NUCLEIC ACIDS DNADeoxyribonucleic acid DNA RNA Ribonucleic acid

NUCLEIC ACIDS DNA(Deoxyribonucleic acid) DNA RNA (Ribonucleic acid)

Brief History of Nucleotides and Nucleic acids Ø Ø Ø 1869 – Johannes Friedrich Miescher isolated “nuclein” from soiled bandages. 1902 – Archibald Garrod studied “Alkaptonuria”; he also concluded that specific gene is associated with absence of specific enzyme. 1903 – Walter Sutton, introduced the chromosome structure. 1913 – Thomas Hunt Morgan “gene mapping” 1926 – James Sumner purified urease and he also identified enzymes to be proteins.

Brief History of Nucleotides and Nucleic acids Ø Ø 1928 –Frederick Griffith “Transforming Principle” a chemical transferred from dead bacteria to living cells caused genetically converted strains. 1944 – Oswald Avery, Maclyn Mc. Carty, and Colin Mac. Leod – identified Griffith’s “transformation principle” as DNA. 1947 - Erwin Chargaff – studied the base pairing 1950’s – Rosalind Franklin “X-ray of DNA”

Brief History of Nucleotides and Nucleic acids n n n 1953: James Watsons and Francis Crick – “DNA double helix”. "Molecular structure of Nucleic Acids: A Structure for Deoxyribose Nucleic Acid" An article published in Nature (April 25, 1953). It was the first publication which described the discovery of the double helix structure of DNA.
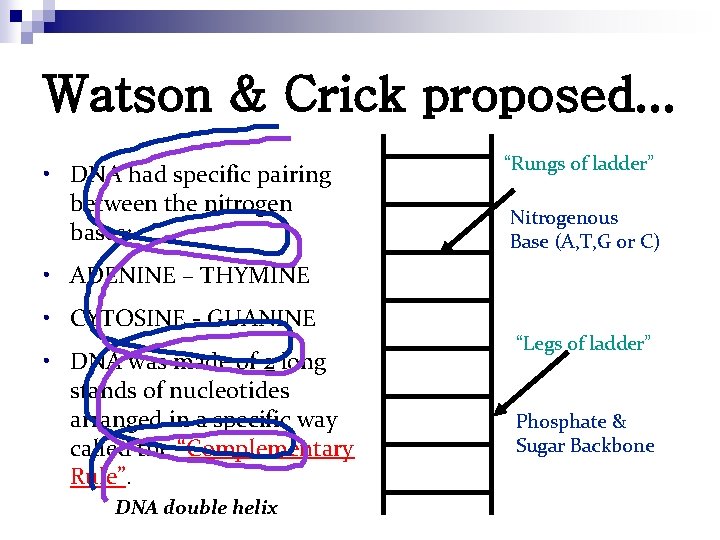
Watson & Crick proposed. . . • DNA had specific pairing between the nitrogen bases: “Rungs of ladder” Nitrogenous Base (A, T, G or C) • ADENINE – THYMINE • CYTOSINE - GUANINE • DNA was made of 2 long stands of nucleotides arranged in a specific way called the “Complementary Rule”. DNA double helix “Legs of ladder” Phosphate & Sugar Backbone
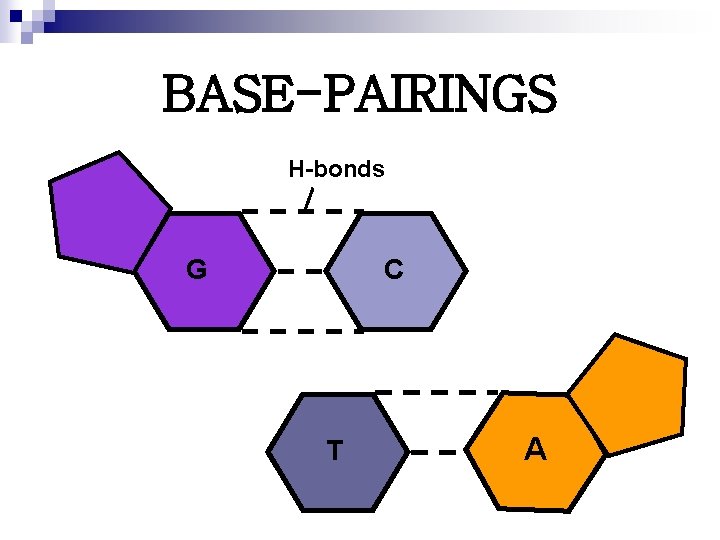
BASE-PAIRINGS H-bonds G C T A
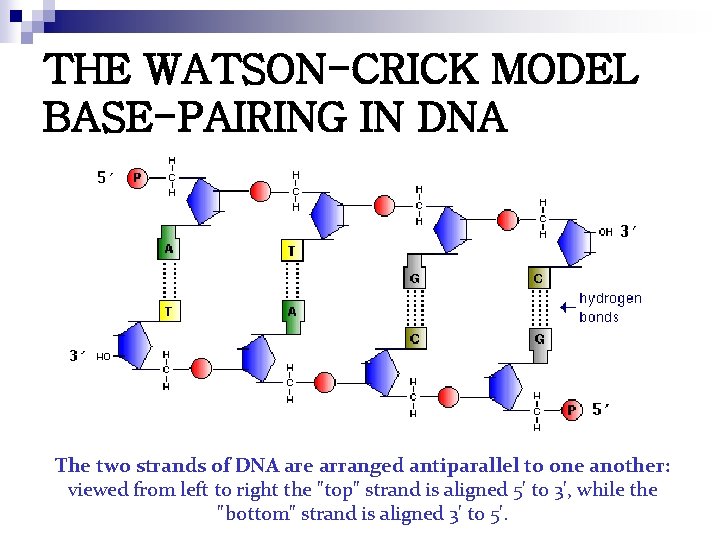
THE WATSON-CRICK MODEL BASE-PAIRING IN DNA The two strands of DNA are arranged antiparallel to one another: viewed from left to right the "top" strand is aligned 5' to 3', while the "bottom" strand is aligned 3' to 5'.
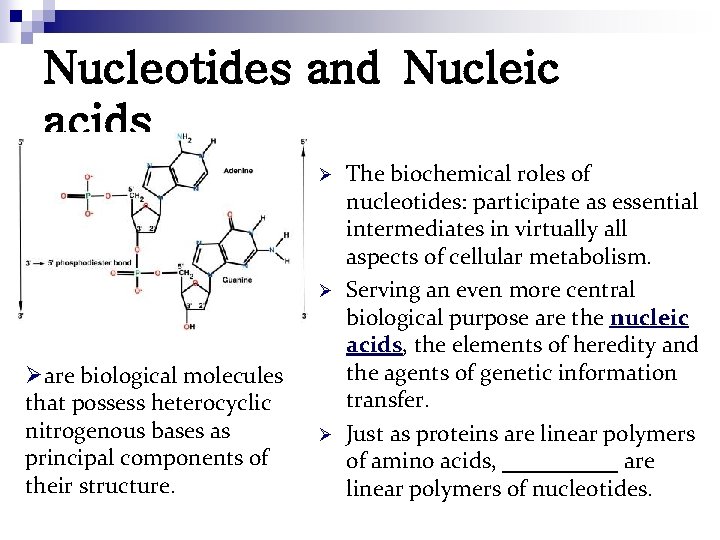
Nucleotides and Nucleic acids Ø Ø Øare biological molecules that possess heterocyclic nitrogenous bases as principal components of their structure. Ø The biochemical roles of nucleotides: participate as essential intermediates in virtually all aspects of cellular metabolism. Serving an even more central biological purpose are the nucleic acids, the elements of heredity and the agents of genetic information transfer. Just as proteins are linear polymers of amino acids, _____ are linear polymers of nucleotides.
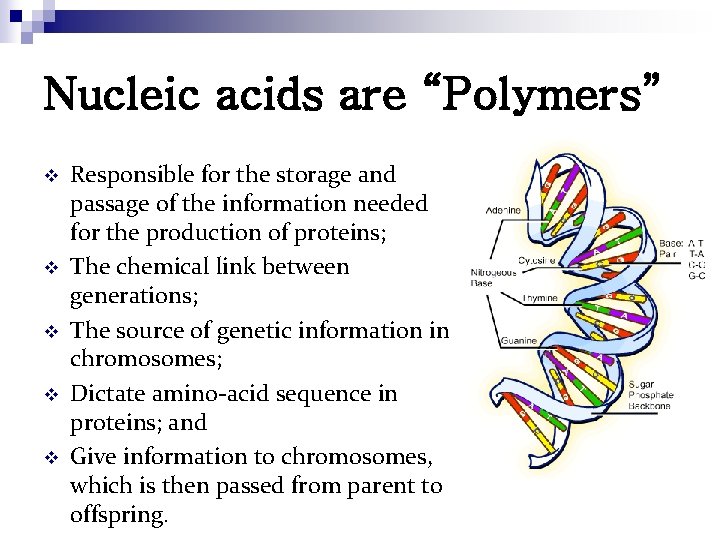
Nucleic acids are “Polymers” v v v Responsible for the storage and passage of the information needed for the production of proteins; The chemical link between generations; The source of genetic information in chromosomes; Dictate amino-acid sequence in proteins; and Give information to chromosomes, which is then passed from parent to offspring.
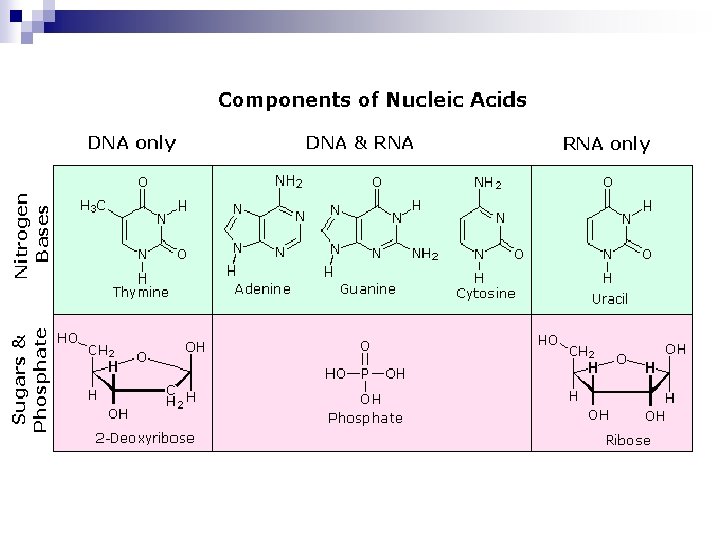

Nitrogenous Bases ü ü ü The bases of nucleotides and nucleic acids are derivatives of either pyrimidine or purine. Q: Look at the structure of pyrimidine, what can you observe? A: 6 member heterocyclic ring, with 2 Nitrogen atoms and it is number clockwise. Q: Look at the structure of purine, what can you observe? A: It is 9 member ring structure of pyrimidine fused with an imidazole ring.

Common Pyrimidines and Purines n n n Q: What are the common pyrimidine bases? A: Cytosine, Uracil and Thymine Q: What is the name of the first 1. pyrimidine base? What is its chemical name? Q: What is the name of the second pyrimidine base? What is its chemical name? Q: What is the name of the third pyrimidine base? What is its chemical name? 2. 3.

Common Pyrimidines and Purines n n Q: What are the common purine bases? A: Adenine and Guanine Q: What is the name of the first purine base? What is its chemical name? Q: What is the name of the first purine base? What is 1. its chemical name? 2.
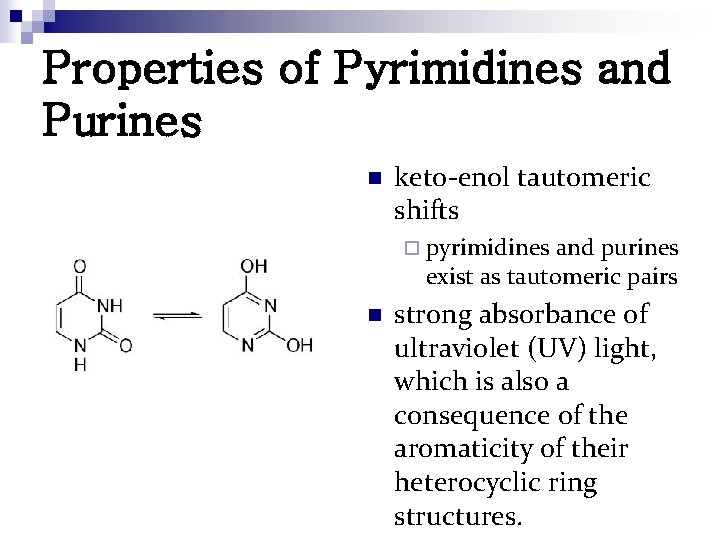
Properties of Pyrimidines and Purines n keto-enol tautomeric shifts ¨ pyrimidines and purines exist as tautomeric pairs n strong absorbance of ultraviolet (UV) light, which is also a consequence of the aromaticity of their heterocyclic ring structures.
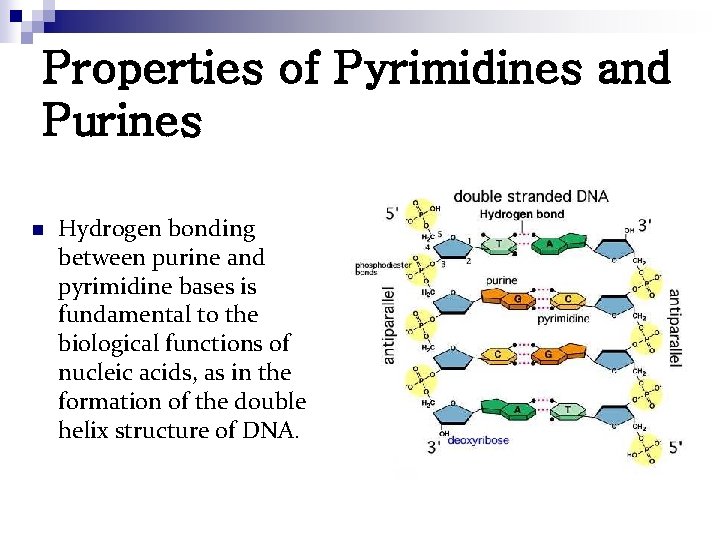
Properties of Pyrimidines and Purines n Hydrogen bonding between purine and pyrimidine bases is fundamental to the biological functions of nucleic acids, as in the formation of the double helix structure of DNA.

