Nuevos paradigmas en 1 era Lnea del CPNM











- Slides: 41

Nuevos paradigmas en 1 era Línea del CPNM avanzado sin mutaciones drivers. Delvys Rodríguez Abreu, MD Medical Oncology Dept. Hospital Universitario Insular de Gran Canaria. Spain

Conflictos de intereses • He recibido honorarios de Conferencias y Advisory Board de Bristol. Myers-Squibb, Merck Sharp & Dohme, Hoffmann-La Roche, Pierre-Fabre, Novartis, Boehringer, Pfizer, Lilly, Astra-Zeneca.

• Monotherapy

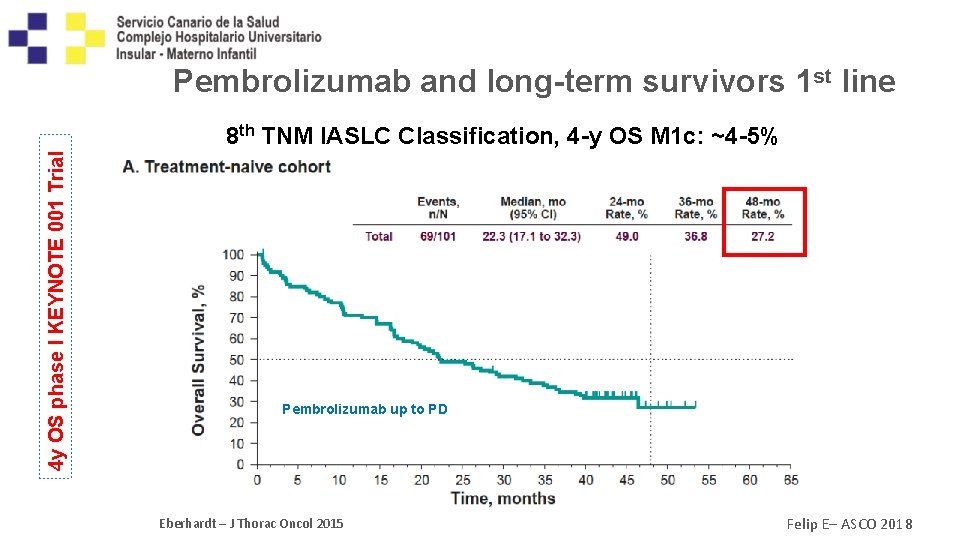
Pembrolizumab and long-term survivors 1 st line 4 y OS phase I KEYNOTE 001 Trial 8 th TNM IASLC Classification, 4 -y OS M 1 c: ~4 -5% PD-L 1 > 50% (N=138) 3 y. OS: 29. 7% Pembrolizumab up to PD Eberhardt – J Thorac Oncol 2015 Felip E– ASCO 2018

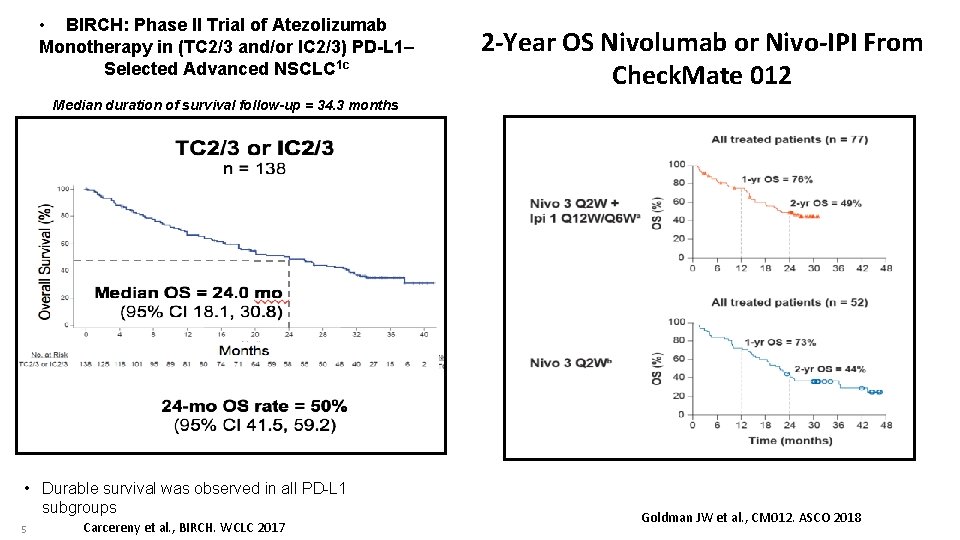
• BIRCH: Phase II Trial of Atezolizumab Monotherapy in (TC 2/3 and/or IC 2/3) PD-L 1– Selected Advanced NSCLC 1 c 2 -Year OS Nivolumab or Nivo-IPI From Check. Mate 012 Median duration of survival follow-up = 34. 3 months • Durable survival was observed in all PD-L 1 subgroups 5 Carcereny et al. , BIRCH. WCLC 2017 Goldman JW et al. , CM 012. ASCO 2018

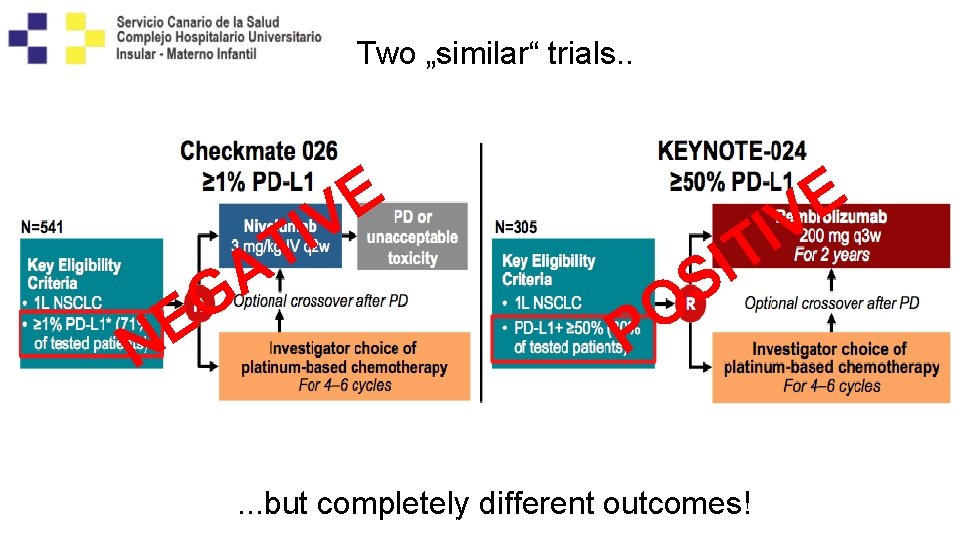
Two „similar“ trials. . E V I T E N A G O P E V I T I S . . . but completely different outcomes! Carbone D et al, NEJM 2017; 376 (25): 2415 -2426; Reck M et al, NEJM 2016; 375 (19):

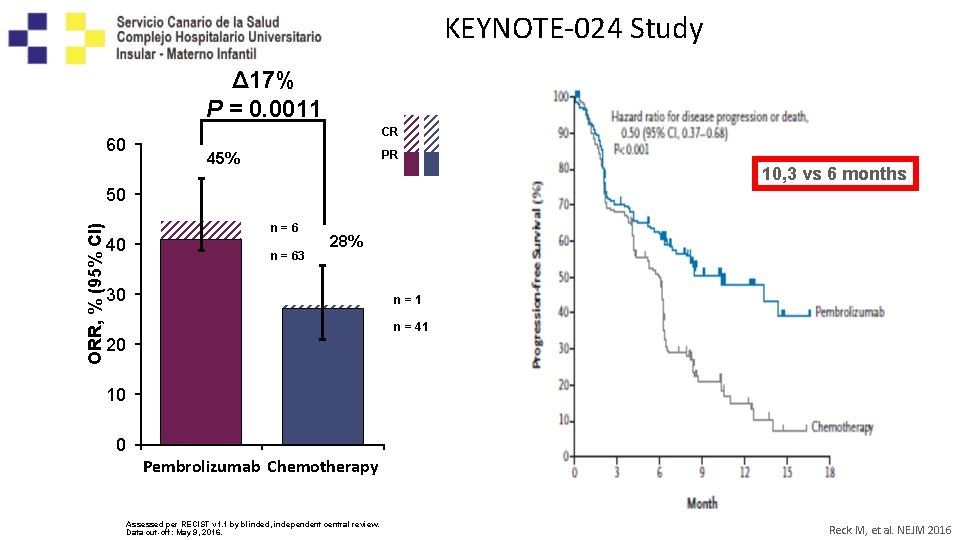
KEYNOTE-024 Study Confirmed Objective Response Rate Δ 17% P = 0. 0011 60 CR PR 45% 10, 3 vs 6 months ORR, % (95% CI) 50 40 n=6 n = 63 28% 30 n=1 n = 41 20 10 0 Pembrolizumab Chemotherapy Assessed per RECIST v 1. 1 by blinded, independent central review. Data cut-off: May 9, 2016. Reck M, et al. NEJM 2016

