Nucleophilic Substitution of Alkyl Halides Part 2 Observations

Nucleophilic Substitution of Alkyl Halides (Part 2)

Observations in Substitution Reactions n Single enantiomer of a starting material resulted in two situations (generally) n n n Rates showed two opposing trends n n n Single enantiomer of product (optically active) Racemic products (optically inactive) Primary halides reacted rapidly and tertiary halides reacted slowly Primary halides reacted slowly and tertiary halides reacted rapidly Kinetics showed two situations n n First order dependence on the nucleophile No concentration dependence on the nucleophile
![SN 2 Mechanism n Bimolecular reaction n Rate = k [alkyl halide]1 [nucleophile]1 Inversion SN 2 Mechanism n Bimolecular reaction n Rate = k [alkyl halide]1 [nucleophile]1 Inversion](http://slidetodoc.com/presentation_image_h2/7a19d09cd550885149ab0a63739a4397/image-3.jpg)
SN 2 Mechanism n Bimolecular reaction n Rate = k [alkyl halide]1 [nucleophile]1 Inversion of chirality at carbon Reactivity of alkyl halides: 1° > 2° > 3°
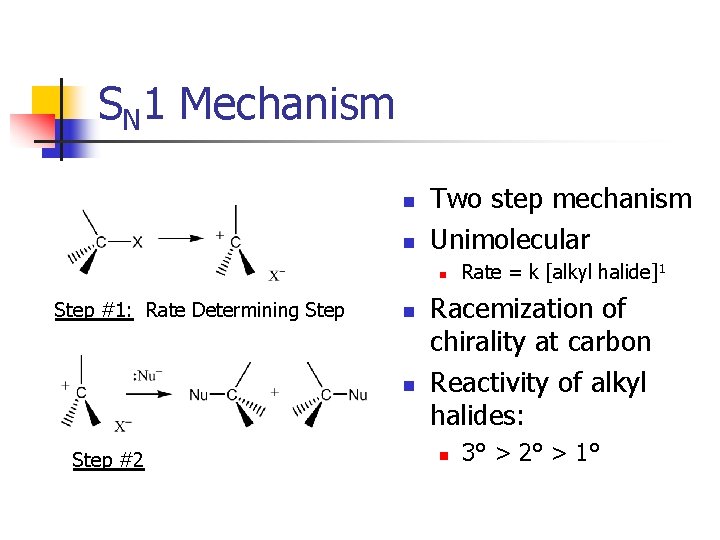
SN 1 Mechanism n n Two step mechanism Unimolecular n Step #1: Rate Determining Step n n Step #2 Rate = k [alkyl halide]1 Racemization of chirality at carbon Reactivity of alkyl halides: n 3° > 2° > 1°
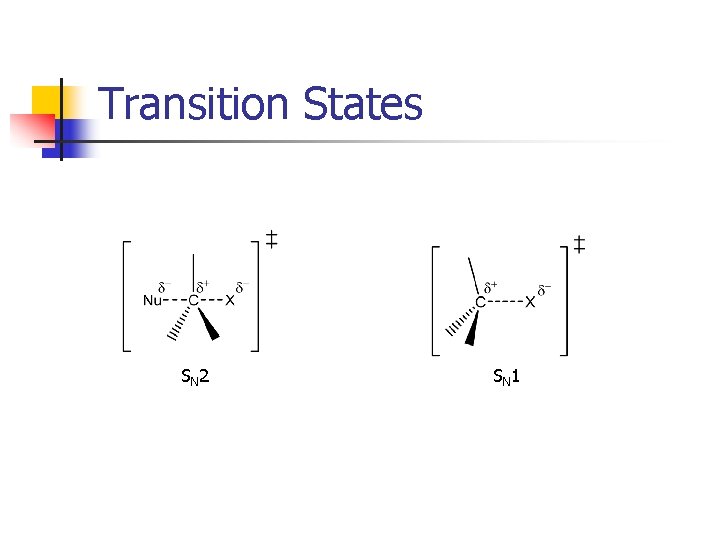
Transition States SN 2 SN 1