Properties of Pyrimidines and Purines n The important functional groups participating in H-bond formation: ¨ amino groups of adenine, cytosine and guanine; ¨ the ring nitrogens at position 3 of pyrimidines and position 1 of purines; and ¨ the strongly electronegative oxygen atoms attached at position 4 of uracil and thymine, position 2 of cytosine, and position 6 of guanine
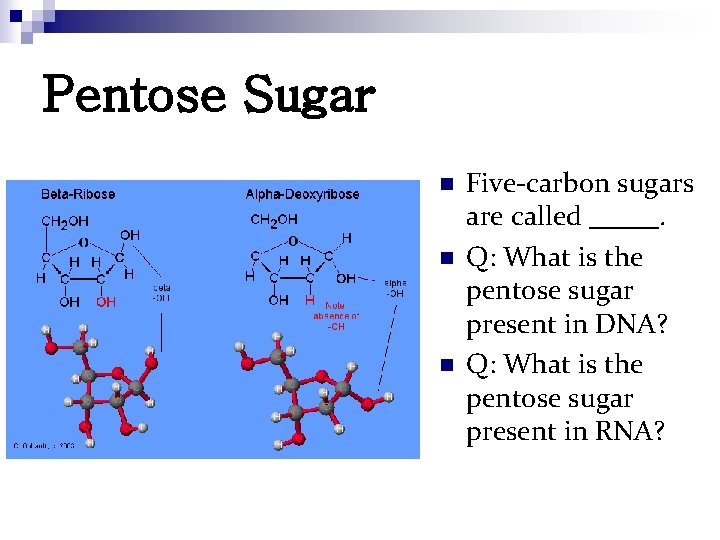
Pentose Sugar n n n Five-carbon sugars are called _____. Q: What is the pentose sugar present in DNA? Q: What is the pentose sugar present in RNA?
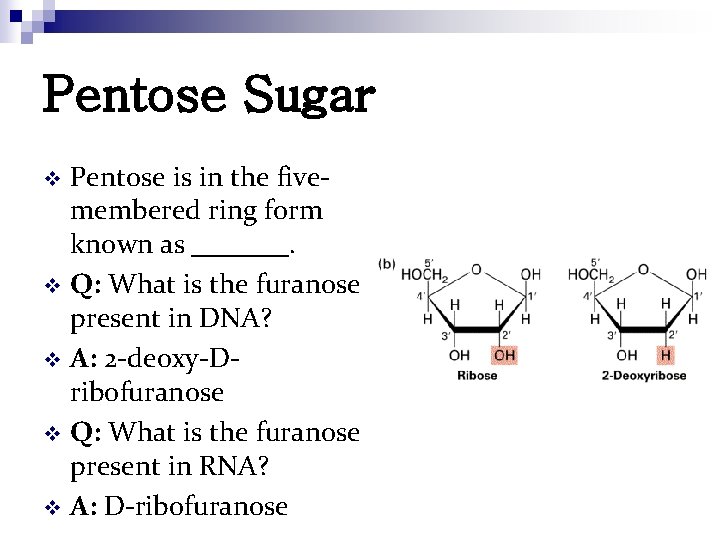
Pentose Sugar Pentose is in the fivemembered ring form known as _______. v Q: What is the furanose present in DNA? v A: 2 -deoxy-Dribofuranose v Q: What is the furanose present in RNA? v A: D-ribofuranose v
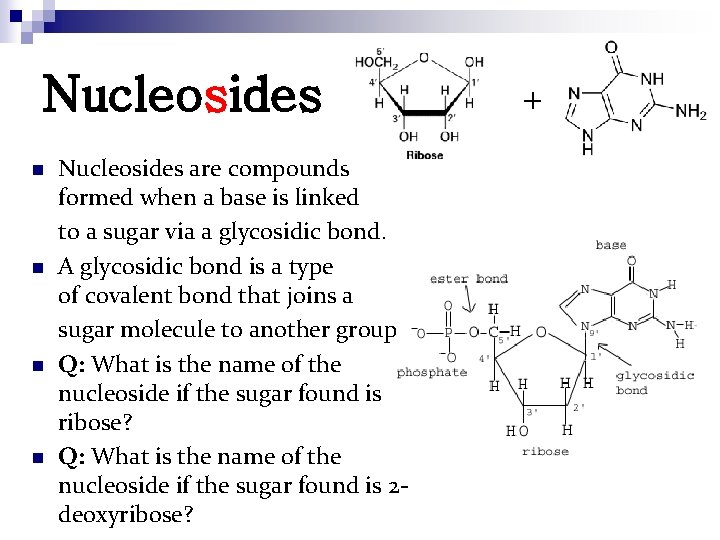
Nucleosides n n Nucleosides are compounds formed when a base is linked to a sugar via a glycosidic bond. A glycosidic bond is a type of covalent bond that joins a sugar molecule to another group. Q: What is the name of the nucleoside if the sugar found is ribose? Q: What is the name of the nucleoside if the sugar found is 2 deoxyribose? +
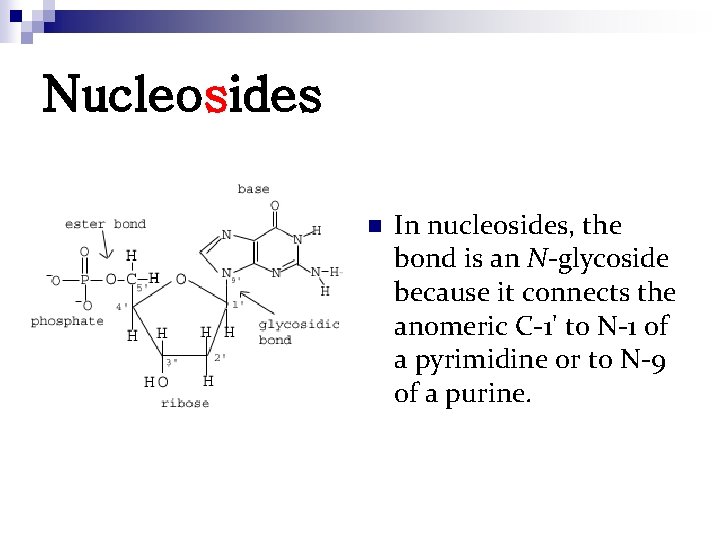
Nucleosides n In nucleosides, the bond is an N-glycoside because it connects the anomeric C-1' to N-1 of a pyrimidine or to N-9 of a purine.

Nucleoside Nomenclature n Nucleosides are named by adding the ending -idine to the root name of a pyrimidine or -osine to the root name of a purine. Nitrogenous base Cytosine + sugar Guanine + sugar Adenine + sugar Uracil + sugar Thymine + sugar Nucleoside name Cytidine Guanosine Adenosine Uridine Thymidine

Nucleoside Conformation n n syn and anti rotation of the base about the glycosidic bond is sterically hindered, principally by the hydrogen atom on the C-2' carbon of the furanose Pyrimidine nucleosides favor the anti conformation Purine nucleosides can adopt either the syn or anti conformation.

Nucleosides Are More Water -Soluble Than Free Bases n n n Nucleosides are much more water-soluble than the free bases because of the hydrophilicity of the sugar moiety. Nucleosides are relatively stable in alkali. Pyrimidine nucleosides are also resistant to acid hydrolysis, but purine nucleosides are easily hydrolyzed in acid to yield the free base and pentose.
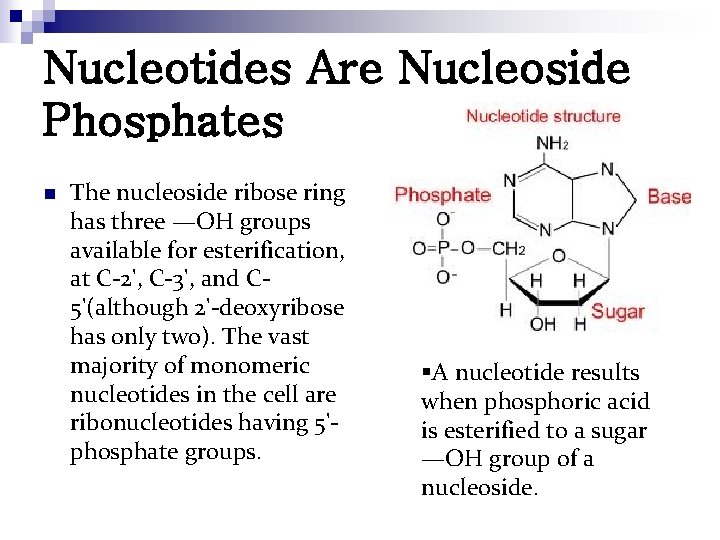
Nucleotides Are Nucleoside Phosphates n The nucleoside ribose ring has three —OH groups available for esterification, at C-2', C-3', and C 5'(although 2'-deoxyribose has only two). The vast majority of monomeric nucleotides in the cell are ribonucleotides having 5'phosphate groups. §A nucleotide results when phosphoric acid is esterified to a sugar —OH group of a nucleoside.
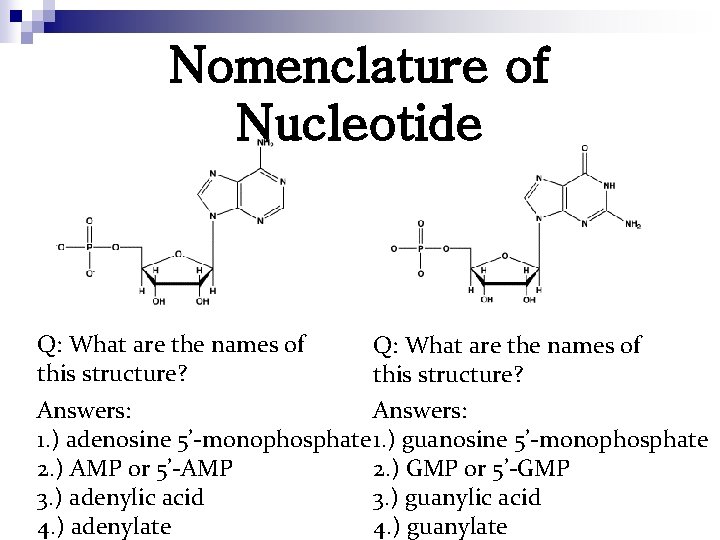
Nomenclature of Nucleotide Q: What are the names of this structure? Answers: 1. ) adenosine 5’-monophosphate 1. ) guanosine 5’-monophosphate 2. ) AMP or 5’-AMP 2. ) GMP or 5’-GMP 3. ) adenylic acid 3. ) guanylic acid 4. ) adenylate 4. ) guanylate
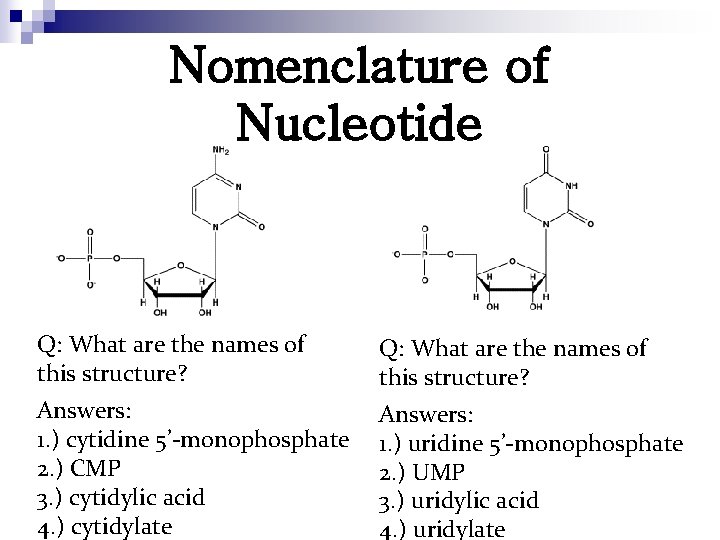
Nomenclature of Nucleotide Q: What are the names of this structure? Answers: 1. ) cytidine 5’-monophosphate 2. ) CMP 3. ) cytidylic acid 4. ) cytidylate Answers: 1. ) uridine 5’-monophosphate 2. ) UMP 3. ) uridylic acid 4. ) uridylate
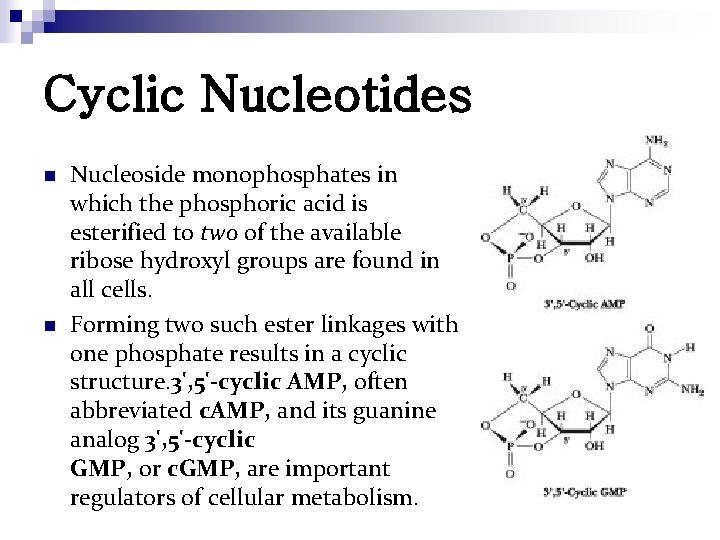
Cyclic Nucleotides n n Nucleoside monophosphates in which the phosphoric acid is esterified to two of the available ribose hydroxyl groups are found in all cells. Forming two such ester linkages with one phosphate results in a cyclic structure. 3', 5'-cyclic AMP, often abbreviated c. AMP, and its guanine analog 3', 5'-cyclic GMP, or c. GMP, are important regulators of cellular metabolism.
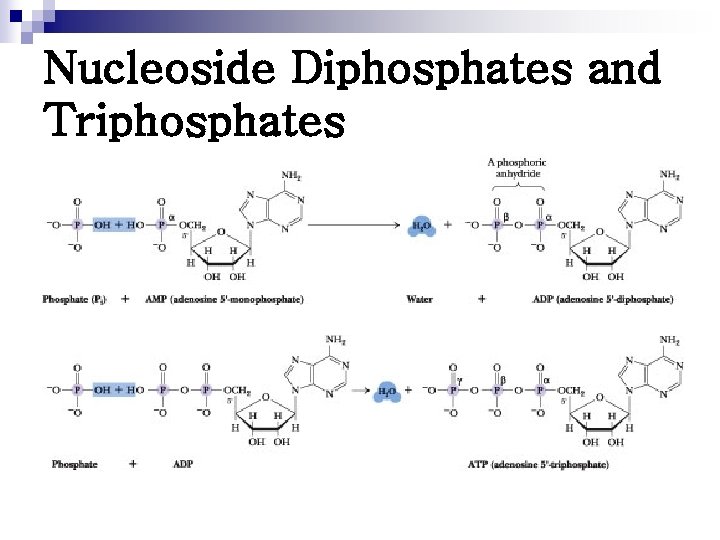
Nucleoside Diphosphates and Triphosphates

Nucleoside 5'-Triphosphates Are Carriers of Chemical Energy n n ATP has been termed the energy currency of the cell. GTP is the major energy source for protein synthesis. CTP is an essential metabolite in phospholipid synthesis UTP forms activated intermediates with sugars that go on to serve as substrates in the biosynthesis of complex carbohydrates and polysaccharides.
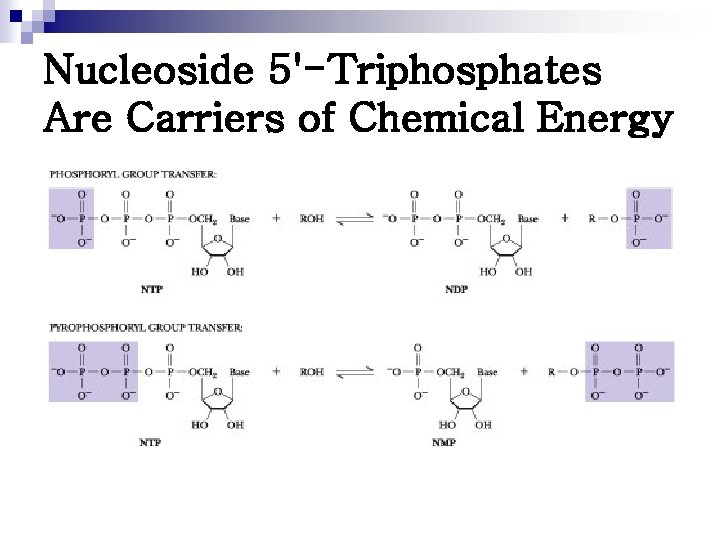
Nucleoside 5'-Triphosphates Are Carriers of Chemical Energy