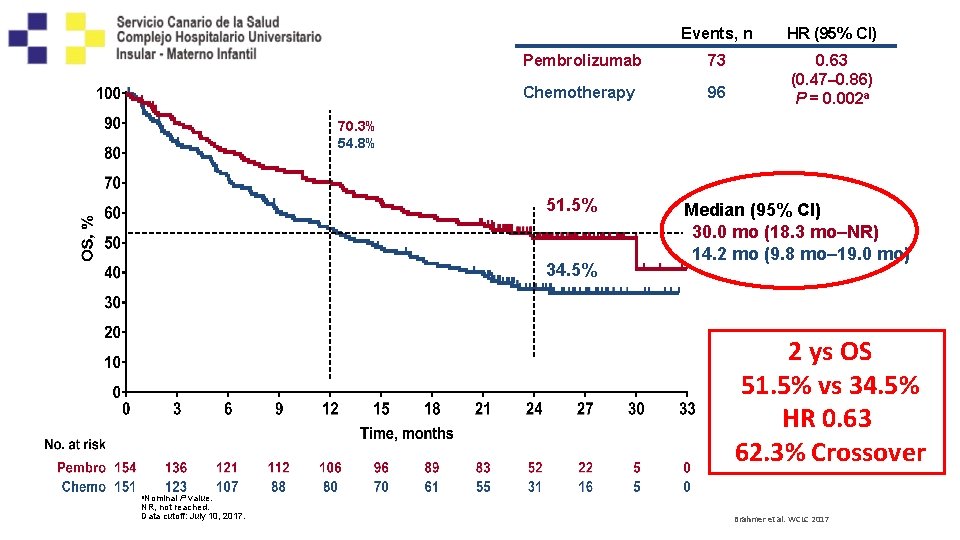
Kaplan-Meier Estimate of OS: Pembrolizumab Updated Analysis Chemotherapy Events, n HR (95% CI) 73 0. 63 (0. 47– 0. 86) P = 0. 002 a 96 70. 3% 54. 8% 51. 5% 34. 5% Median (95% CI) 30. 0 mo (18. 3 mo–NR) 14. 2 mo (9. 8 mo– 19. 0 mo) 2 ys OS 51. 5% vs 34. 5% HR 0. 63 62. 3% Crossover a. Nominal P value. NR, not reached. Data cutoff: July 10, 2017. Brahmer et al. WCLC 2017

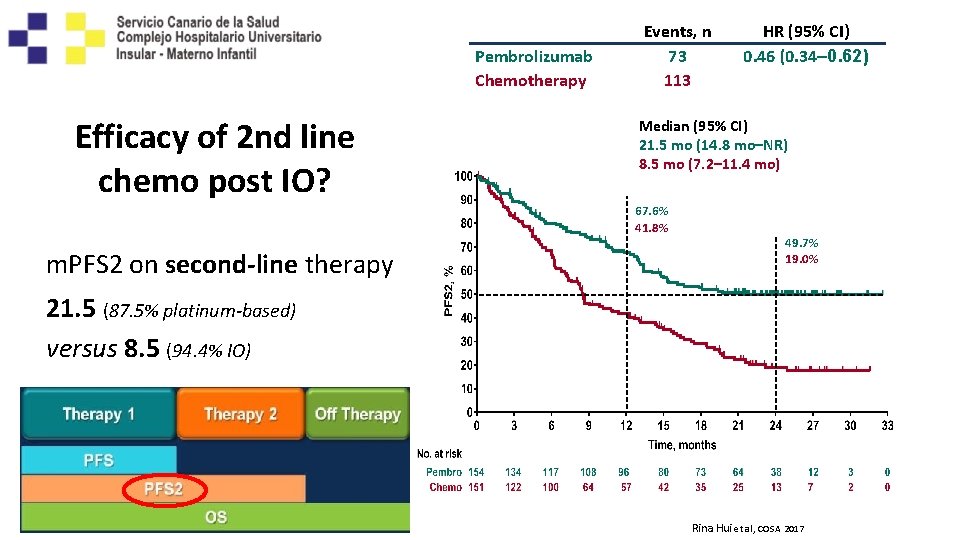
Pembrolizumab Chemotherapy Efficacy of 2 nd line chemo post IO? Events, n 73 113 Median (95% CI) 21. 5 mo (14. 8 mo–NR) 8. 5 mo (7. 2– 11. 4 mo) 67. 6% 41. 8% m. PFS 2 on second-line therapy HR (95% CI) 0. 46 (0. 34– 0. 62) 49. 7% 19. 0% 21. 5 (87. 5% platinum-based) versus 8. 5 (94. 4% IO) Rina Hui et al, COSA 2017

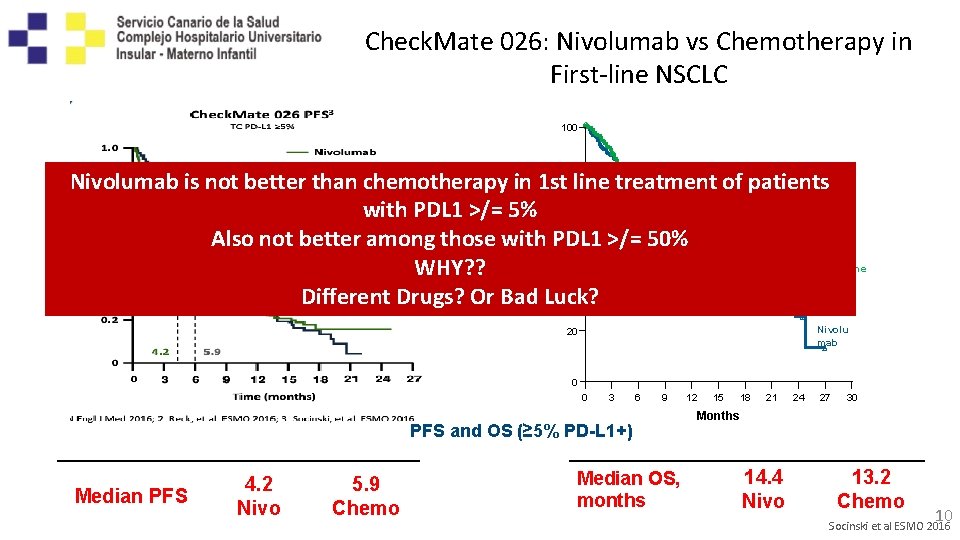
Check. Mate 026: Nivolumab vs Chemotherapy in First-line NSCLC 100 OS (%) Nivolumab is not better than chemotherapy in 1 st 80 line treatment of patients with PDL 1 >/= 5% 60 HR = 1. 02 Also not better among those with PDL 1 >/= 50% Chemothe WHY? ? 40 rapy Different Drugs? Or Bad Luck? Nivolu mab 20 0 0 3 6 9 PFS and OS (≥ 5% PD-L 1+) Median PFS 4. 2 Nivo 5. 9 Chemo Median OS, months 12 15 18 21 24 27 30 Months 14. 4 Nivo 13. 2 Chemo 10 Socinski et al ESMO 2016

