers greater than 1 have a positive exponent, numbers less than 1 a negative exponent
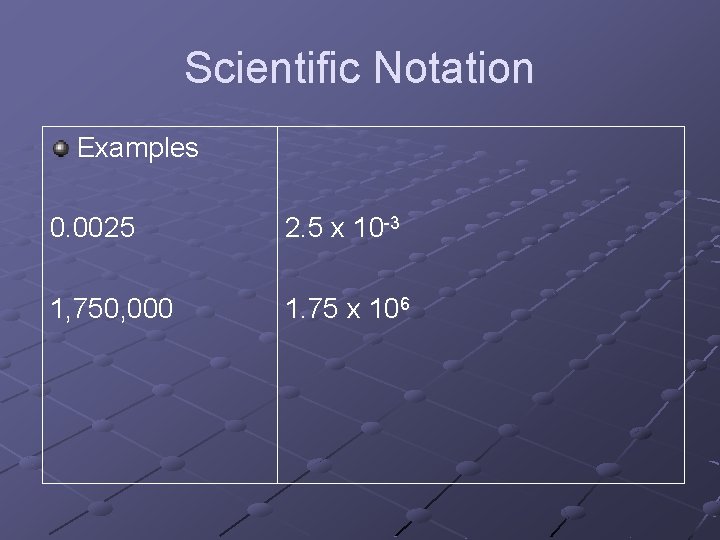
Scientific Notation Examples 0. 0025 2. 5 x 10 -3 1, 750, 000 1. 75 x 106

Scientific Notation Problems 0. 0000678 Express in Scientific Notation 998953000000 0. 5768

Scientific Notation Problems 1. 567 x 10 -3 Express in Standard Notation 6. 02 x 1023 3. 14 x 102

Sig Figs in Scientific Notation The numbers expressed in the scientific notation are significant Examples: 5. 02 x 104 3 S. F The number of significant figures in a set of numbers will be the # of sig figs in the scientific notation. Examples: 50. 200 5 SF 5. 0200 x 10 -1

Survivor Science Convert the following to exponential notation or to ordinary notation Tell me how many Sig Figs. 1. 2. 3. 4. 5. 76 896745 8. 9 x 103 3. 45 x 10 -1 0. 222

6. 5. 38 x 10 -3 7. 5 million 8. 8. 00 x 104 9. 0. 00859 10. 953. 6

Significant Figures in Calculations What are Significant Digits? Examples n n Triple Beam Balance Graduated Cylinder All the certain digits plus the estimated digit in a measurement. How many decimal places can we count

Significant Figures in Calculations Exact Numbers Examples n Infinite # of sig figs Do not affect the number of significant digits in the final answer. They are not measurements!! 1000 m = 1 km 12 in = 1 foot

Significant Figures in Calculations Multiplication and Division The number with the smallest number of significant digits determines how many significant digits are allowed in the final answer. Example Volume of a box n n Lx. Wx. H (3. 05 m)(2. 10 m)(0. 75 m) 2 sig figs 4. 8 m 3

Significant Figures in Calculations Example n n Density of a penny M = 2. 53 g V = 0. 3 m. L n n n D=M/V # significant figures allowed D = 8 g/m. L

Significant Figures in Calculations Addition and Subtraction Example The number of significant digits depends on the number with the largest uncertainty. (you may be using different scales) Shoes 951. 0 g Clothes 1407 g Ring 23. 911 g Glasses 158. 18 g Total 2540. g

Significant Figures in Calculations Example What is the mass of a penny if, the weighing paper alone has a mass 0. 67 g and weighing paper plus the penny has a mass of 3. 2 g -0. 67 g 2. 5 g

Significant Figures in Calculations Remember A calculated number can only be as precise as the least precise measurement in the calculation.

Practice Calculate each of the following to the correct number of significant figures. Include units on your answer. 1. (25 g/mol)(4. 0 mol) = 2. (3. 48 in)(1. 28 in)(0. 010 in) = 3. 2. 06 cm + 1. 8 cm + 0. 004 cm = 4. If the mass of a lead cube is 176. 91 g and it measures 2. 51 cm x 2. 49 cm, what is the density of lead?