Nucleoside 5'-Triphosphates Are Carriers of Chemical Energy The evolution of metabolism has led to the dedication of one of these four NTPs to each of the major branches of metabolism. n To complete the picture, the four NTPs and their d. NTP counterparts are the substrates for the synthesis of the remaining great class of biomolecules—the nucleic acids. n

The Bases of Nucleotides Serve as “Information Symbols” n n ATP: serve as the primary nucleotide in central pathways of energy metabolism, GTP: is used to drive protein synthesis. Various nucleotides are channeled in appropriate metabolic directions through specific recognition of the base of the nucleotide. This role as information symbols extends to nucleotide polymers, the nucleic acids, where the bases serve as the information symbols for the code of genetic information.
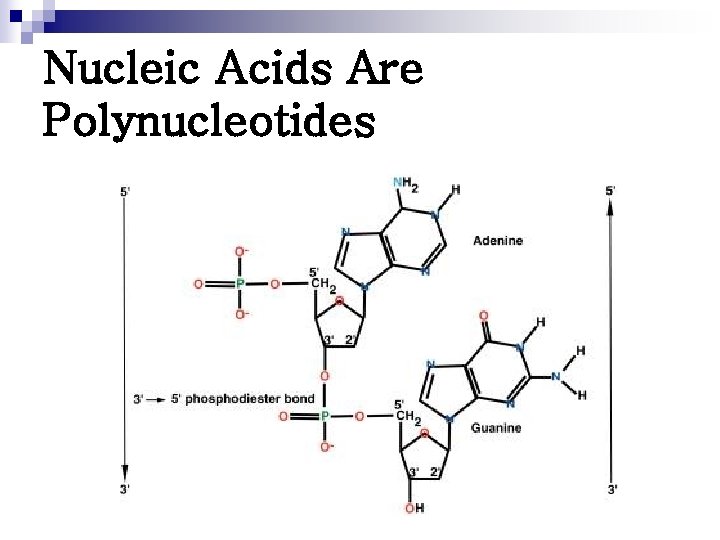
Nucleic Acids Are Polynucleotides
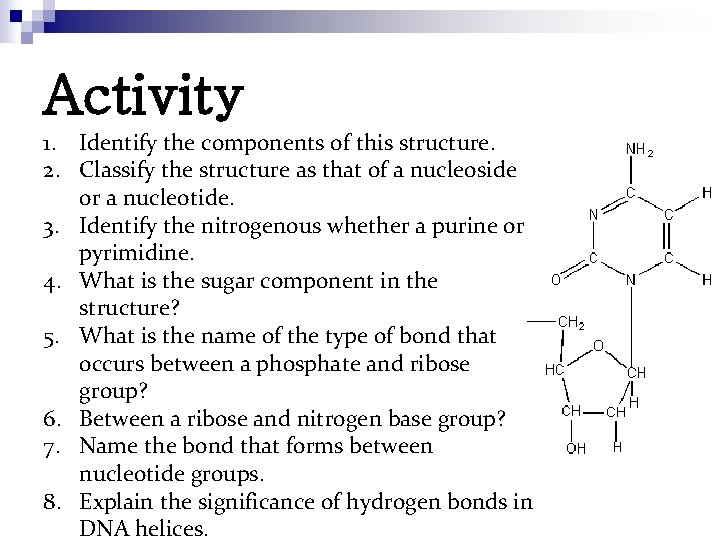
Activity 1. Identify the components of this structure. 2. Classify the structure as that of a nucleoside or a nucleotide. 3. Identify the nitrogenous whether a purine or pyrimidine. 4. What is the sugar component in the structure? 5. What is the name of the type of bond that occurs between a phosphate and ribose group? 6. Between a ribose and nitrogen base group? 7. Name the bond that forms between nucleotide groups. 8. Explain the significance of hydrogen bonds in DNA helices.
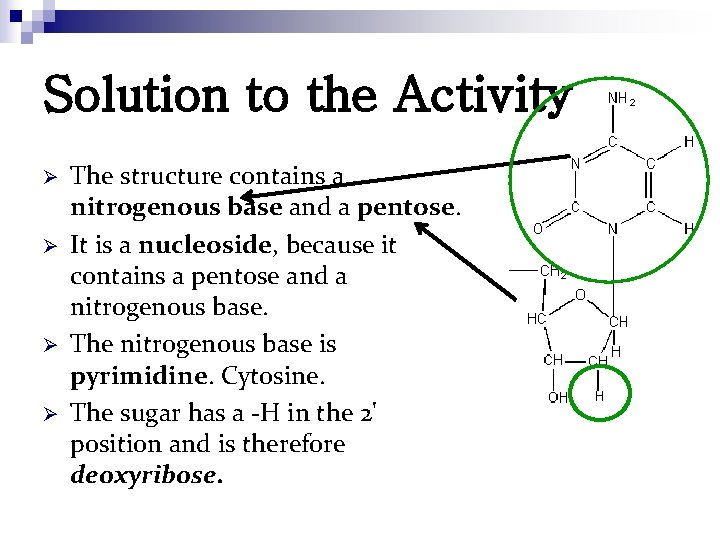
Solution to the Activity Ø Ø The structure contains a nitrogenous base and a pentose. It is a nucleoside, because it contains a pentose and a nitrogenous base. The nitrogenous base is pyrimidine. Cytosine. The sugar has a -H in the 2' position and is therefore deoxyribose.

Solution to the Activity n n An ester bond forms between a phosphate group and a ribose group. A glycosidic bond forms between a ribose group and a nitrogen base group A phosphodiester bond forms between nucleotides to form nucleic acids. While hydrogen bonds do contribute a small amount to the stability of helices, their main contribution is to the specificity of a helix. Hydrogen bonds dictate the complementary base pairing that aligns anti-parallel nucleic acids strands in a DNA helix.
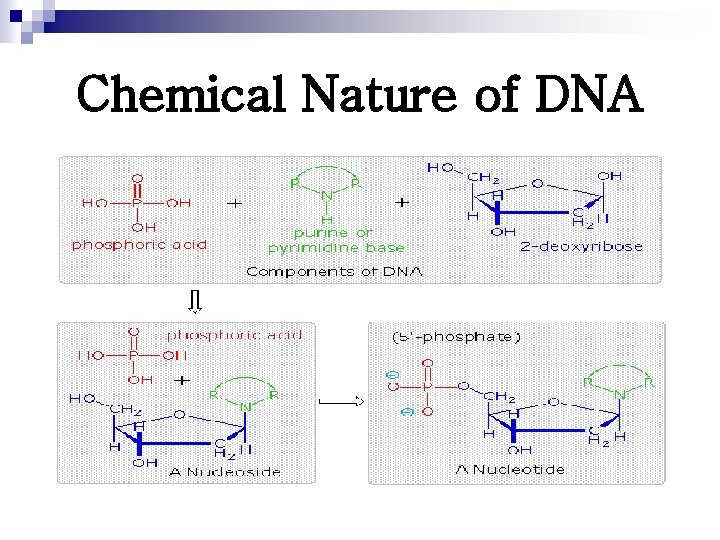
Chemical Nature of DNA
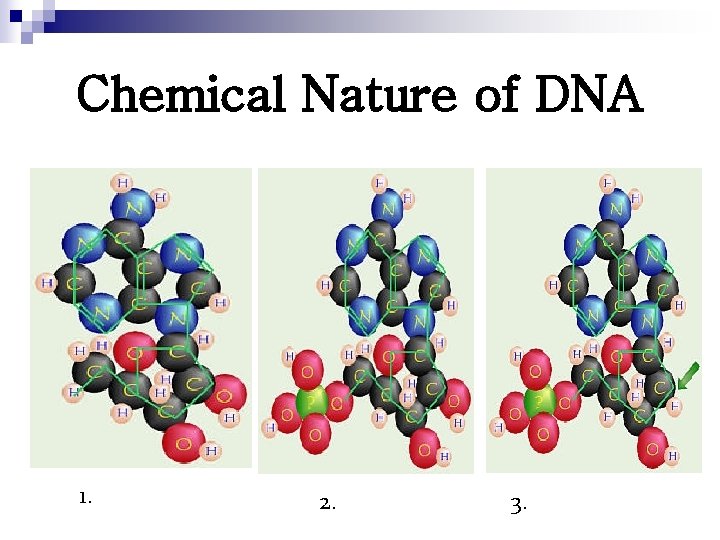
Chemical Nature of DNA 1. 2. 3.

DNA at a glance. . . n n DNA is organized into chromosomes, which are found within the nuclei of cells. A gene is a segment of DNA on a chromosome that codes for specific protein and thus determines a trait. The genetic code is determined by the order of bases in the gene, which specifies what type of proteins will be produced. Remember this! DNA contains the genetic material.
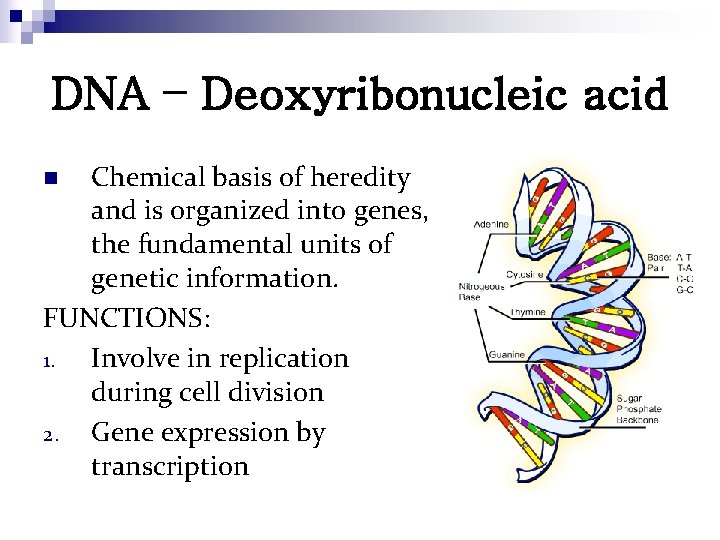
DNA – Deoxyribonucleic acid Chemical basis of heredity and is organized into genes, the fundamental units of genetic information. FUNCTIONS: 1. Involve in replication during cell division 2. Gene expression by transcription n
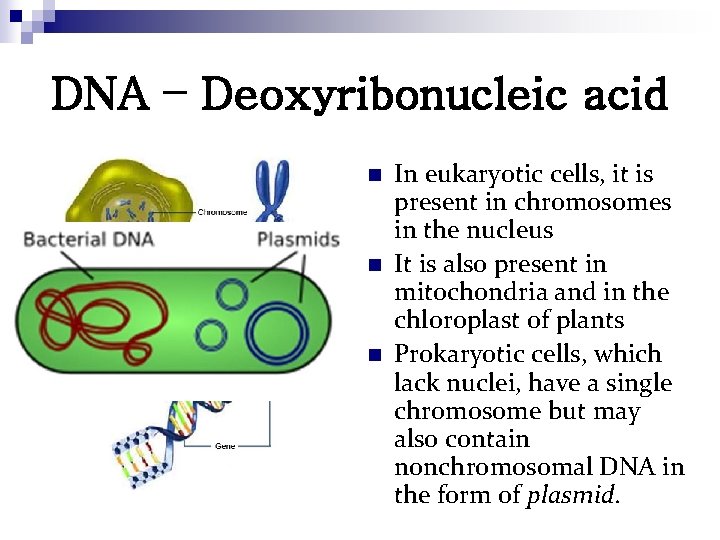
DNA – Deoxyribonucleic acid n n n In eukaryotic cells, it is present in chromosomes in the nucleus It is also present in mitochondria and in the chloroplast of plants Prokaryotic cells, which lack nuclei, have a single chromosome but may also contain nonchromosomal DNA in the form of plasmid.
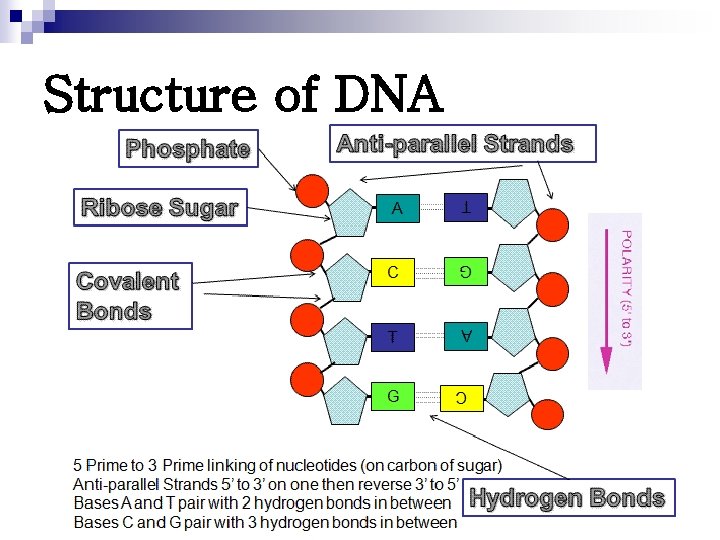
Structure of DNA