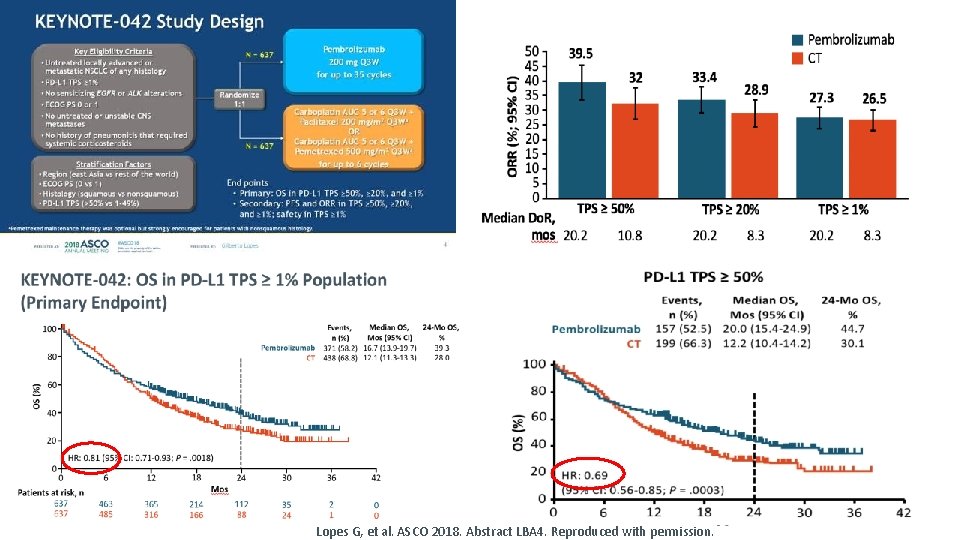
Lopes G, et al. ASCO 2018. Abstract LBA 4. Reproduced with permission.

• Combinations

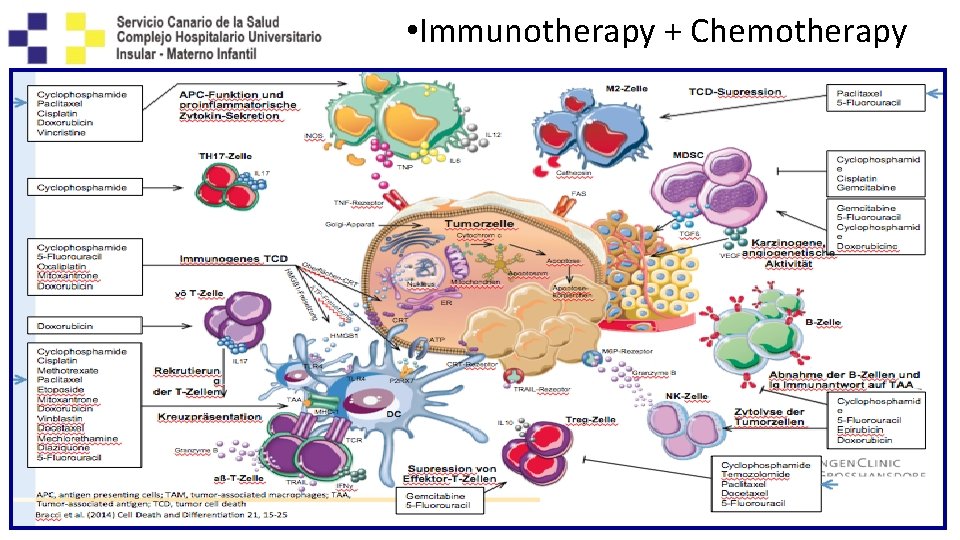
• Immunotherapy + Chemotherapy

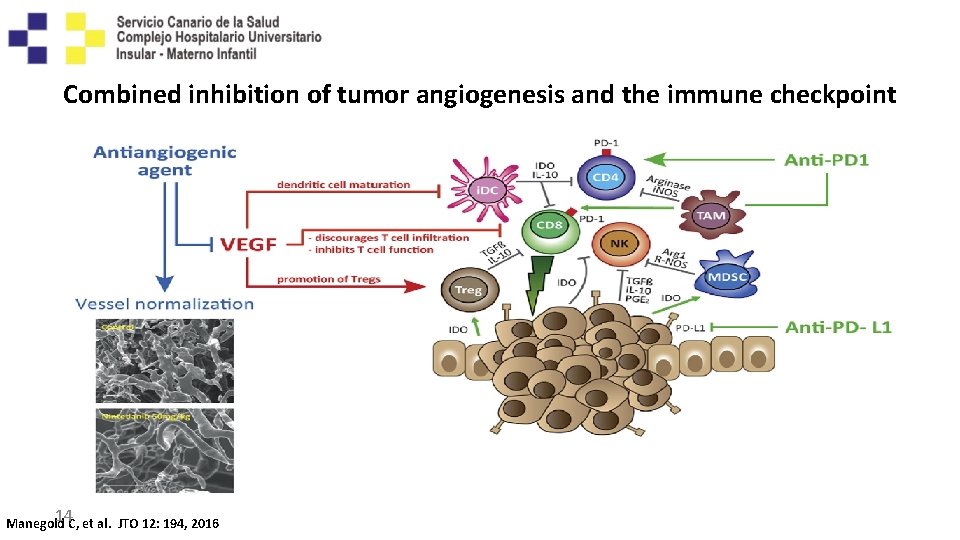
Combined inhibition of tumor angiogenesis and the immune checkpoint 14 Manegold C, et al. JTO 12: 194, 2016

• Non-squamous NSCLC

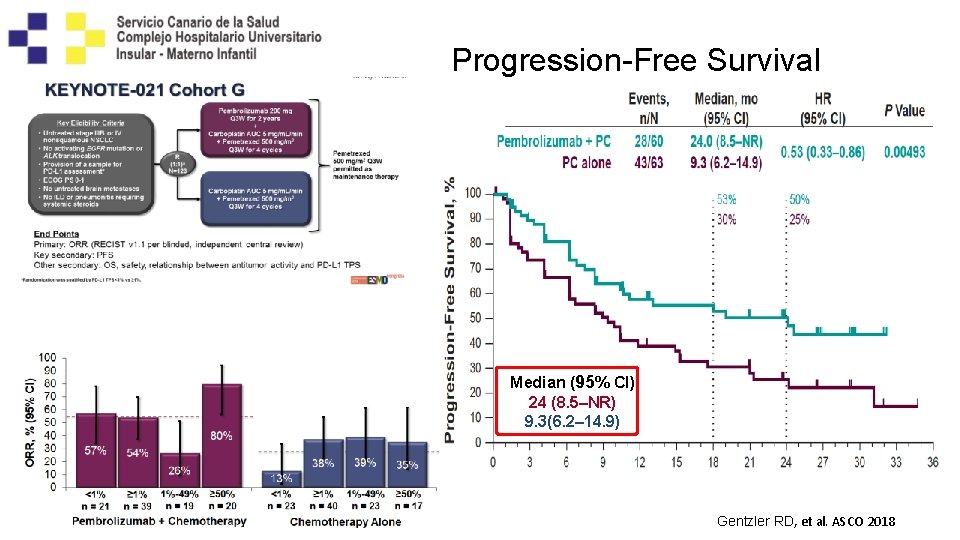
Progression-Free Survival Median ((95% % CI) 24 (8. 5–NR) 24 9. 3(6. 2– 14. 9) Gentzler RD, et al. ASCO 2018