Practice Calculate each of the following to the correct number of significant figures. Include units on your answer. 1. (25 g/mol)(4. 0 mol) =1. 0 x 102 2. (3. 48 in)(1. 28 in)(0. 010 in) =. 045 in 3 3. 2. 06 cm + 1. 8 cm + 0. 004 cm = 3. 9 cm 4. If the mass of a lead cube is 176. 91 g and it measures 2. 51 cm x 2. 49 cm, what is the density of lead? 11. 3 g/cm 3

Rally rows Sig figs in Calculations 1. 2. 3. 4. 5. 12 cm + 0. 031 cm + 7. 969 cm = (41. 025 g - 23. 38 g) ÷ 8. 01 m. L= 17. 3 cm x 6. 2 cm + 3. 28 cm 2 = 109. 3758 m 2 45. 813 m = What is the mass of Salt (Na. Cl) if the sodium has a mass of 22. 99 g and the Cl a mass of 35. 5 g?


Brain Teaser 1. 2. 3. 12 cm + 0. 031 cm + 7. 969 cm = (41. 025 g - 23. 38 g) ÷ 8. 01 m. L= Round 546. 51 to 3 sig figs and 2 sig figs

Answers 1. 2. 3. 12 cm + 0. 031 cm + 7. 969 cm =20 cm (41. 025 g - 23. 38 g) ÷ 8. 01 m. L=2. 00 g/ml Round 546. 51 to 3 sig figs and 2 sig figs 547 550

Convert the following to either numbers or scientific notation 6. 5. 38 x 10 -3 7. 50. 00 8. 8. 00 x 104 9. 0. 00859 10. 953. 6

Brain Teaser Find the mass of each penny. Each row will take the average of the penny mass (remember sig figs!!!!) Then each row will take all pennies in that row and find the volume using a graduated cylinder. Calculate the Density Put your group answer on the board.

Conservation of Copper Pre-lab Law of conservation of Energy n The observed fact that in any chemical or physical process energy is neither created nor destroyed. n Matter is energy Einstein’s famous equation n E = mc 2

Conservation of Copper Pre-lab The total amount of energy in the universe never changes. The Law of Conservation of Energy/Matter can be applied to n Closed systems Well defined groups of objects that can transfer energy among one another.

Symbols in chemical equations Aqueous (aq) Solid (s) Liquid (l) Gaseous (g)

Pre-Lab Title Purpose Background Materials Procedures

Lab Discussion

Conservation of Copper Postlab % error calculation Mass Cu Start - Mass Cu End x 100 = Mass Cu Start What are some of the reasons for this error?

Post-Lab Use Rubric as Guide n Organized Data (part 1, 2 etc) % error n Data Analysis Paragraph form – refer to equations to identify substances in observations. n Conclusion Restate purpose and how lab met this purpose Discussion of error, be specific what happened during your experiment? How could the error be controlled or improved?

Brainteaser 2. 0 years to seconds 6. 32 moles = _____ liters (if 22. 4 L = 1 mole)

Brain Sampler You have 5 minutes. Each person in a group will turn in a paper. (5. 31 x 10 -2 cm) x ( 2. 46 x 105 cm) 3. 56 g + 9. 0 g + 2. 300 g = The density of a substance is 3. 9 g/ml. It’s mass is 50. 0 g. What is the volume of the substance? (4. 5 ml x 3. 49 ml) + 45 ml 2

Dimensional Analysis

Dimensional Analysis § A tool. An organized technique of converting between units. Example § How many eggs are in two cases? 1 case = 24 cartons 1 carton = 12 eggs

Dimensional Analysis Equivalence Statement Examples Conversion Factors Examples Relates the same quantity in two different units An equation that is always equal. 1 gallon = 3. 785 L 2. 54 cm = 1 inch Ratio between units in the equivalence statement

Dimensional Analysis How to use Dimensional Analysis (DA) 1. Identify the unknown. What is the problem asking you to find? 2. Start with the given (often the first # in the problem) 3. Identify and set up the conversion factors for each unit of change 4. Make sure all units cancel EXCEPT the one you are looking for. 5. Check that you have the correct number of significant figures. 6. Ask whether your answer makes sense.