Structure of DNA Polydeoxyribonucleotide Ø Linked by 3’, 5’-phosphodiester bonds Ø Double stranded molecules Ø In eukaryotic cells: nucleoproteins Ø In prokaryotic cells: nucleoid Ø
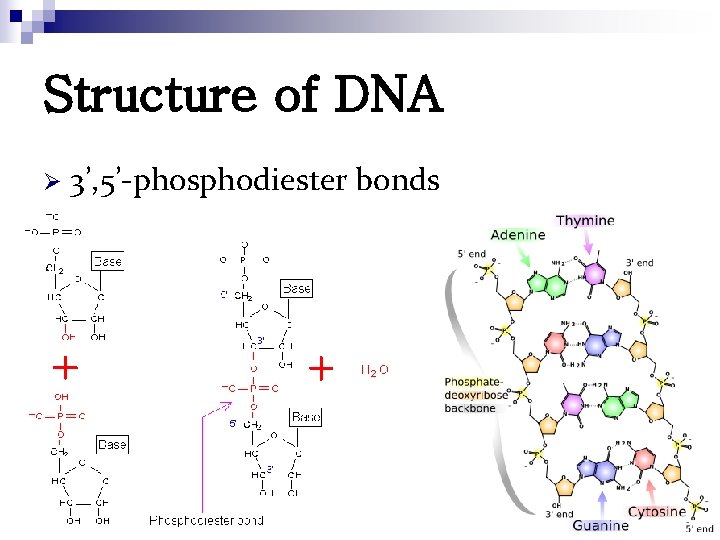
Structure of DNA Ø 3’, 5’-phosphodiester bonds
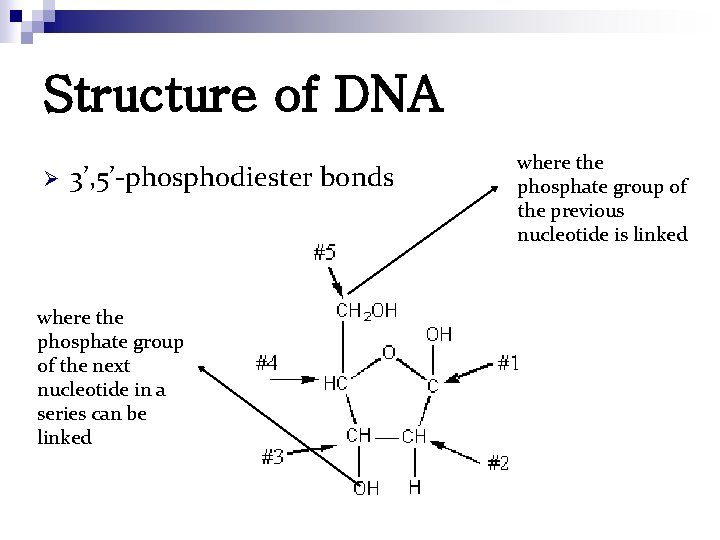
Structure of DNA Ø 3’, 5’-phosphodiester bonds where the phosphate group of the next nucleotide in a series can be linked where the phosphate group of the previous nucleotide is linked
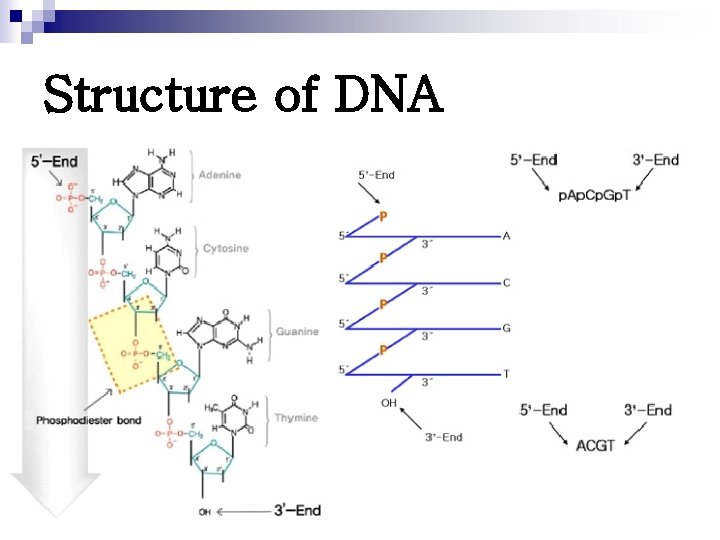
Structure of DNA
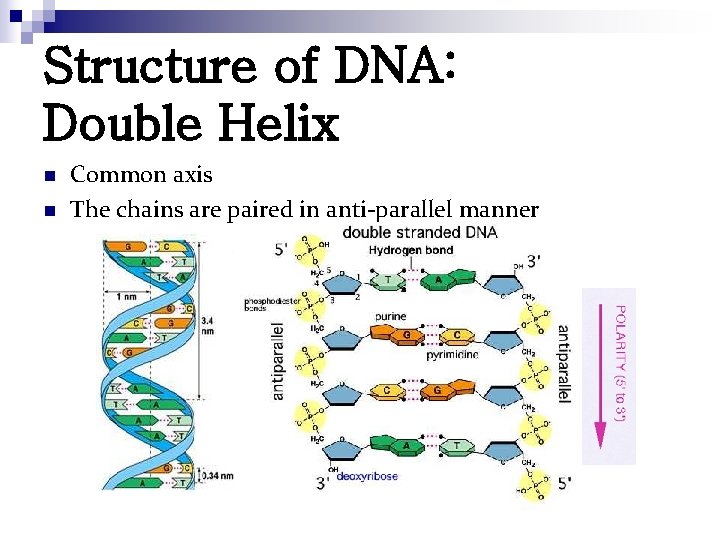
Structure of DNA: Double Helix n n Common axis The chains are paired in anti-parallel manner
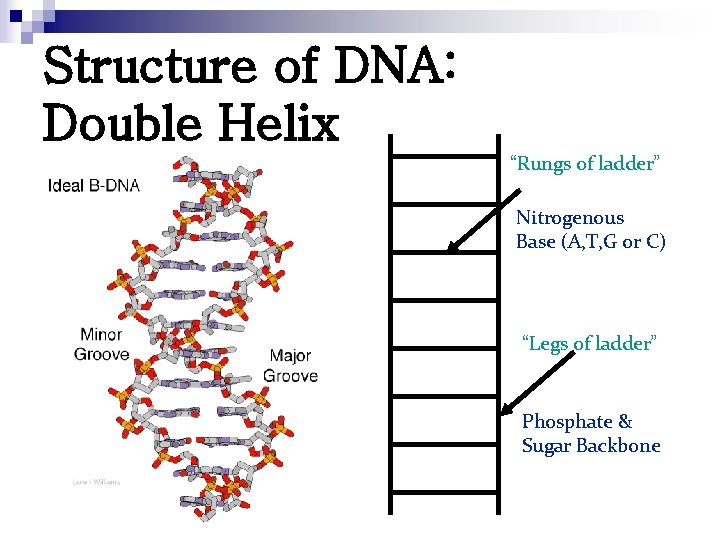
Structure of DNA: Double Helix “Rungs of ladder” Nitrogenous Base (A, T, G or C) “Legs of ladder” Phosphate & Sugar Backbone
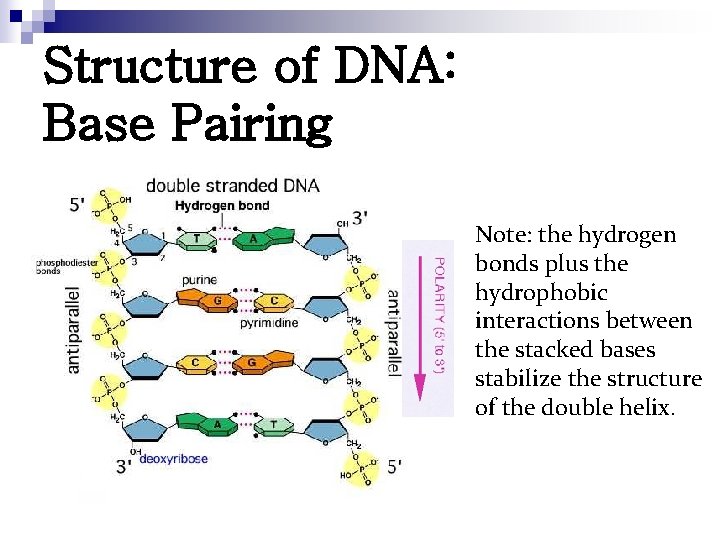
Structure of DNA: Base Pairing Note: the hydrogen bonds plus the hydrophobic interactions between the stacked bases stabilize the structure of the double helix.

Separation of the two DNA strands in the double helix ü ü ü Q: How the separation occurs? A: The 2 strands of the double helix occur when the hydrogen bonds between the paired bases are disrupted. Q: What is the mechanism of disruption? A: The p. H of the DNA solution is altered so that the nucleotide bases ionize or if the solution is heated. Note: no phosphodiester bonds are broken by such treatment.

Separation of the two DNA strands in the double helix n Remember these terms: ¨ ¨ ¨ n Melting Temperature (Tm): the temperature at which the half of the helical structure is lost. DNA denaturation: loss of helical structure of the DNA by increasing the temperature(melting) or treatment with alkali. Hyperchromic effect: DNA absorbance to UV is at 260 nm(max), it will increase if the DNA dissociate. Renaturation(Re-annealing): occurs when the complementary DNA strands can reform the double helix. Hypochromic effect: absorbance at 260 nm decreases. Note: DNA saturated with Adenine-Thymine denature at low temperature.
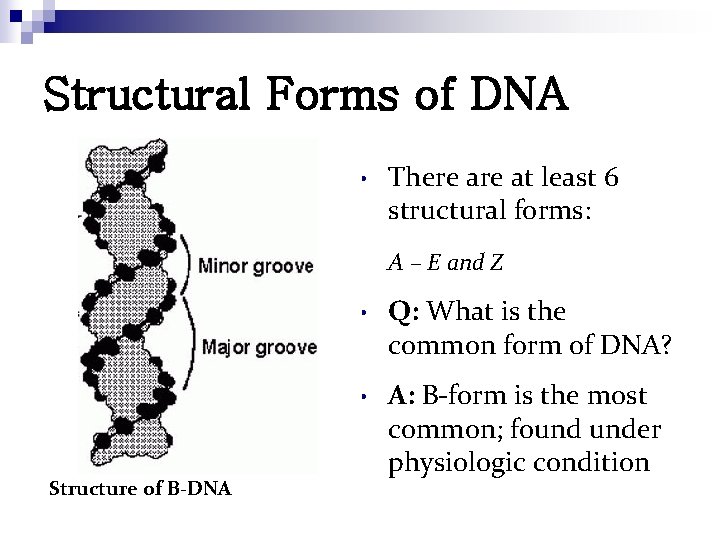
Structural Forms of DNA • There at least 6 structural forms: A – E and Z Structure of B-DNA • Q: What is the common form of DNA? • A: B-form is the most common; found under physiologic condition

Structural Forms of DNA
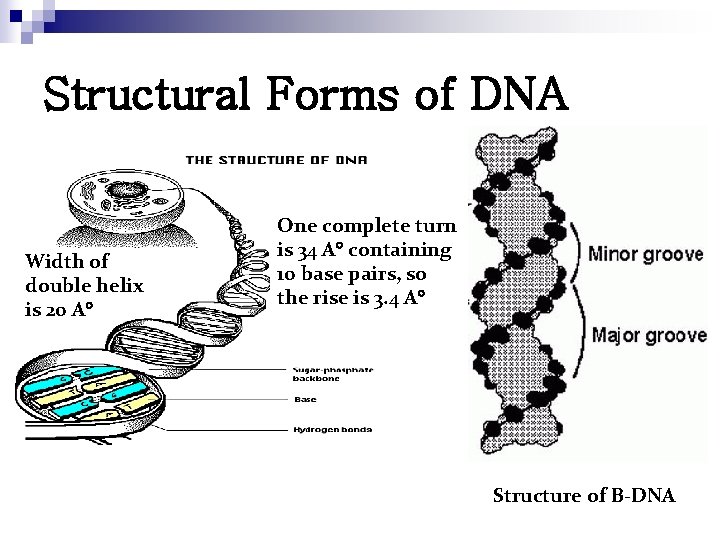
Structural Forms of DNA Width of double helix is 20 A One complete turn is 34 A containing 10 base pairs, so the rise is 3. 4 A Structure of B-DNA
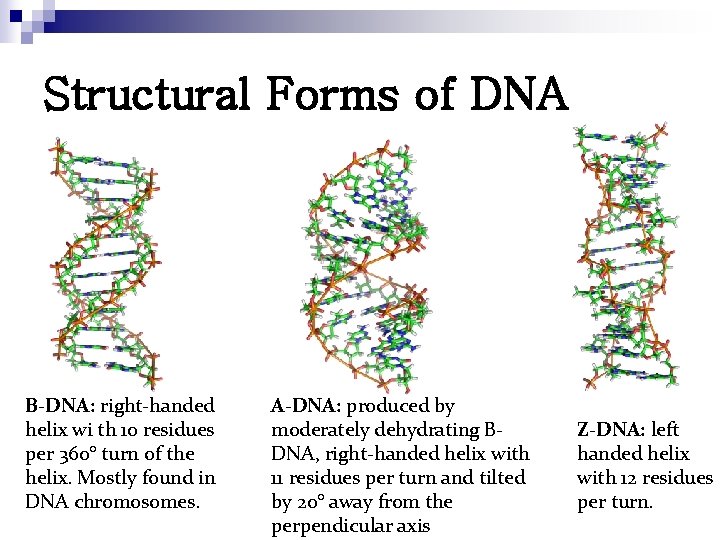
Structural Forms of DNA B-DNA: right-handed helix wi th 10 residues per 360° turn of the helix. Mostly found in DNA chromosomes. A-DNA: produced by moderately dehydrating BDNA, right-handed helix with 11 residues per turn and tilted by 20° away from the perpendicular axis Z-DNA: left handed helix with 12 residues per turn.