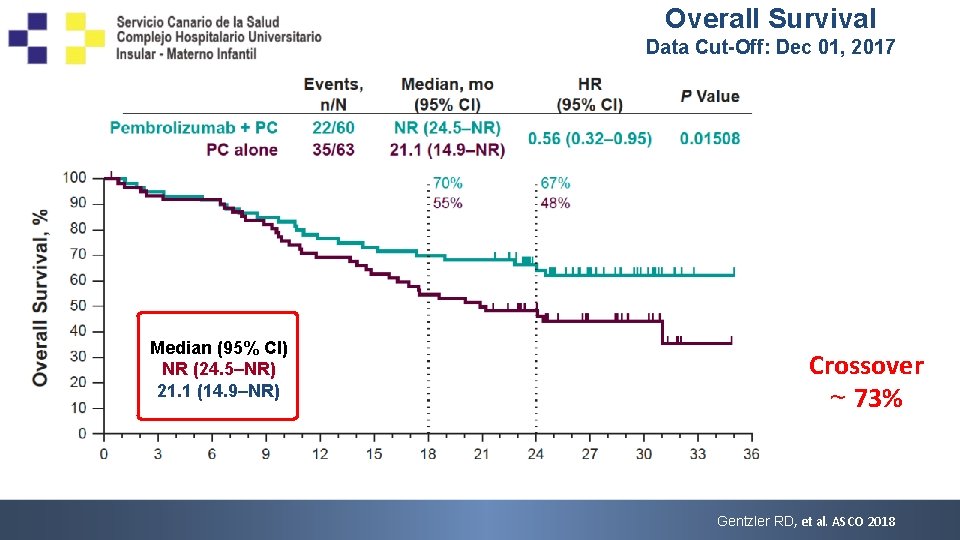
Overall Survival Data Cut-Off: Dec 01, 2017 Median (95% CI) NR (24. 5–NR) 21. 1 (14. 9–NR) • a 24 Crossover ~ 73% additional deaths since primary analysis (pembro + PC, n = 7; PC alone, n = 17). b. P value is descriptive (one-sided P < 0. 025). Gentzler RD, et al. ASCO 2018

May 10, 2017 FDA approved Pembrolizumab in combination with Carbo-Pemetrexed in 1 L NSCLC.

• Phase 3 Trials in Non-squamous NSCLC

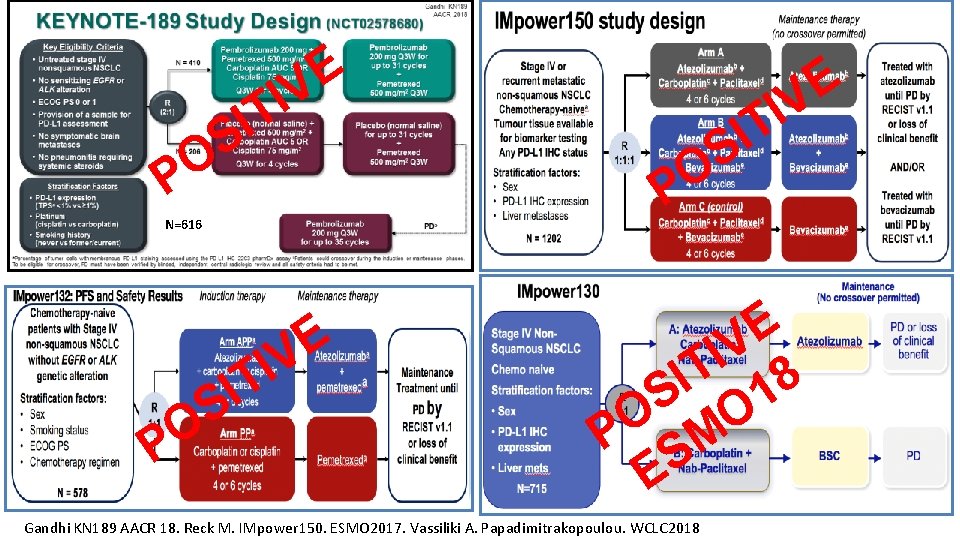
E V I T I S O P N=616 E IV T I S O P E V I T 8 I 1 S O O P SM E Gandhi KN 189 AACR 18. Reck M. IMpower 150. ESMO 2017. Vassiliki A. Papadimitrakopoulou. WCLC 2018

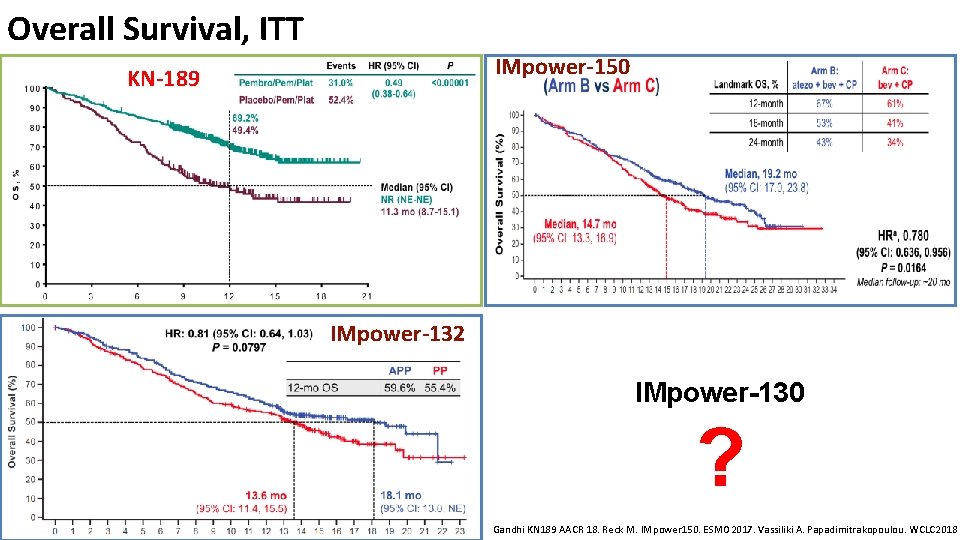
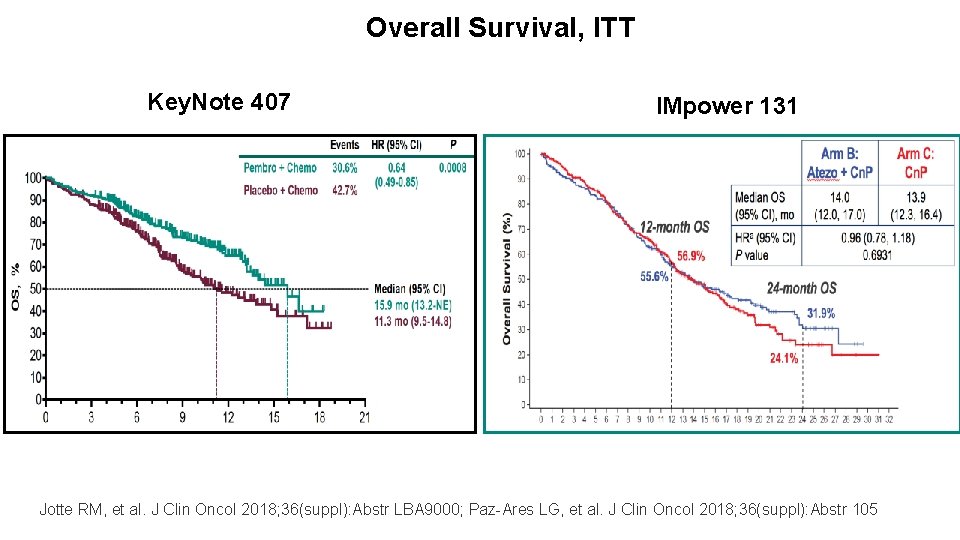
Overall Survival, ITT IMpower-150 KN-189 IMpower-132 IMpower-130 ? Gandhi KN 189 AACR 18. Reck M. IMpower 150. ESMO 2017. Vassiliki A. Papadimitrakopoulou. WCLC 2018