Alkyl Halide (Substrate) n Steric effects: carbon is being attacked by the nucleophile in an SN 2 mechanism n n n Steric strain experienced by nucleophile increases in the order: 1° < 2° << 3° So reactivity is CH 3 X > 1° > 2° Carbocation: intermediate in the SN 1 mechanism n Electron-donation by attached carbons stabilizes carbocation intermediates: 3° > 2° > 1°
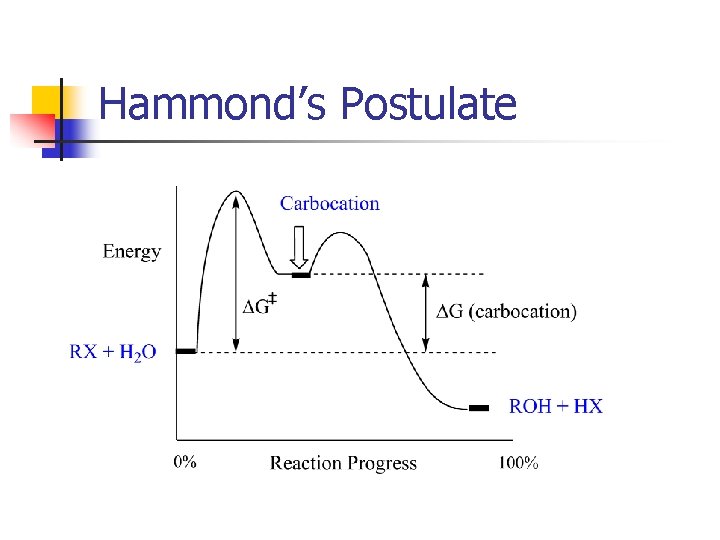
Hammond’s Postulate
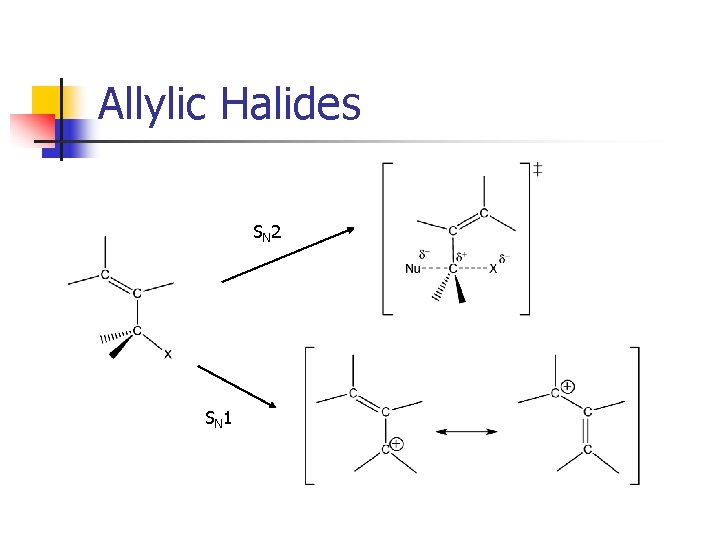
Allylic Halides SN 2 SN 1
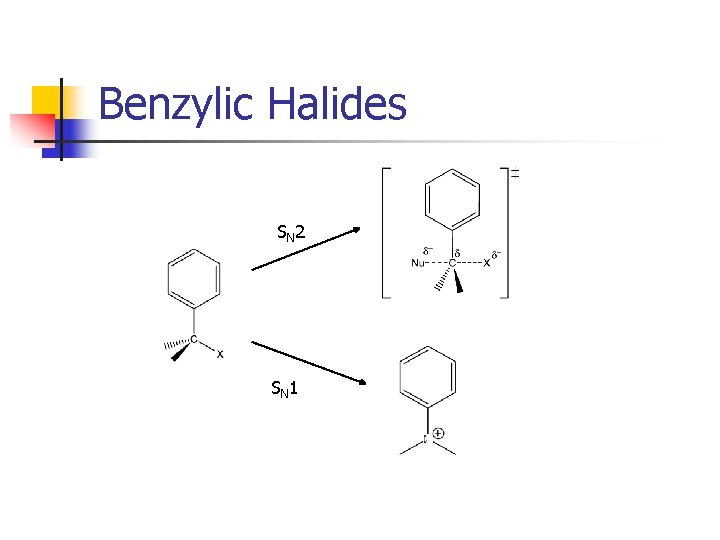
Benzylic Halides SN 2 SN 1
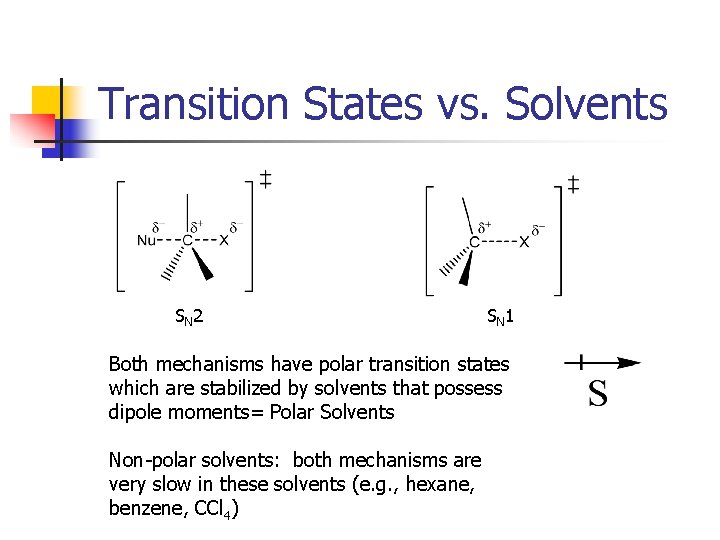
Transition States vs. Solvents SN 2 SN 1 Both mechanisms have polar transition states which are stabilized by solvents that possess dipole moments= Polar Solvents Non-polar solvents: both mechanisms are very slow in these solvents (e. g. , hexane, benzene, CCl 4)