Modifications of DNA n n n Can be modified by translation process(after DNA synthesis). Most common: methylation of cytosine that precedes guanosines to produce methyl-cytosine. Frequency of methylation: <10% of all cytosines. Function of methylation: the presence of methylcytosines in genes is strongly correlated with transcriptionally inactive genes. Note: Methylcytosine in sequence of alternating CG doublets favors the transition of DNA from the B-form to Z-form
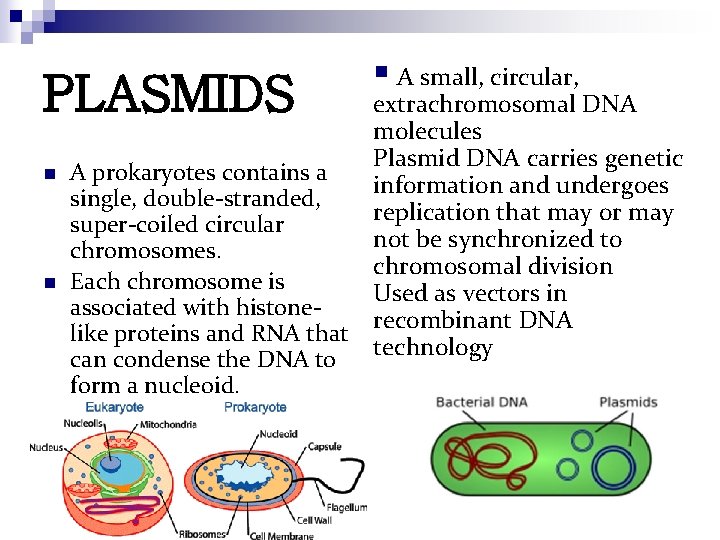
PLASMIDS n n § A small, circular, extrachromosomal DNA molecules Plasmid DNA carries genetic A prokaryotes contains a information and undergoes single, double-stranded, replication that may or may super-coiled circular not be synchronized to chromosomes. chromosomal division Each chromosome is Used as vectors in associated with histonerecombinant DNA like proteins and RNA that technology can condense the DNA to form a nucleoid.
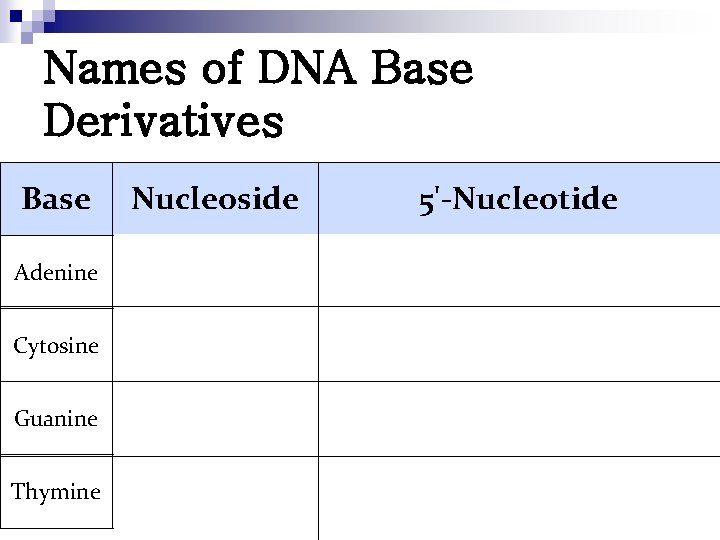
Names of DNA Base Derivatives Base Nucleoside 5'-Nucleotide Adenine 2'-Deoxyadenosine-5'-monophosphate Cytosine 2'-Deoxycytidine-5'-monophosphate Guanine 2'-Deoxyguanosine-5'-monophosphate Thymine 2'-Deoxythymidine-5'-monophosphate
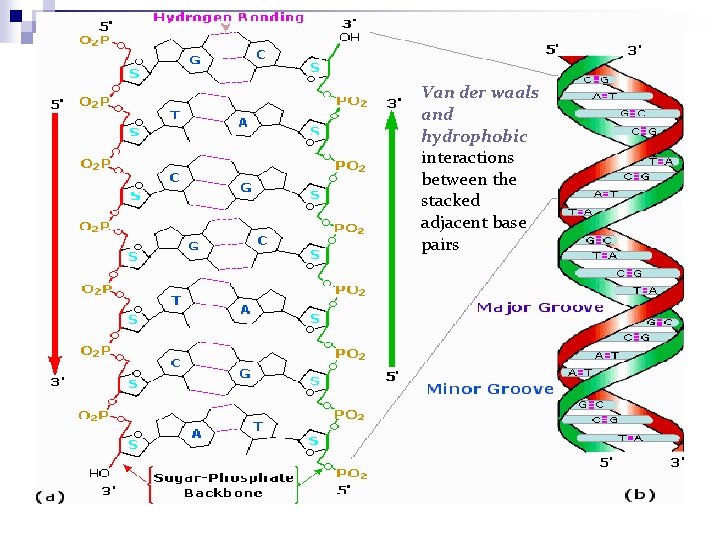
Van der waals and hydrophobic interactions between the stacked adjacent base pairs
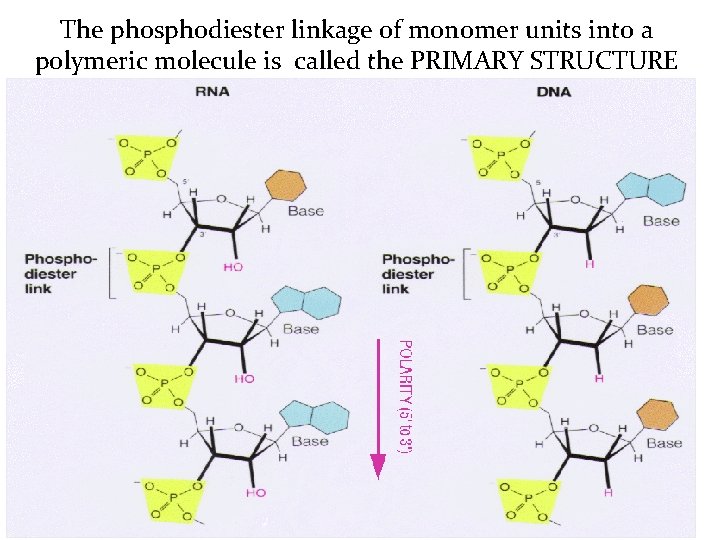
The phosphodiester linkage of monomer units into a polymeric molecule is called the PRIMARY STRUCTURE

The Primary Structure n n The arrangement (or order) of specific nucleotides along the chain is called the sequence. The sequence is genetic information. A sequence can be written simply as: ACGTT (or 5'-ACGTT-3'). Alternatively, the sequence could also be written as: 3'-TTGCA-5'.

Higher Order Structure of DNA n I. Supercoils: studies of circular DNA have shown that it can be twisted into compact supercoiled or superhelical form. ¨ Right-handed(positive) supercoil: are twisted in the same direction as the right-handed helix of B-form DNA about its axis. ¨ Left-handed(negative) supercoil: twisted on the opposite direction.

Higher Order Structure of DNA Topoisomerases: enzymes responsible for altering the superhelicity of the cellular DNA. This enzyme is very important in the process of replication. n Note: Supercoils have a high free energy compared to relaxed DNA. n Supercoil exist naturally, examples, bacterial genomic DNA and plasmid(mostly are negative). n
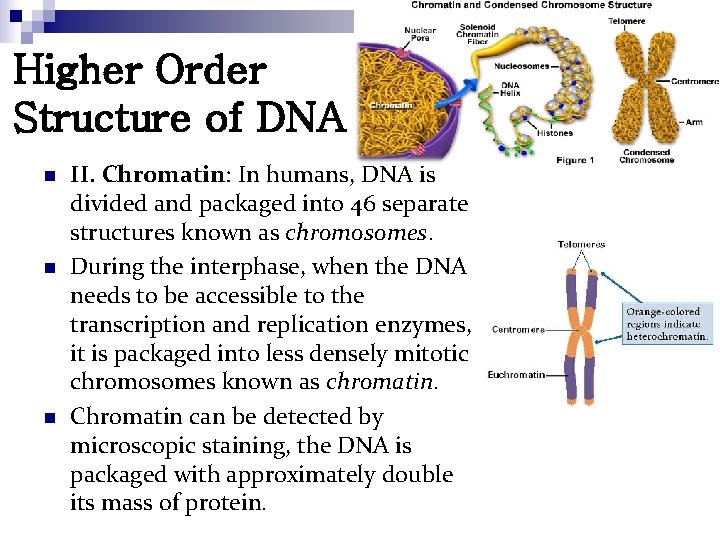
Higher Order Structure of DNA n n n II. Chromatin: In humans, DNA is divided and packaged into 46 separate structures known as chromosomes. During the interphase, when the DNA needs to be accessible to the transcription and replication enzymes, it is packaged into less densely mitotic chromosomes known as chromatin. Chromatin can be detected by microscopic staining, the DNA is packaged with approximately double its mass of protein.
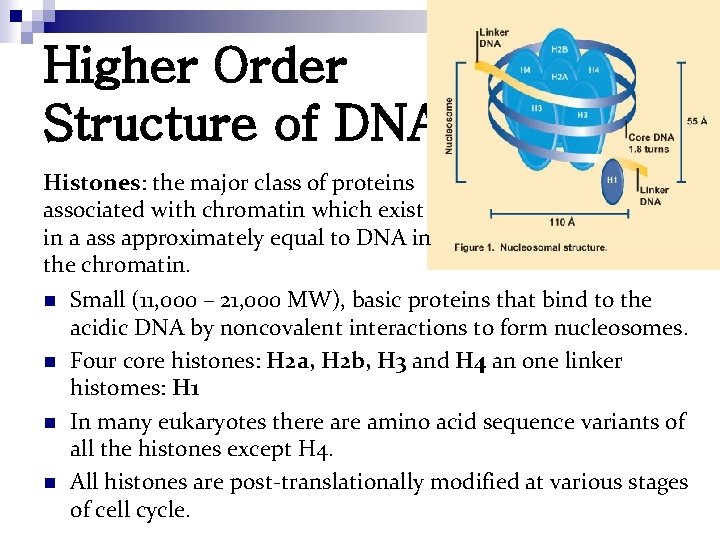
Higher Order Structure of DNA Histones: the major class of proteins associated with chromatin which exist in a ass approximately equal to DNA in the chromatin. n n Small (11, 000 – 21, 000 MW), basic proteins that bind to the acidic DNA by noncovalent interactions to form nucleosomes. Four core histones: H 2 a, H 2 b, H 3 and H 4 an one linker histomes: H 1 In many eukaryotes there amino acid sequence variants of all the histones except H 4. All histones are post-translationally modified at various stages of cell cycle.

DNA SYNTHESIS http: //highered. mcgrawhill. com/sites/0072437316/student_view 0/chapter 14/ani mations. html# View: 1. How Nucleotides are added in DNA 2. DNA replication fork

QUIZ 1. DNA polymerase III can only add nucleotides to an existing chain, so _________ is required. A) an RNA primer B) DNA polymerase I C) helicase D) a DNA primer 2. The enzymes that break hydrogen bonds and unwind DNA are: A) Primers B) Forks C) Helicases D) Polymerases

QUIZ 3. Replication begins at a specific site in the DNA called ______. 4. During replication within the fork, ____ bind to the single-stranded regions preventing the strands from rejoining. 5. This enzyme replaces the primase and is able to add DNA nucleotides to the RNA primer 6. Short fragment or sequences of discontinuous DNA

QUIZ 7. DNA replication is from 5’ to 3’ direction. True or False. 8. -9. In which strand DNA is synthesized continuously? Discontinuously? 10. The DNA fragments on the lagging strand are hooked together by the enzyme____.
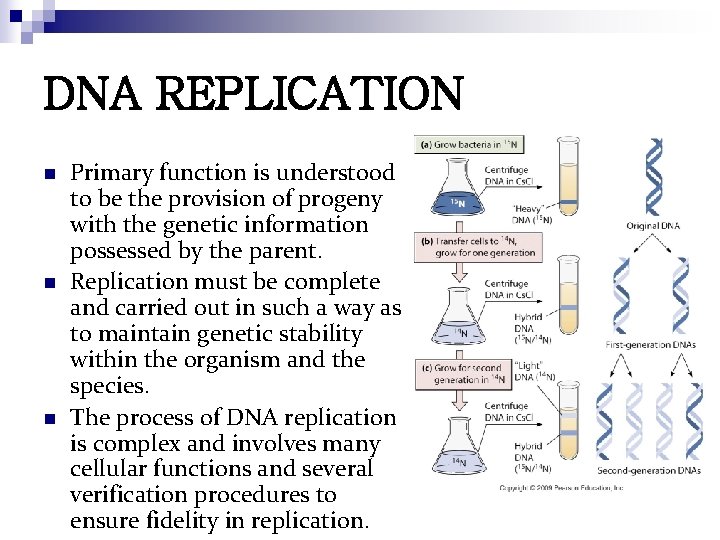
DNA REPLICATION n n n Primary function is understood to be the provision of progeny with the genetic information possessed by the parent. Replication must be complete and carried out in such a way as to maintain genetic stability within the organism and the species. The process of DNA replication is complex and involves many cellular functions and several verification procedures to ensure fidelity in replication.

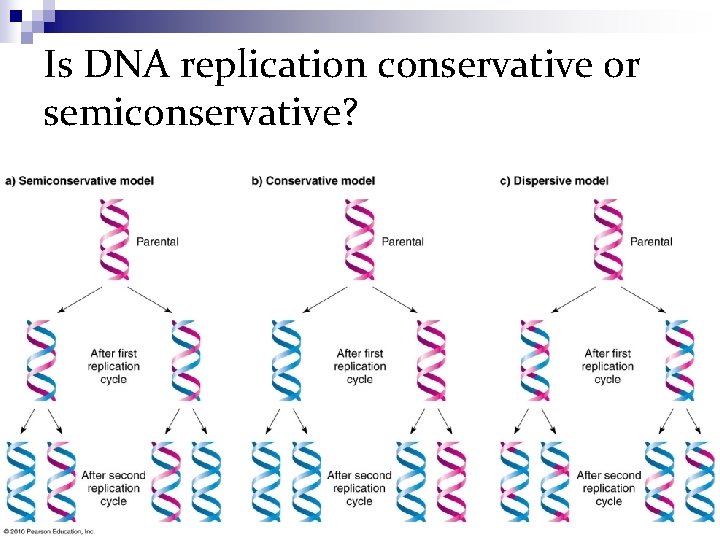
Is DNA replication conservative or semiconservative? n Conservative - old strand acts as a template ¨ One daughter strand is the original template while the other strand is composed entirely out of new nucleotides n Semiconservative - old strand splits apart and acts as a template ¨ Both daughter strands are composed of one of the old strands and one comprised out of new nucleotides

Is DNA replication conservative or semiconservative? n In their brief paper, "Molecular structure of Nucleic Acids: A Structure for Deoxyribose Nucleic Acid" , James Watson and Francis Crick (1953) wrote: ¨ "It has not escaped our notice that the specific pairing we have postulated immediately suggests a possible copying mechanism for the genetic material. " n This mechanism for DNA replication is known as the semiconservative model.
Is DNA replication conservative or semiconservative?

Semi-Conservative Model ØThe two parental strands separate and each makes a copy of itself. ØAfter one round of replication, the two daughter molecules each comprises one old and one new strand. ØNote that after two rounds, two of the DNA molecules consist only of new material, while the other two contain one old and one new strand.
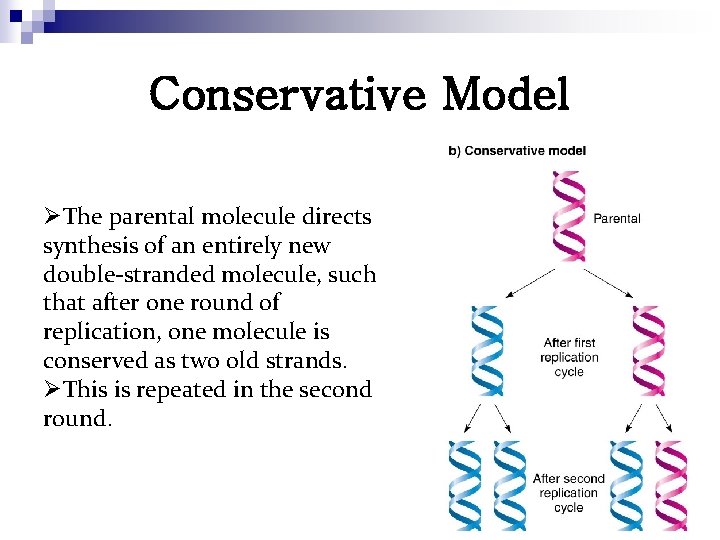
Conservative Model ØThe parental molecule directs synthesis of an entirely new double-stranded molecule, such that after one round of replication, one molecule is conserved as two old strands. ØThis is repeated in the second round.