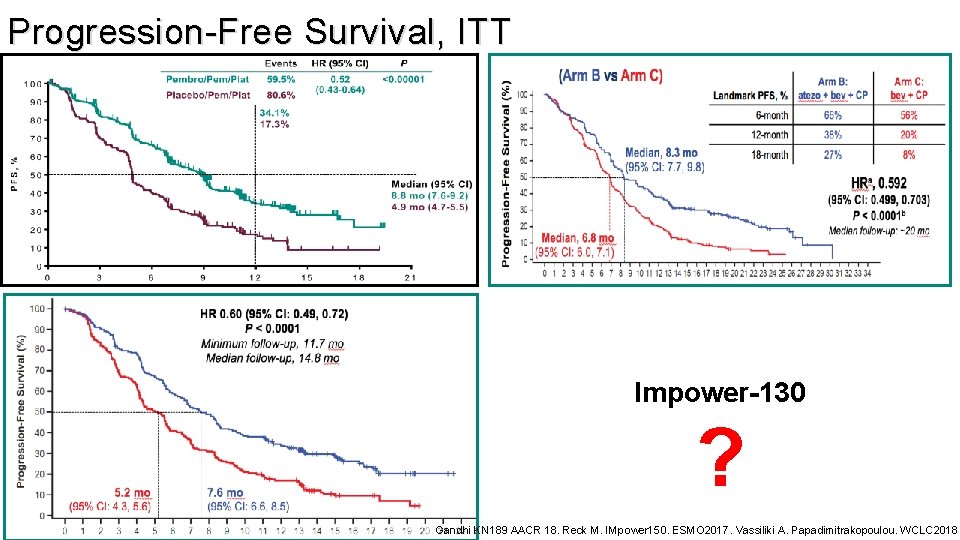
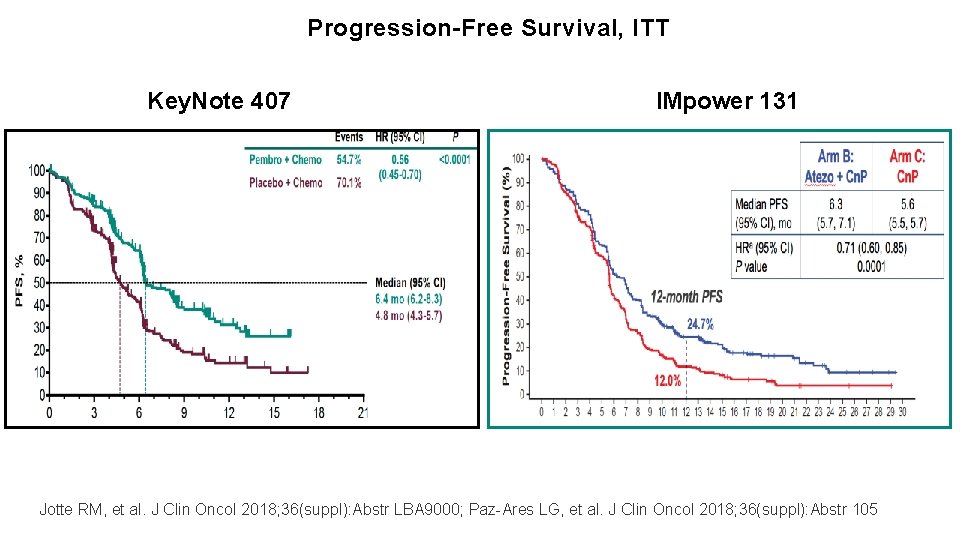
Progression-Free Survival, ITT Impower-130 ? Gandhi KN 189 AACR 18. Reck M. IMpower 150. ESMO 2017. Vassiliki A. Papadimitrakopoulou. WCLC 2018

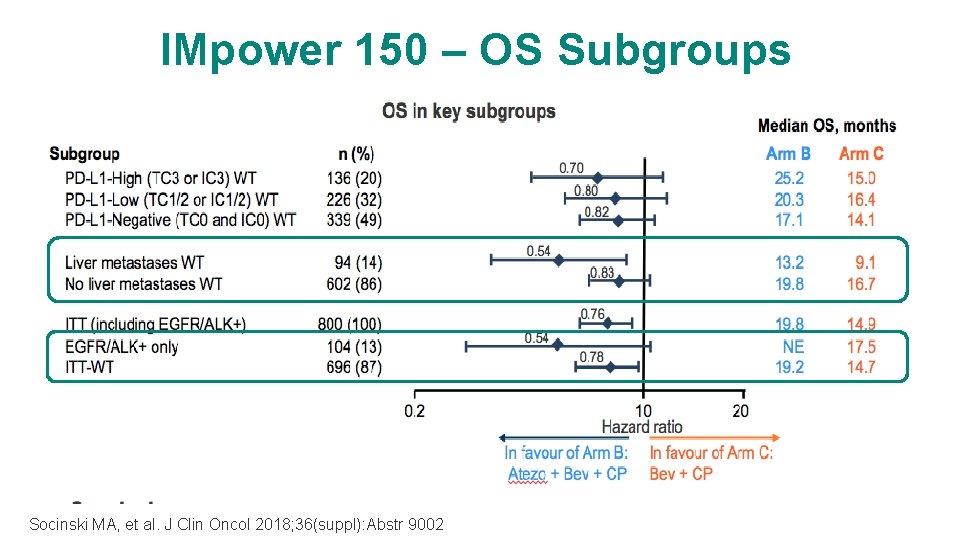
IMpower 150 – OS Subgroups Socinski MA, et al. J Clin Oncol 2018; 36(suppl): Abstr 9002

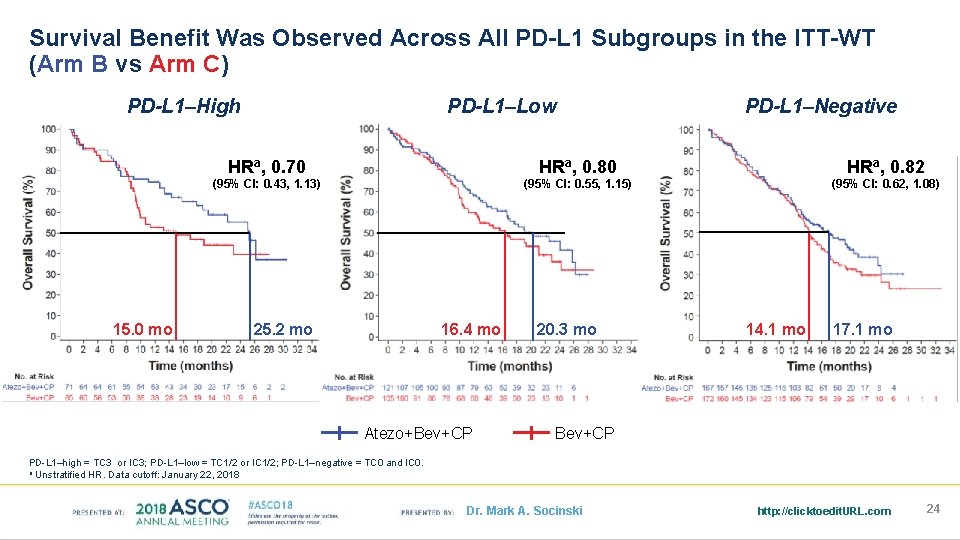
Survival Benefit Was Observed Across All PD-L 1 Subgroups in the ITT-WT (Arm B vs Arm C) PD-L 1–High 15. 0 mo PD-L 1–Low PD-L 1–Negative HRa, 0. 70 HRa, 0. 82 (95% CI: 0. 43, 1. 13) (95% CI: 0. 55, 1. 15) (95% CI: 0. 62, 1. 08) 25. 2 mo 16. 4 mo Atezo+Bev+CP 20. 3 mo 14. 1 mo 17. 1 mo Bev+CP PD-L 1–high = TC 3 or IC 3; PD-L 1–low = TC 1/2 or IC 1/2; PD-L 1–negative = TC 0 and IC 0. a Unstratified HR. Data cutoff: January 22, 2018 Dr. Mark A. Socinski http: //clicktoedit. URL. com 24