Best Solvents for SN 2
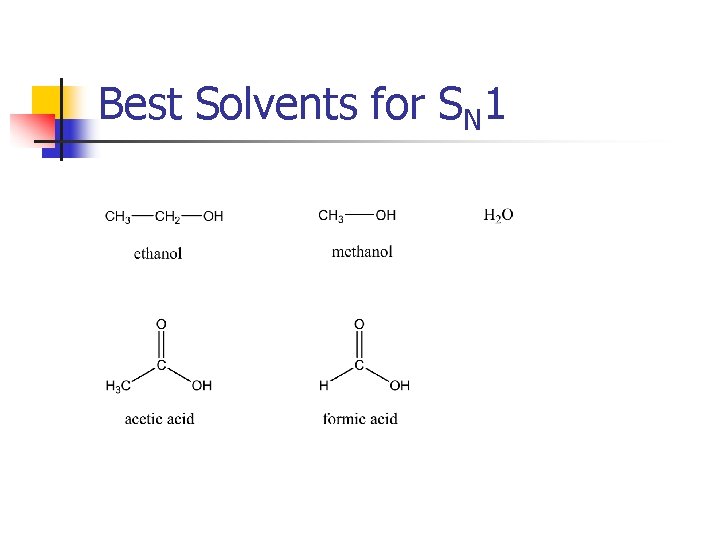
Best Solvents for SN 1

Nucleophile n Concentration and reactivity of the nucleophile important in SN 2 n n n Good electon-pair donors but weak bases Anions >> neutral nucleophiles Nucleophile is not involved in the rate determining step of the SN 1 mechanism n n Poor electron-pair donors often favor SN 1 Conjugate acids of nucleophiles especially as solvents
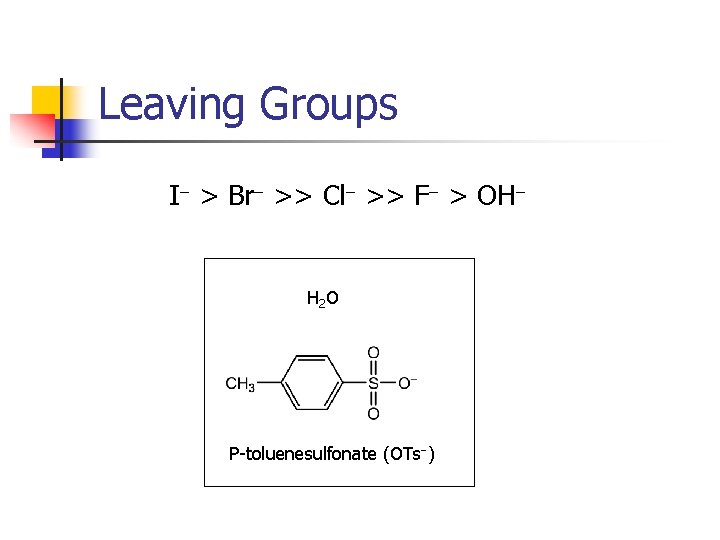
Leaving Groups I- > Br- >> Cl- >> F- > OH- H 2 O P-toluenesulfonate (OTs-)
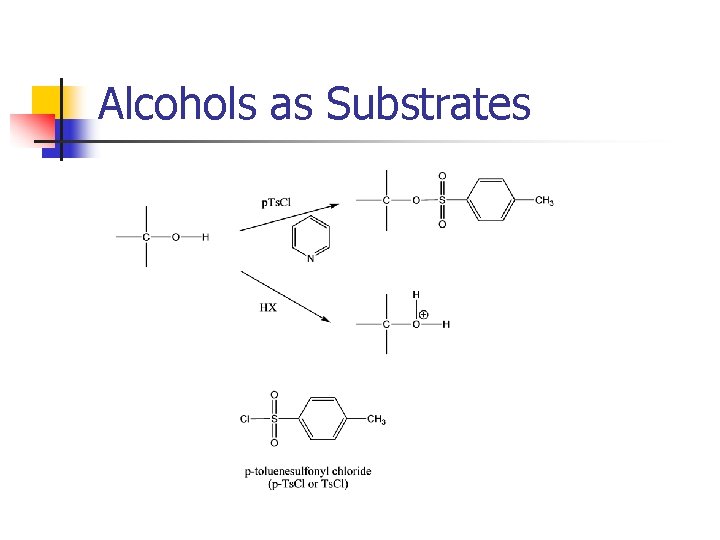
Alcohols as Substrates
- Slides: 15