Dispersive Model ØMaterial in the two parental strands is distributed more or less randomly between two daughter molecules. ØIn the model shown here, old material is distributed symmetrically between the two daughters molecules. ØOther distributions are possible.
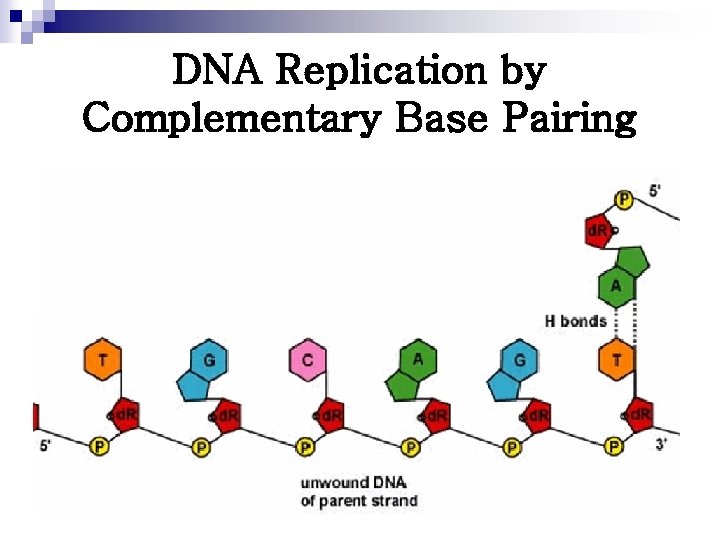
DNA Replication by Complementary Base Pairing
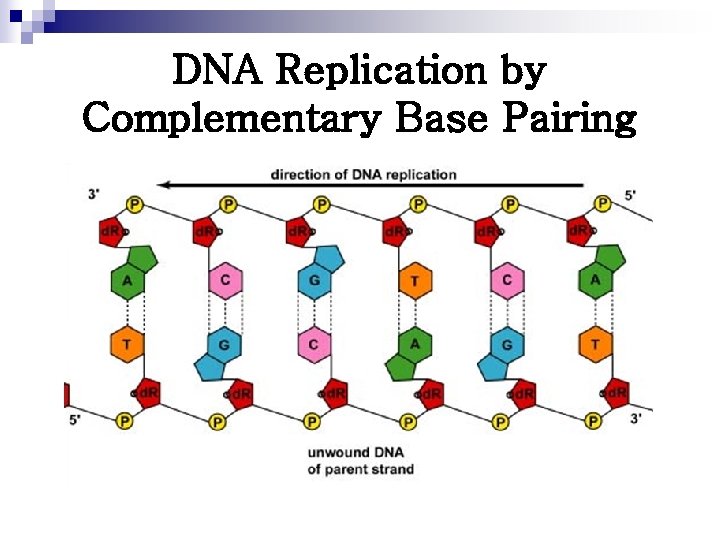
DNA Replication by Complementary Base Pairing

Steps involved in DNA Replication in Eukaryotes Identification of origin of replication Unwinding (denaturation) of ds. DNA to provide a ss. DNA template Formation of the replication fork Initiation of DNA synthesis and elongation Formation of replication bubbles with ligation of the newly synthesized DNA segments Reconstitution of chromatin structure 1. 2. 3. 4. 5. 6. • • E. coli is used to describe the replication process in prokaryotes Less complex in prokaryotes
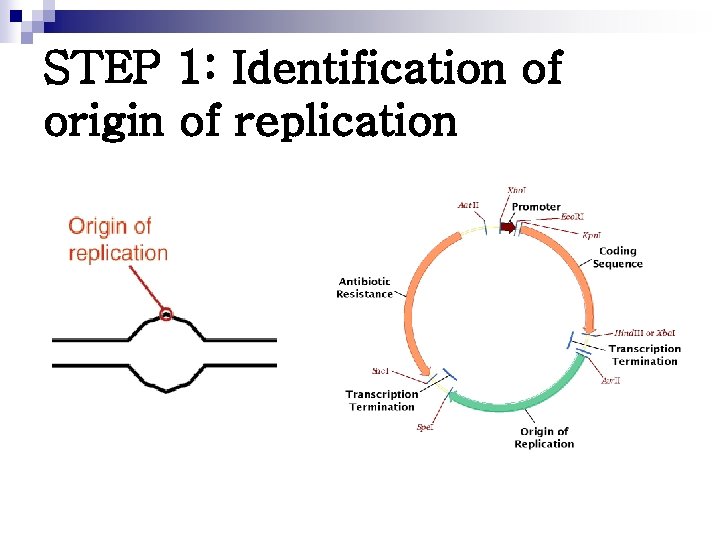
STEP 1: Identification of origin of replication

Origin of replication in eukaryotic DNA n Multiple sites ¨ Provides mechanism for rapidly replicating the great length of the eukaryotic DNA n These sites include a short sequence composed almost exclusively of A-T base pairs called CONSENSUS SEQUENCE ¨ The order of nucleotide is essentially same at each site
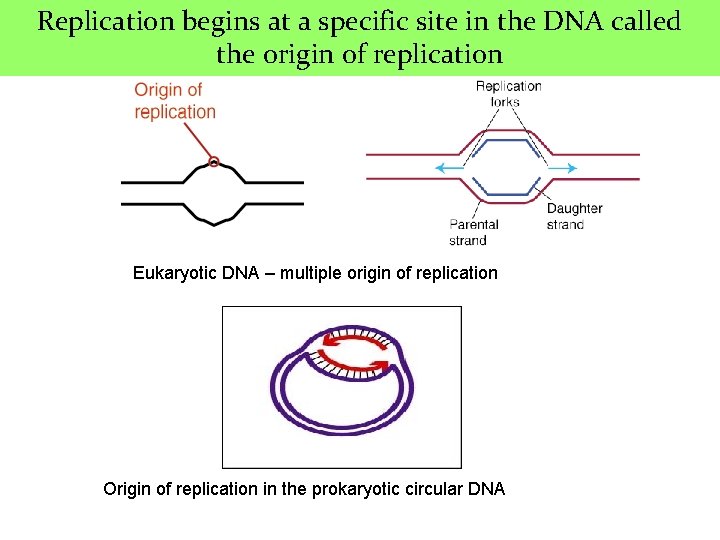
Replication begins at a specific site in the DNA called the origin of replication Eukaryotic DNA – multiple origin of replication Origin of replication in the prokaryotic circular DNA
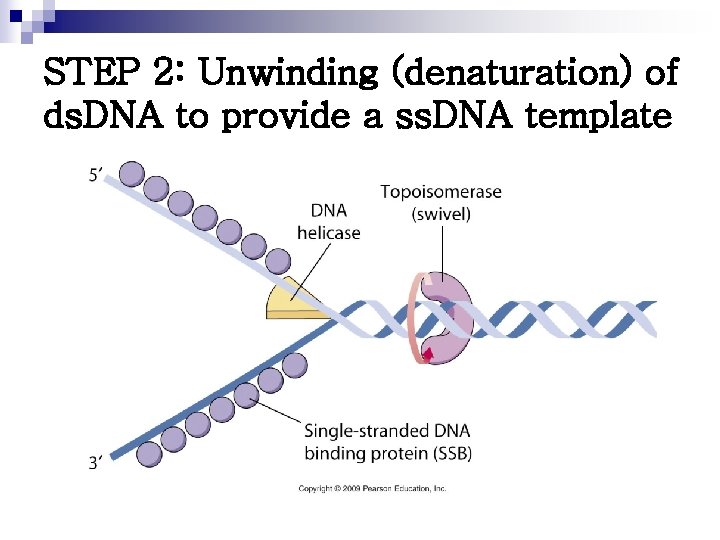
STEP 2: Unwinding (denaturation) of ds. DNA to provide a ss. DNA template
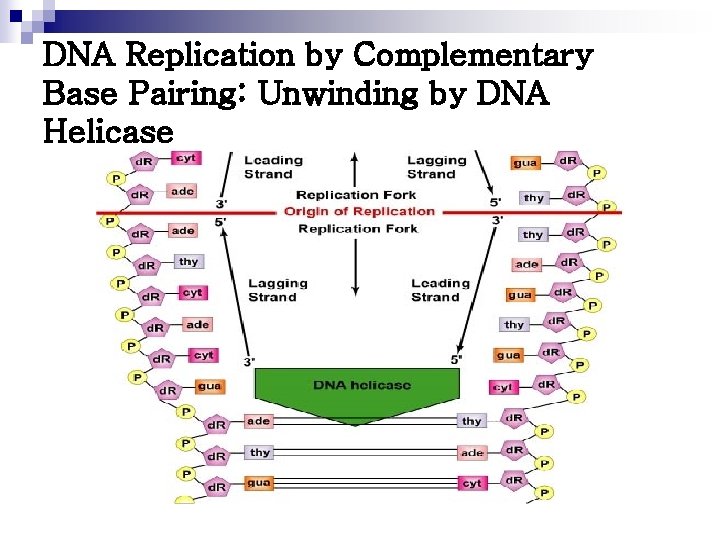
DNA Replication by Complementary Base Pairing: Unwinding by DNA Helicase
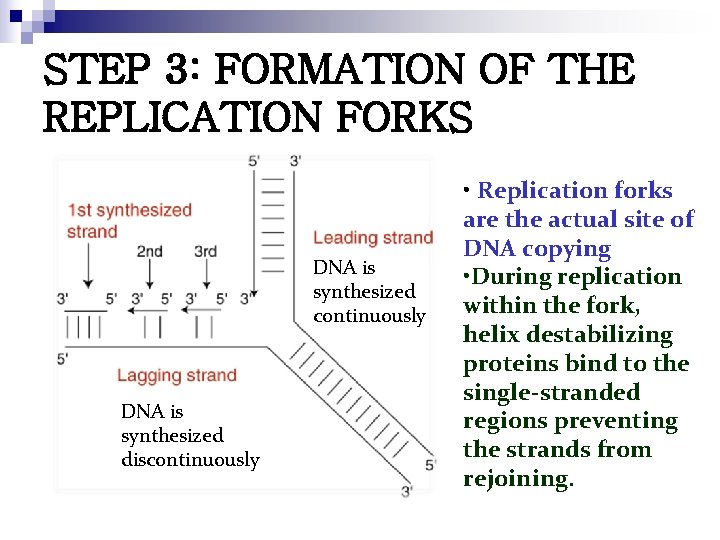
STEP 3: FORMATION OF THE REPLICATION FORKS DNA is synthesized continuously DNA is synthesized discontinuously • Replication forks are the actual site of DNA copying • During replication within the fork, helix destabilizing proteins bind to the single-stranded regions preventing the strands from rejoining.
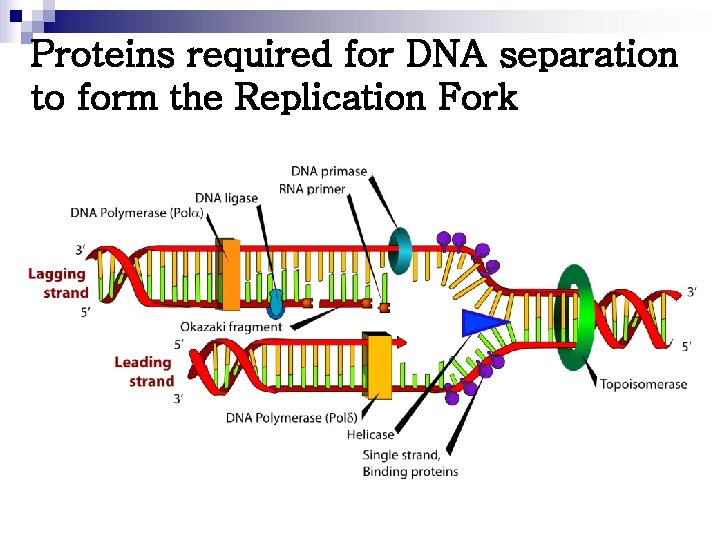
Proteins required for DNA separation to form the Replication Fork

n Dna. A protein ¨ Bind to specific nucleotide sequences at the ORIC ¨ Causes the double-stranded DNA to melt n n Strands separate, forming localized regions of single-stranded Single-stranded DNA-binding (SSB) proteins ¨ Also called helix-destabilizing proteins ¨ Keep 2 strands of DNA separated in the area of replication origin ¨ Also protect DNA from nucleases that cleave ss. DNA n DNA helicases ¨ Bind to ss. DNA near the replication fork ¨ Unwind the double helix ¨ Requires energy provided by ATP
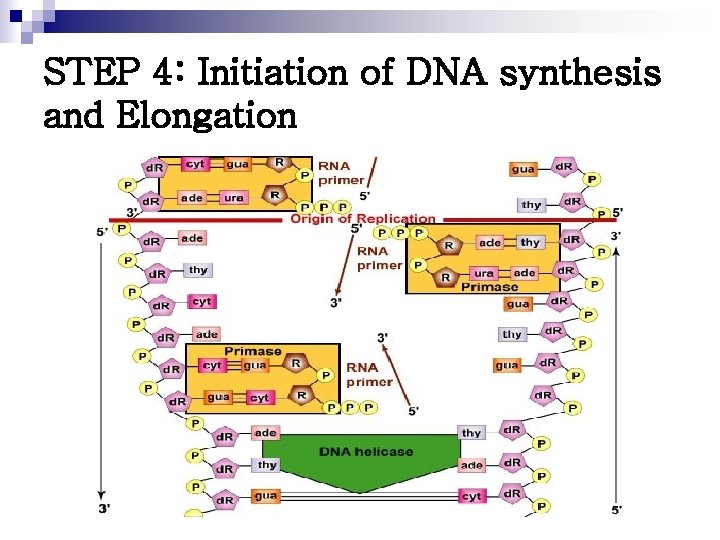
STEP 4: Initiation of DNA synthesis and Elongation
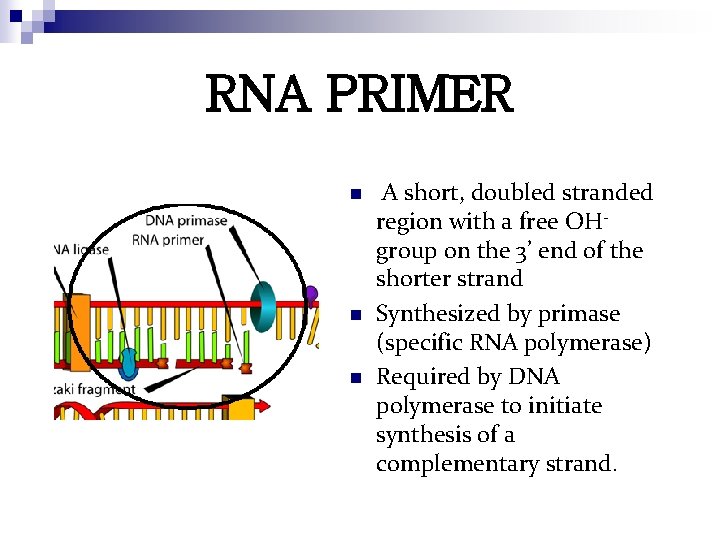
RNA PRIMER n n n A short, doubled stranded region with a free OHgroup on the 3’ end of the shorter strand Synthesized by primase (specific RNA polymerase) Required by DNA polymerase to initiate synthesis of a complementary strand.
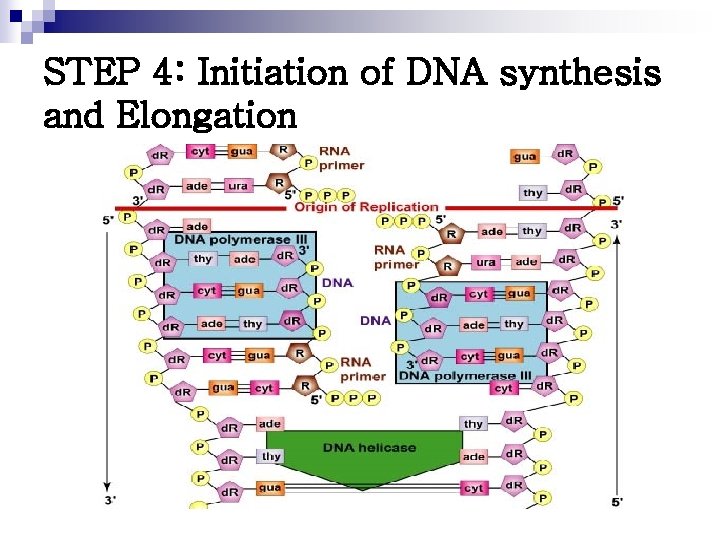
STEP 4: Initiation of DNA synthesis and Elongation