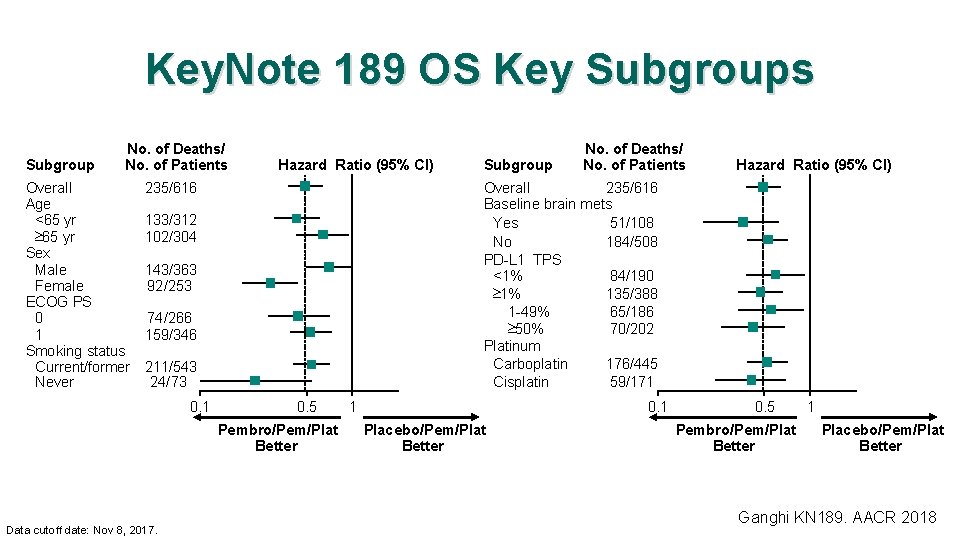
Key. Note 189 OS Key Subgroups Subgroup No. of Deaths/ No. of Patients Overall Age <65 yr ³ 65 yr Sex Male Female ECOG PS 0 1 Smoking status Current/former Never Hazard Ratio (95% CI) 235/616 143/363 92/253 74/266 159/346 211/543 24/73 0. 5 Pembro/Pem/Plat Better Data cutoff date: Nov 8, 2017. Hazard Ratio (95% CI) Overall 235/616 Baseline brain mets Yes 51/108 No 184/508 PD-L 1 TPS <1% 84/190 ³ 1% 135/388 1 -49% 65/186 ³ 50% 70/202 Platinum Carboplatin 176/445 Cisplatin 59/171 133/312 102/304 0. 1 Subgroup No. of Deaths/ No. of Patients 1 0. 1 Placebo/Pem/Plat Better 0. 5 Pembro/Pem/Plat Better 1 Placebo/Pem/Plat Better Ganghi KN 189. AACR 2018

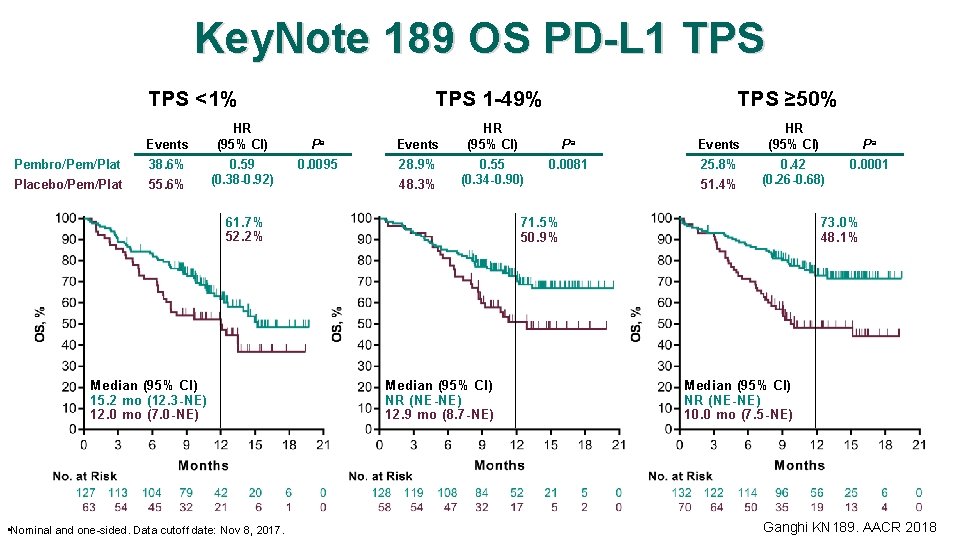
Key. Note 189 OS PD-L 1 TPS <1% Events Pembro/Pem/Plat 38. 6% Placebo/Pem/Plat 55. 6% HR (95% CI) 0. 59 (0. 38 -0. 92) TPS 1 -49% Pa Events 0. 0095 28. 9% 48. 3% HR (95% CI) a Nominal and one-sided. Data cutoff date: Nov 8, 2017. Pa 0. 55 (0. 34 -0. 90) 61. 7% 52. 2% Median (95% CI) 15. 2 mo (12. 3 -NE) 12. 0 mo (7. 0 -NE) TPS ≥ 50% 0. 0081 Events 25. 8% 51. 4% HR (95% CI) 0. 42 (0. 26 -0. 68) 71. 5% 50. 9% Median (95% CI) NR (NE-NE) 12. 9 mo (8. 7 -NE) Pa 0. 0001 73. 0% 48. 1% Median (95% CI) NR (NE-NE) 10. 0 mo (7. 5 -NE) Ganghi KN 189. AACR 2018

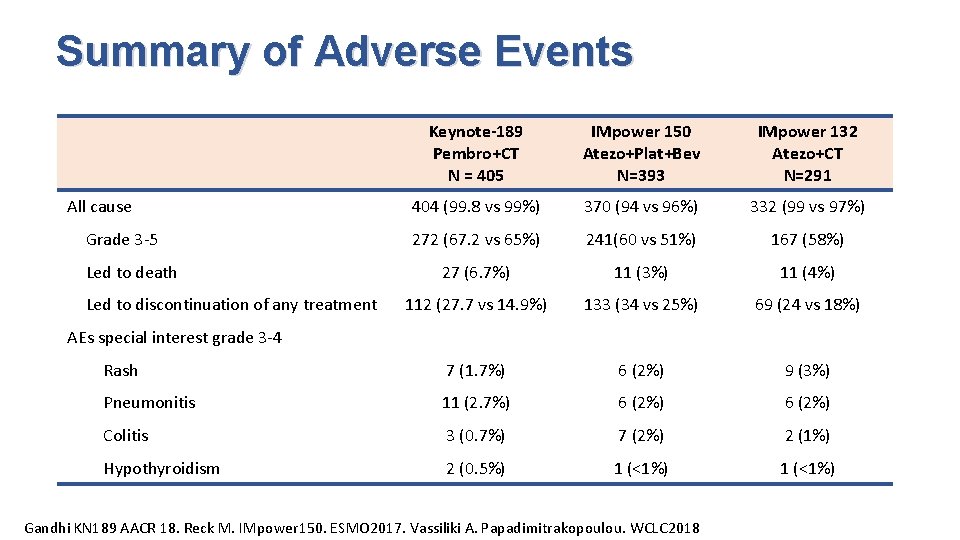
Summary of Adverse Events Keynote-189 Pembro+CT N = 405 IMpower 150 Atezo+Plat+Bev N=393 IMpower 132 Atezo+CT N=291 404 (99. 8 vs 99%) 370 (94 vs 96%) 332 (99 vs 97%) 272 (67. 2 vs 65%) 241(60 vs 51%) 167 (58%) 27 (6. 7%) 11 (3%) 11 (4%) 112 (27. 7 vs 14. 9%) 133 (34 vs 25%) 69 (24 vs 18%) Rash 7 (1. 7%) 6 (2%) 9 (3%) Pneumonitis 11 (2. 7%) 6 (2%) Colitis 3 (0. 7%) 7 (2%) 2 (1%) Hypothyroidism 2 (0. 5%) 1 (<1%) All cause Grade 3 -5 Led to death Led to discontinuation of any treatment AEs special interest grade 3 -4 Gandhi KN 189 AACR 18. Reck M. IMpower 150. ESMO 2017. Vassiliki A. Papadimitrakopoulou. WCLC 2018

THESE ARE PRACTICE CHANGING DATA!!!!