Brain Teaser Usain Bolt, a Jamaican Olympian, just ran a world record for a 100 meter dash sprint. He ran this race in 9. 68 seconds beating the world record by 1 -one hundredth of a second (9. 69 s). How fast was he running? How fast was he running in miles per hour? n n n 1 mile = 1609. 34 meter 60 seconds = 1 minute 60 minutes = 1 hour

Sample Problems 1. How many kilograms are in 125 g of salt? 2. Chloroform is a liquid once used as and anesthetic. What is the volume in m. L of 5. 0 g of chloroform. Density = 1. 49 g/m. L? 3. The length of a marathon race is approximately 26. 2 miles. What is the distance of the race in kilometers? 5280. feet = 1 mile 4. Convert the freeway speed limit of 65 miles per hour to kilometers per second. 1 km = 0. 62 mi



Start with the given, end with what you need!

Practice 1. How many pieces of paper are in 3 boxes of copy paper if each box contains 16 reams and each ream is 500 pieces of paper?

TEAM The Perfect Cup of Kool-Aid It is a hot summer day and you are extremely thirsty. You decide that a cup of Kool-aid would be perfect to quench your thirst. You find the following recipe for the perfect cup of Kool-aid: n 0. 05 cups of Kool-aid powder should be mixed with 7. 5 fluid ounces of water. However, in your kitchen you only have teaspoon with which to measure. Using dimensional analysis and the given equivalence statements, determine the number of teaspoons of Kool-aid and water you need to make your drink. 5 m. L = 1 tsp 17 Tbsp = 1 cup 1 Tbsp = 15 m. L 8 fl oz = 1 cup

Summary What is dimensional analysis used for? Why is it useful? What must accompany every number (measurement) in chemistry?

Warm Up 6 Describe at least 3 qualitative and quantitative measurements on the Regular coke and diet coke. Are they the same? If not, why not?

Density Intensive Physical Property of Matter. n Does not matter how much matter is present the density will not change. Unique for each type of matter Mass per unit of volume g/m. L or g/cm 3 1 m. L = 1 cm 3

Density Calculation An unknown colorless liquid has a mass of 30. 6 g and a volume of 38. 9 m. L. What is the density of the liquid? What is the identity of the liquid? An unknown metal has a mass of 1. 25 g and a volume of 0. 0587 cm 3? What is the identity of the metal?

Density Demos Which soda has a greater density? Diet or Regular What is the density? How could this be measured?

Graphing Density What is the density of rubber? Material given 5 different rubber stoppers Balance Ruler Graduated cylinder

Graphing Must include n n n Title – that is descriptive of the graph Labeled axes - with units Scaled axes – adjust to fill paper


Density as a conversion factor What is the density of rubber? If a rubber stopper has a volume of 8. 2 m. L, what is the mass in grams?

Density Calculation If a block of rubber measures 10. 32 cm wide, 46. 15 cm long and 0. 14 cm thick what is the mass of the rubber?

Error in Density of Rubber Percent Error What is the density of rubber determined form our graph? What is the density of rubber listed on the density table? Actual - Experimental x 100 = Actual

D. A. Practice Dimensional Analysis: Factor Label Method

Review Homework Whiteboard assigned problems Each group member must present an aspect of the problem to the class n n What is the problem asking you to find? What units? What information are you starting with? What units? How to set up the conversion factors How the significant figures are determined

Practice 1. A large river flows at the rate of 2. 6 x 105 cm 3/s into a boat lock that has a volume of 40, 500 m 3. How long before the lock will be filled with water? 2. Convert 3. 0 x 108 m/s to miles per hour. (1600 m =1 mile)

DA Quiz Turn quiz over on desk when finished Wait quietly

Penny Lab During this Lab you will investigate pennies that were minted before and after 1980. Some things to consider n n n What equipment is available to use? How many significant digits can be measured? How could error enter into your experiment? Have procedures approved n Must have sketched data table Perform the investigation.

Board Meeting Sketch of your Graph What year did the counterfeiting begin? Does it continue today? What is the counterfeit penny made of? n How do you know?
- Slides: 85