DNA Polymerases n n n Responsible for copying the DNA templates Only able to “read” in the 3’ 5’ direction and synthesize new DNA in the 5’ 3’ direction Elongate a new DNA strand by adding deoxyribonucleotide, one at a time, to the 3’-end of the growing chain DNA POLYMERASE I Replaces the primase and is able to add DNA nucleotides to the RNA primer n Catalyze DNA chain elongation n
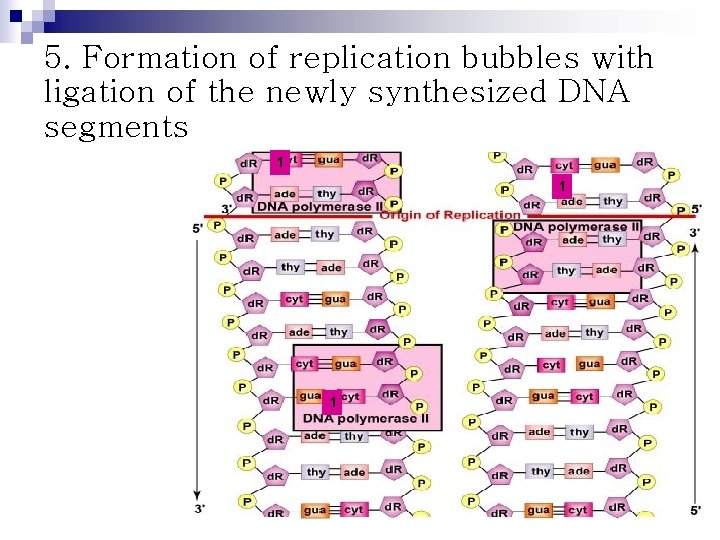
5. Formation of replication bubbles with ligation of the newly synthesized DNA segments 1 1 1
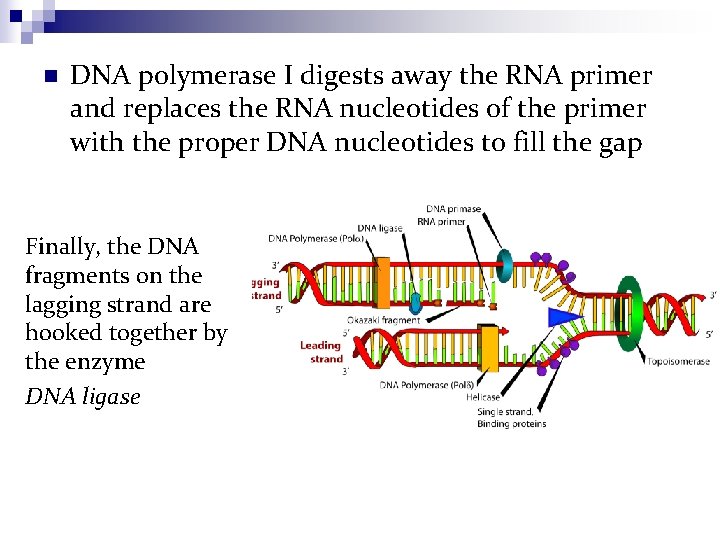
n DNA polymerase I digests away the RNA primer and replaces the RNA nucleotides of the primer with the proper DNA nucleotides to fill the gap Finally, the DNA fragments on the lagging strand are hooked together by the enzyme DNA ligase
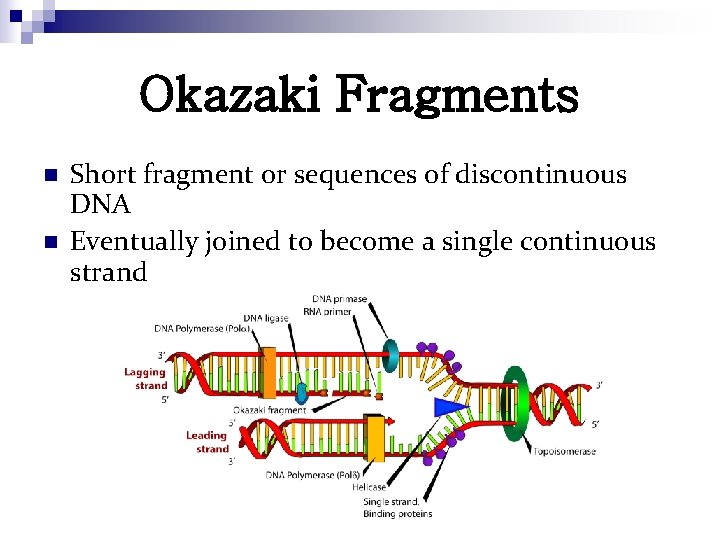
Okazaki Fragments n n Short fragment or sequences of discontinuous DNA Eventually joined to become a single continuous strand

Classes of Proteins involved in Replication PROTEIN FUNCTION DNA polymerases Deoxynucleotide polymerization Helicases Processive unwinding of DNA Topoisomerases Relieve torsional strain that results from helicase-induced unwinding Primase Initiates synthesis of RNA primers Single-strand binding proteins Prevent premature reannealing of ds. DNA ligase Seals the single strand nick between the nascent chain and Okazaki fragments on lagging strand

DNA Topoisomerase I § Solves the problem caused by tension generated by winding/unwinding of DNA. n It wraps around DNA and makes a cut (producing a “nick”) permitting the helix to spin. n Once DNA is relaxed, topoisomerase reconnects broken strands n Relax negative supercoils in E. coli, and both negative and positive supercoils in eukaryotic cells Note: Have both strand-cutting (nuclease) and strandresealing (ligase) activities
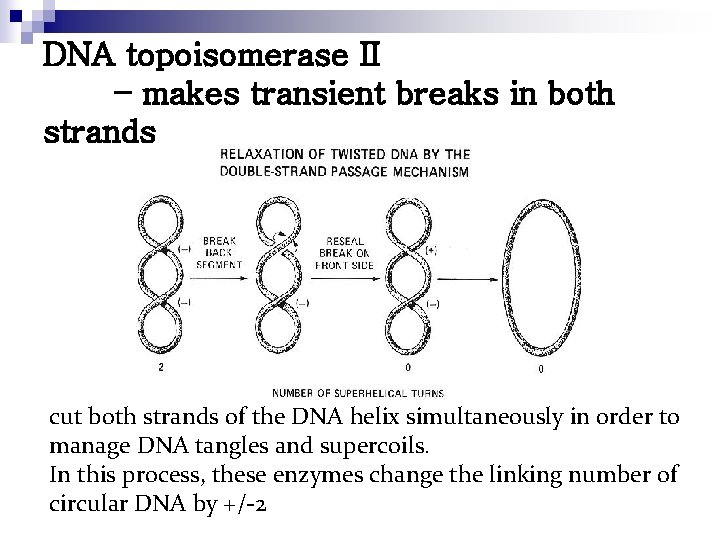
DNA topoisomerase II – makes transient breaks in both strands cut both strands of the DNA helix simultaneously in order to manage DNA tangles and supercoils. In this process, these enzymes change the linking number of circular DNA by +/-2

The DNA Polymerases Three (3) important properties 1. Chain elongation 2. Processivity 3. Proofreading – ensures replication fidelity

Prokaryotic and Eukaryotic DNA polymerases E. coli Mammalian Function I Gap filling and synthesis of lagging strand II DNA proofreading and repair DNA repair Mitochondrial DNA synthesis Processive, leading strand synthesis III
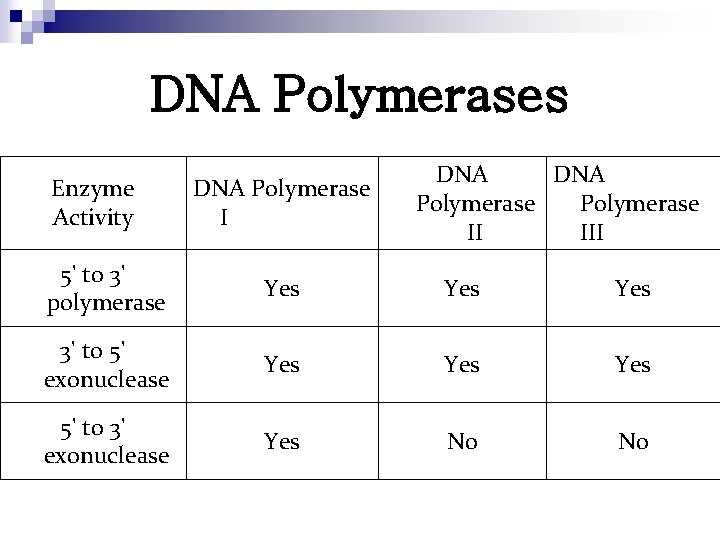
DNA Polymerases Enzyme Activity DNA Polymerase II III 5' to 3' polymerase Yes Yes 3' to 5' exonuclease Yes Yes 5' to 3' exonuclease Yes No No

E. coli DNA Polymerase I n 5'-to-3' DNA Polymerase activity ¨ n n Gap is filled 3'-to-5' exonuclease (Proofreading activity) 5'-to-3' exonuclease (Nick translation activity) Remove one nucleotide at a time from region of DNA that is properly base-paired ¨ Also remove groups of altered nucleotides in the 5’→ 3’ direction, removing from 1 to 10 nucleotides at a time ¨
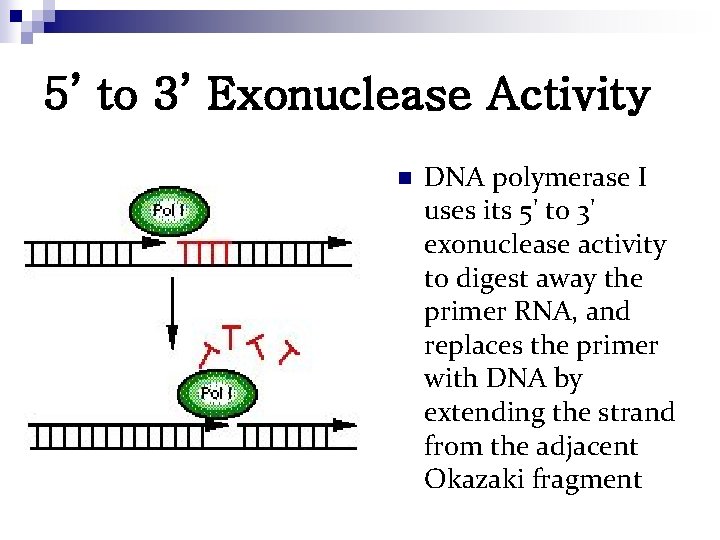
5’ to 3’ Exonuclease Activity n DNA polymerase I uses its 5' to 3' exonuclease activity to digest away the primer RNA, and replaces the primer with DNA by extending the strand from the adjacent Okazaki fragment
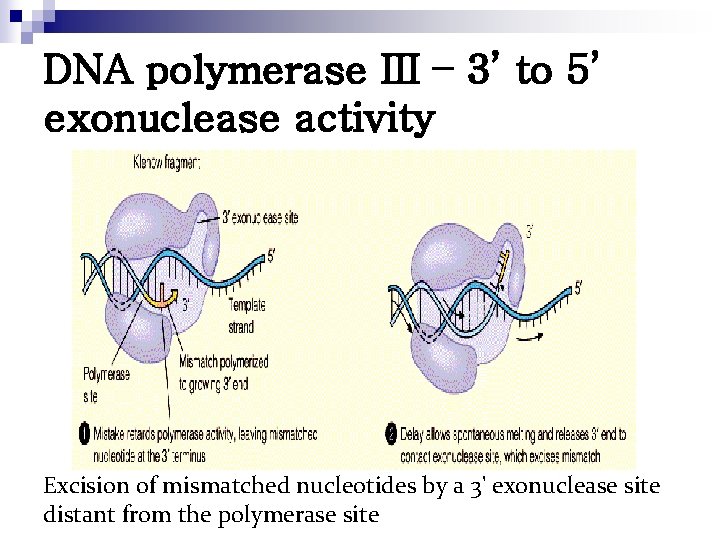
DNA polymerase III – 3’ to 5’ exonuclease activity Excision of mismatched nucleotides by a 3' exonuclease site distant from the polymerase site
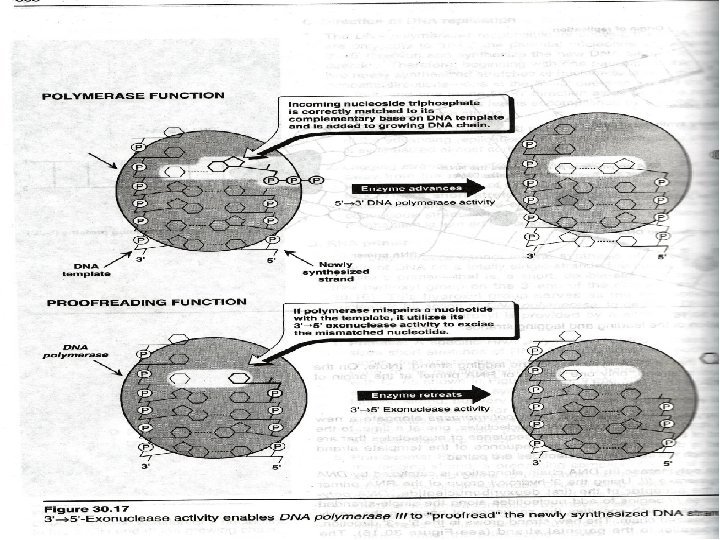
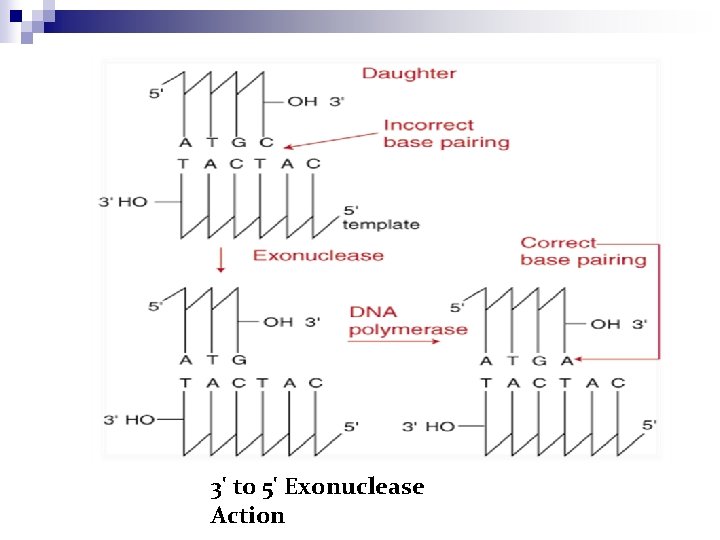
3' to 5' Exonuclease Action
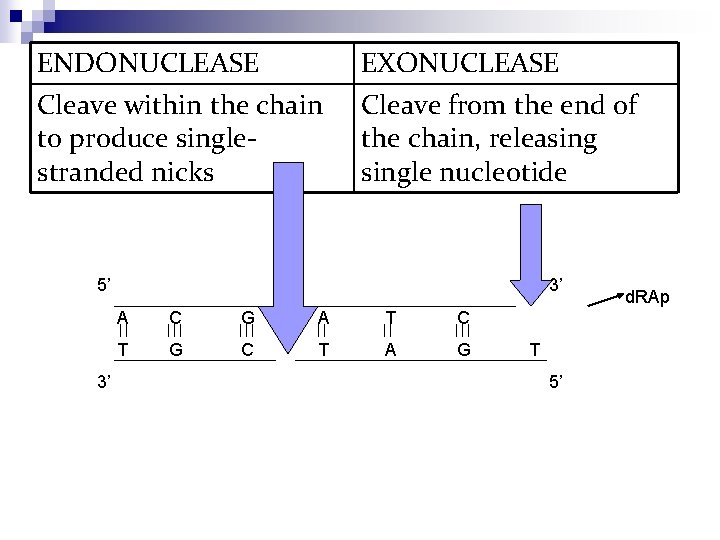
ENDONUCLEASE Cleave within the chain to produce singlestranded nicks EXONUCLEASE Cleave from the end of the chain, releasingle nucleotide 5’ 3’ 3’ A C G A T C A T G C T A G T 5’ d. RAp