• Squamous NSCLC

Key. Note 407 IMpower 131 E IV T I S O P E V I T I S O P N=559 Jotte RM, et al. J Clin Oncol 2018; 36(suppl): Abstr LBA 9000; Paz-Ares LG, et al. J Clin Oncol 2018; 36(suppl): Abstr 105

Overall Survival, ITT Key. Note 407 IMpower 131 Jotte RM, et al. J Clin Oncol 2018; 36(suppl): Abstr LBA 9000; Paz-Ares LG, et al. J Clin Oncol 2018; 36(suppl): Abstr 105

Progression-Free Survival, ITT Key. Note 407 IMpower 131 Jotte RM, et al. J Clin Oncol 2018; 36(suppl): Abstr LBA 9000; Paz-Ares LG, et al. J Clin Oncol 2018; 36(suppl): Abstr 105

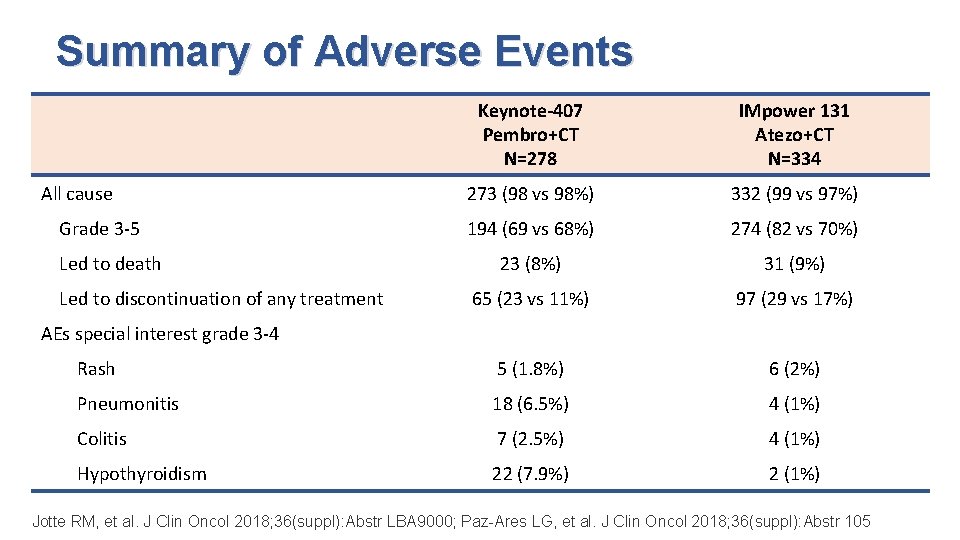
Summary of Adverse Events Keynote-407 Pembro+CT N=278 IMpower 131 Atezo+CT N=334 273 (98 vs 98%) 332 (99 vs 97%) 194 (69 vs 68%) 274 (82 vs 70%) 23 (8%) 31 (9%) 65 (23 vs 11%) 97 (29 vs 17%) Rash 5 (1. 8%) 6 (2%) Pneumonitis 18 (6. 5%) 4 (1%) Colitis 7 (2. 5%) 4 (1%) Hypothyroidism 22 (7. 9%) 2 (1%) All cause Grade 3 -5 Led to death Led to discontinuation of any treatment AEs special interest grade 3 -4 Jotte RM, et al. J Clin Oncol 2018; 36(suppl): Abstr LBA 9000; Paz-Ares LG, et al. J Clin Oncol 2018; 36(suppl): Abstr 105

Immunotherapy + Immunotherapies

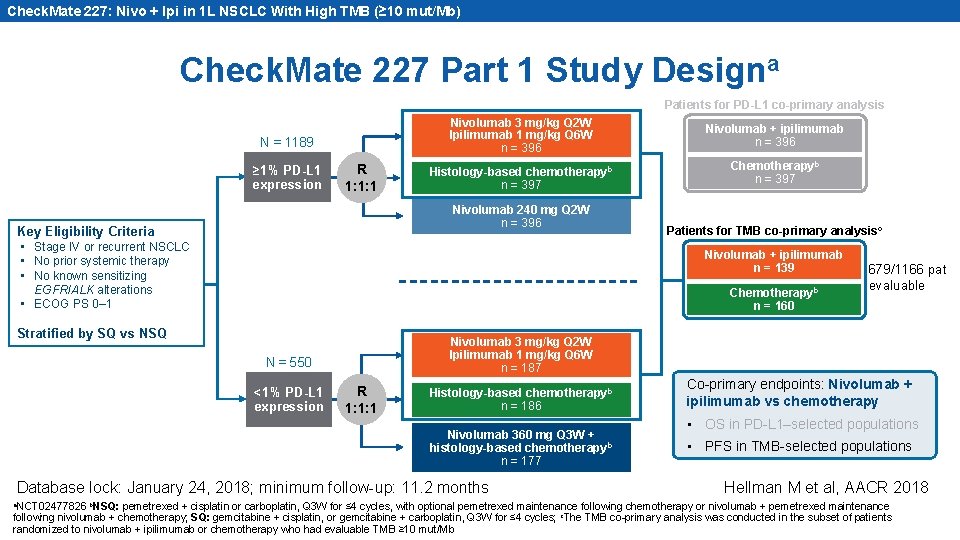
Check. Mate 227: Nivo + Ipi in 1 L NSCLC With High TMB (≥ 10 mut/Mb) Check. Mate 227 Part 1 Study Designa Patients for PD-L 1 co-primary analysis N = 1189 ≥ 1% PD-L 1 expression R 1: 1: 1 Nivolumab 3 mg/kg Q 2 W Ipilimumab 1 mg/kg Q 6 W n = 396 Nivolumab + ipilimumab n = 396 Histology-based chemotherapyb n = 397 Chemotherapyb n = 397 Nivolumab 240 mg Q 2 W n = 396 Key Eligibility Criteria • Stage IV or recurrent NSCLC • No prior systemic therapy • No known sensitizing EGFR/ALK alterations • ECOG PS 0– 1 Nivolumab + ipilimumab n = 139 Chemotherapyb n = 160 Stratified by SQ vs NSQ <1% PD-L 1 expression 679/1166 pat evaluable Nivolumab 3 mg/kg Q 2 W Ipilimumab 1 mg/kg Q 6 W n = 187 N = 550 R 1: 1: 1 Histology-based chemotherapyb n = 186 Nivolumab 360 mg Q 3 W + histology-based chemotherapyb n = 177 Database lock: January 24, 2018; minimum follow-up: 11. 2 months a. NCT 02477826 b. NSQ: Patients for TMB co-primary analysisc Co-primary endpoints: Nivolumab + ipilimumab vs chemotherapy • OS in PD-L 1–selected populations • PFS in TMB-selected populations Hellman M et al, AACR 2018 pemetrexed + cisplatin or carboplatin, Q 3 W for ≤ 4 cycles, with optional pemetrexed maintenance following chemotherapy or nivolumab + pemetrexed maintenance following nivolumab + chemotherapy; SQ: gemcitabine + cisplatin, or gemcitabine + carboplatin, Q 3 W for ≤ 4 cycles; c. The TMB co-primary analysis was conducted in the subset of patients randomized to nivolumab + ipilimumab or chemotherapy who had evaluable TMB ≥ 10 mut/Mb

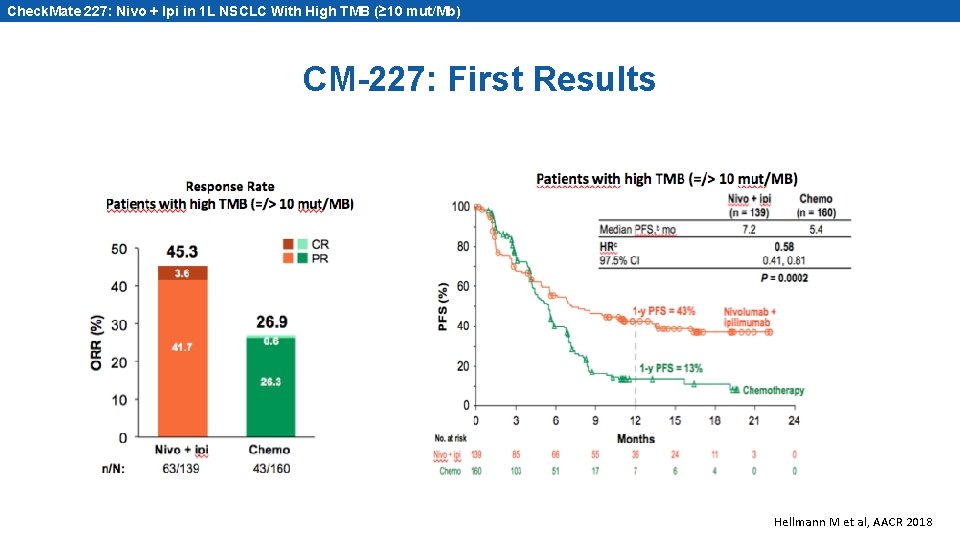
Check. Mate 227: Nivo + Ipi in 1 L NSCLC With High TMB (≥ 10 mut/Mb) CM-227: First Results Hellmann M et al, AACR 2018