Eukaryotic DNA Replication Polymerases are designated by Greek letter n Pol - has primase activity n Pol - elongate the leading strand n Pol - elongate the lagging strand in mammals ¨ Pol and Pol both have 3’ to 5’ exonuclease activity n n Pol - analogous to E. coli DNA polymerase—that is, it can, in association with other proteins, excise primers and carry out “repairs” Pol - replicates mitochondrial DNA
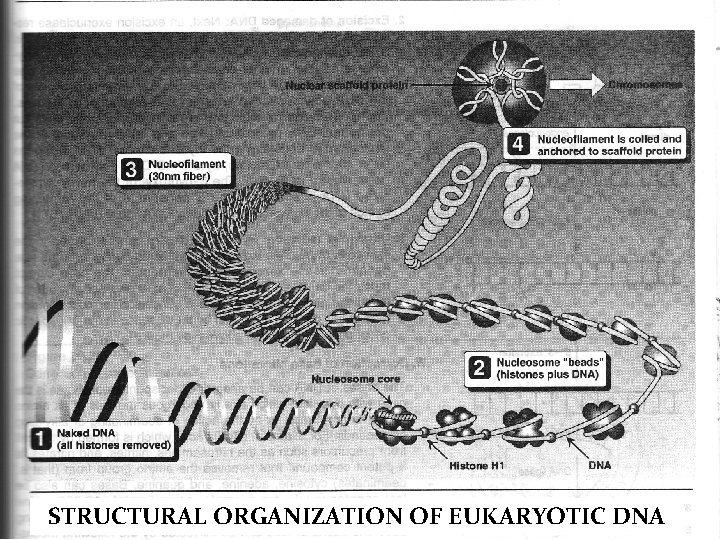
STRUCTURAL ORGANIZATION OF EUKARYOTIC DNA

Organization of Eukaryotic DNA HISTONES n Basic proteins tightly bound to eukaryotic DNA n 5 classes: H 1, H 2 A, H 2 B, H 3, and H 4 n H 1 binds to the DNA chain between the nucleosome beads n H 1 appears to aid the packing of nucleosomes into the more structures NUCLEOSOMES n Nucleosomes “beads” is formed by two molecules each of H 2 A, H 2 B, H 3, and H 4 with DNA double helix wound around nearly twice n Nucleosomes, joined by “linker” DNA (approx. 50 nucleotides in length) form a polynucleosome or nucleofilament.
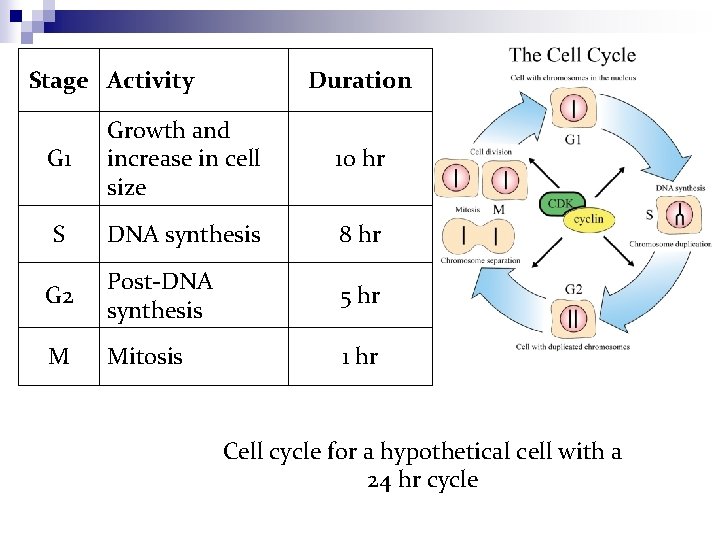
Stage Activity Duration G 1 Growth and increase in cell size 10 hr S DNA synthesis 8 hr G 2 Post-DNA synthesis 5 hr M Mitosis 1 hr Cell cycle for a hypothetical cell with a 24 hr cycle

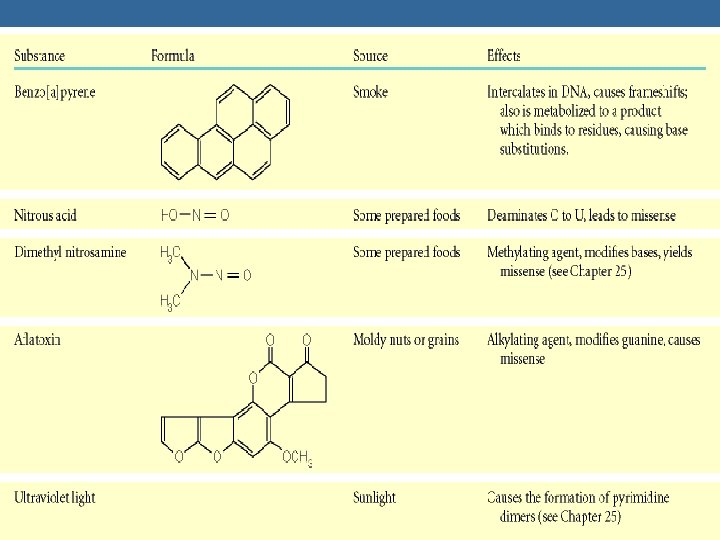
DNA DAMAGE CAUSES OF DNA DAMAGE 1. UV LIGHT - RADIATION 2. CHEMICALS 3. OTHER AGENTS DNA DAMAGE CAN BE: 1. BASE ALTERATION 2. REMOVAL OR LOST OF NUCLEOTIDE BASE

Four Mechanisms of DNA Repair MECHANISM PROBLEM SOLUTION MISMATCH REPAIR Copying errors Methyl-directed strand cutting, exonuclease digestion, and replacement BASE EXCISION REPAIR Spontaneous, chemical, or radiation damage to a single base Base removal by Nglycosylase, abasic sugar removal, replacement NUCLEOTIDE EXCISION Spontaneous, chemical, REPAIR or radiation damage to a DNA segment Removal of an approx. 30 nucleotide oligomer and replacement DOUBLE-STRAND BREAK REPAIR Synapsis, unwinding, alignment, ligation Ionizing radiation, chemotherapy, oxidative free radicals

Proteins involved in the DNA repairing of E. coli
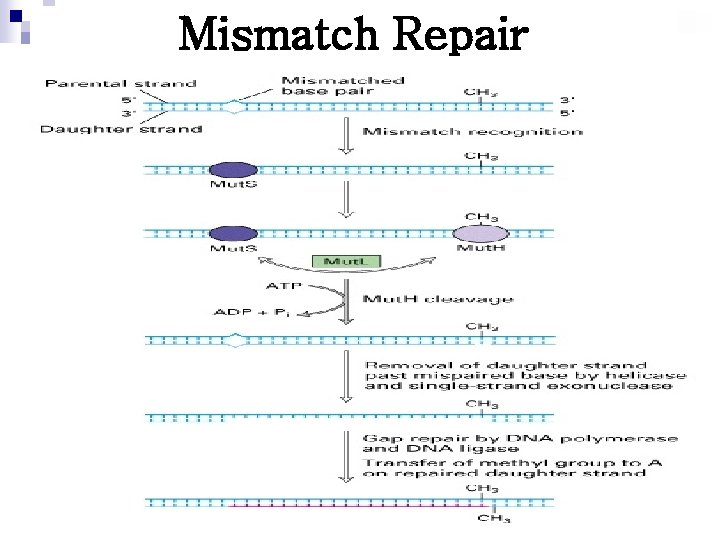
Mismatch Repair
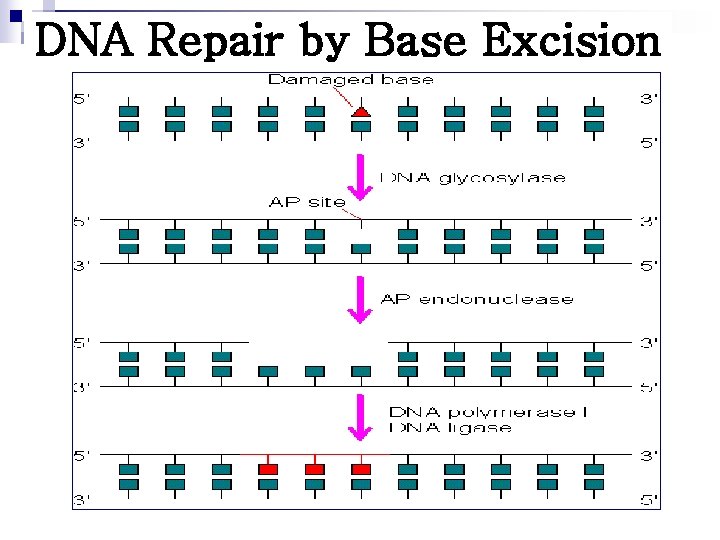
DNA Repair by Base Excision
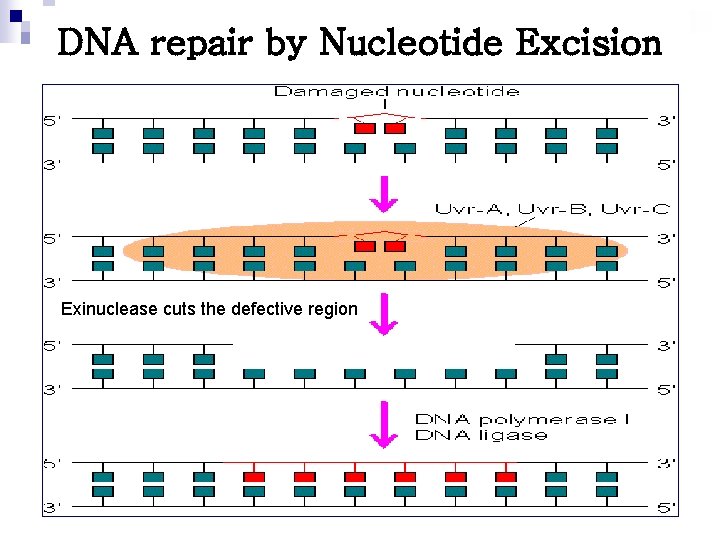
DNA repair by Nucleotide Excision Exinuclease cuts the defective region
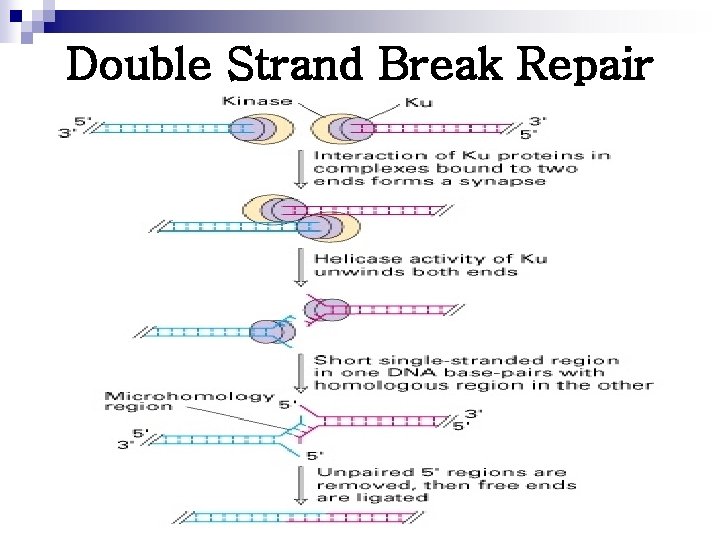
Double Strand Break Repair
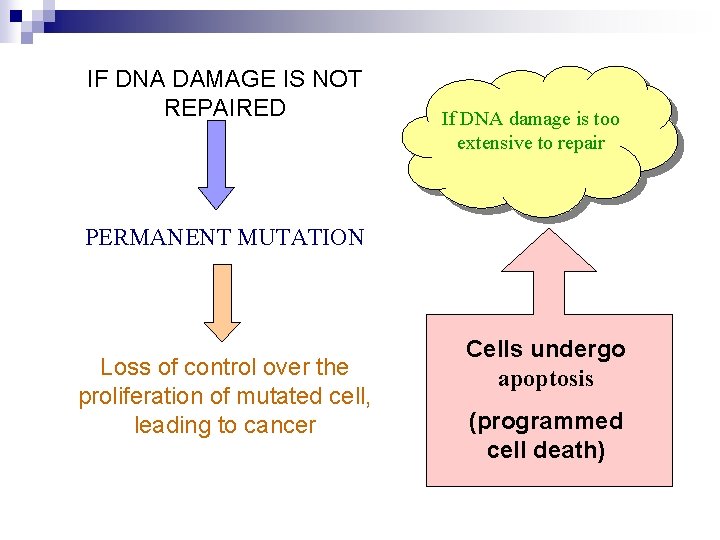
IF DNA DAMAGE IS NOT REPAIRED If DNA damage is too extensive to repair PERMANENT MUTATION Loss of control over the proliferation of mutated cell, leading to cancer Cells undergo apoptosis (programmed cell death)

Diseases resulting from disordered DNA repair capabilities n Xeroderma Pigmentosum (XP) ¨ XP is a rare inherited disease of humans which, among other things, predisposes the patient to pigmented lesions on areas of the skin exposed to the sun and an elevated incidence of skin cancer. ¨ XP can be caused by mutations in any one of several genes — all of which have roles to play in NER

Diseases resulting from disordered DNA repair capabilities FANCONI’S ANEMIA • An inherited disease that mainly affects the bone marrow. • The disease is caused by a genetic defect. • This defect prevents cells from fixing damaged DNA or removing toxic substances called oxygen-free radicals that damage cells. • People with certain birth defects or who develop low blood counts may have this disease.

Diseases resulting from disordered DNA repair capabilities ATAXIA-TELANGIECTASIA n n n Rare, inherited, childhood disease that affects the brain and other parts of the body. Ataxia refers to uncoordinated movements, such as walking. Telangiectasis is the enlargement of blood vessels (capillaries) just below the surface of the skin. Telangiectasis appear as tiny, red, spider-like veins. The disease results from defects in the ataxia telangiectasia mutated (ATM) gene. n Defects in this gene can lead to abnormal cell death in various places of the body, including the part of the brain that helps coordinate movement.
- Slides: 124