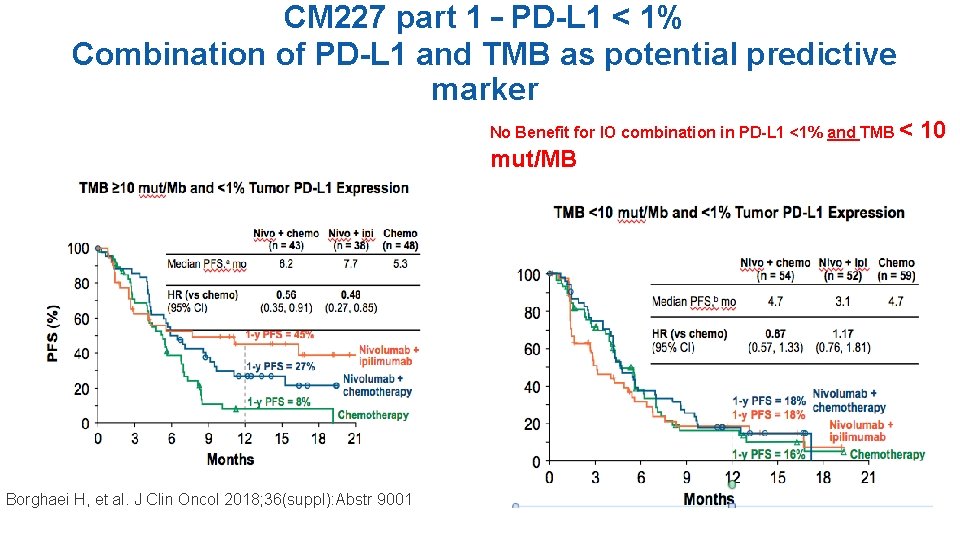
CM 227 part 1 – PD-L 1 < 1% Combination of PD-L 1 and TMB as potential predictive marker No Benefit for IO combination in PD-L 1 <1% and TMB < mut/MB Borghaei H, et al. J Clin Oncol 2018; 36(suppl): Abstr 9001 10

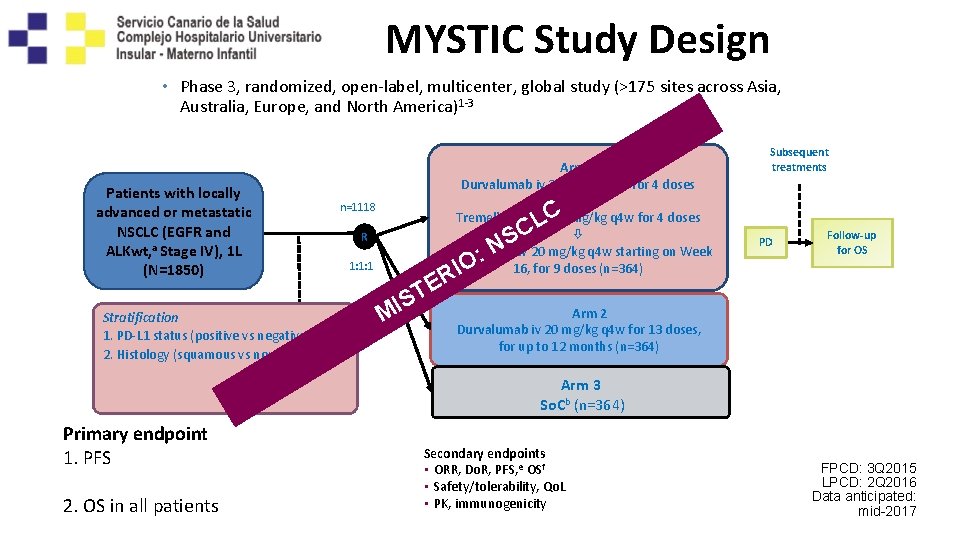
MYSTIC Study Design • Phase 3, randomized, open-label, multicenter, global study (>175 sites across Asia, Australia, Europe, and North America)1 -3 Patients with locally advanced or metastatic NSCLC (EGFR and ALKwt, a Stage IV), 1 L (N=1850) Stratification 1. PD-L 1 status (positive vs negative) 2. Histology (squamous vs nonsquamous) Arm 1 Durvalumab iv 20 mg/kg q 4 w for 4 doses + Tremelimumab iv 1 mg/kg q 4 w for 4 doses Durvalumab iv 20 mg/kg q 4 w starting on Week 16, for 9 doses (n=364) n=1118 R : 1: 1: 1 M E T IS O RI C L C NS Subsequent treatments PD Follow-up for OS Arm 2 Durvalumab iv 20 mg/kg q 4 w for 13 doses, for up to 12 months (n=364) Arm 3 So. Cb (n=364) Primary endpoint 1. PFS in all patients and in PD-L 1(+) patients 2. OS in all patients Secondary endpoints • ORR, Do. R, PFS, e OSf • Safety/tolerability, Qo. L • PK, immunogenicity FPCD: 3 Q 2015 LPCD: 2 Q 2016 Data anticipated: mid-2017

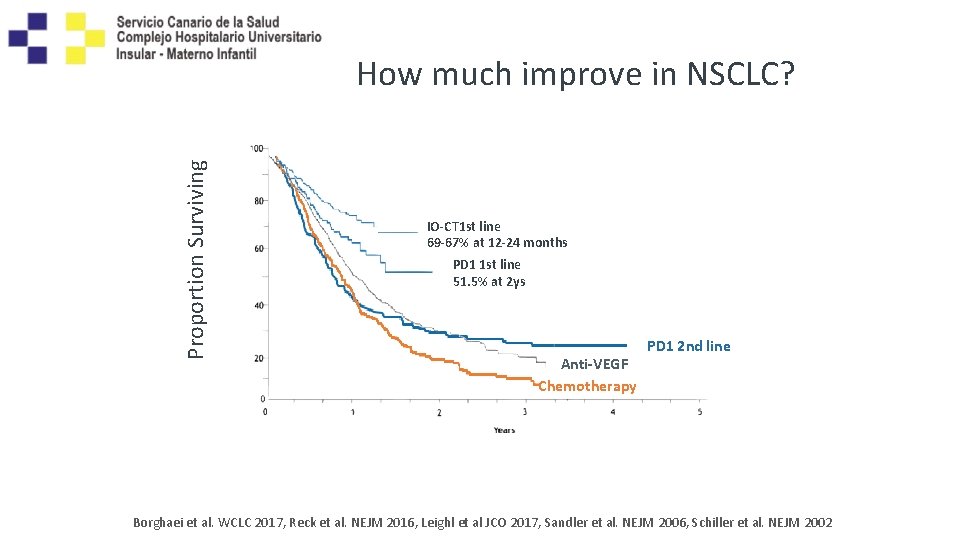
Proportion Surviving How much improve in NSCLC? IO-CT 1 st line 69 -67% at 12 -24 months PD 1 1 st line 51. 5% at 2 ys Anti-VEGF Chemotherapy PD 1 2 nd line Borghaei et al. WCLC 2017, Reck et al. NEJM 2016, Leighl et al JCO 2017, Sandler et al. NEJM 2006, Schiller et al. NEJM 2002

Some take home messages • PEMBROLIZUMAB MONO STILL THE SOC in 1 L NSCLC…PD-L 1>=50% • CT plus IO is a NEW STANDARD OF CARE IN 1 L nonsquamous NSCLC, in all PD-L 1 expression and WILL be a new standard in squamous soon!! • Combos have worse tolerability but no new ir. AEs • We STILL needing betters Biomarkers • Patients access to this NEW OPTIONS of therapy must be our goal!!!

THANK YOU!! Dr. Delvys Rodríguez Abreu Servicio Oncología Médica Hospital Universitario Insular de Gran Canaria drodabr@gobiernodecanarias. org