Nuclearchemical synthesis of iron compounds and their Mssbauer

Nuclear-chemical synthesis of iron compounds and their Mössbauer identification Proposal to the ISOLDE and Neutron Time-of. Flight Committee H. P. Gunnlaugsson, S. K. Dedushenko, L. Hemmingsen et al. ,

Outline Introduction about 57 Mn (T½ = 1. 5 min. ) → 57 Fe Chemistry with 57 Mn Physics/chemistry that can be addressed here Mössbauer collaboration at ISOLDE/CERN Beam request 2
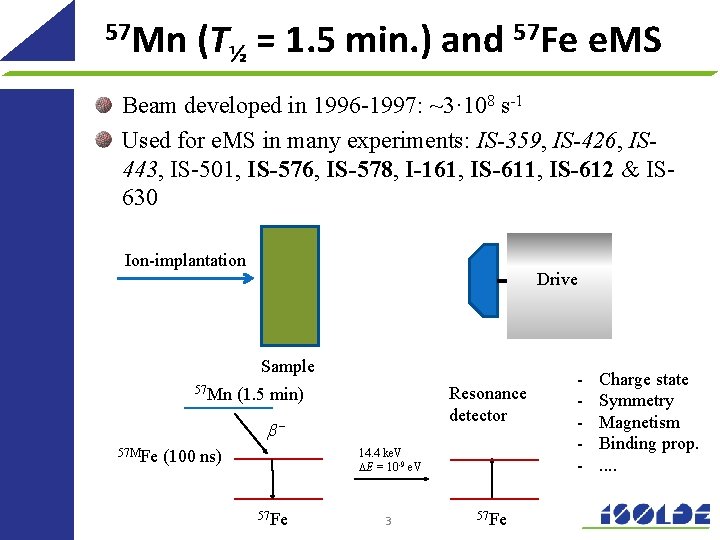
57 Mn (T½ = 1. 5 min. ) and 57 Fe e. MS Beam developed in 1996 -1997: ~3· 108 s-1 Used for e. MS in many experiments: IS-359, IS-426, IS 443, IS-501, IS-576, IS-578, I-161, IS-612 & IS 630 Ion-implantation Drive Sample 57 Mn Resonance detector (1. 5 min) b– 57 MFe 14. 4 ke. V DE = 10 -9 e. V (100 ns) 57 Fe 3 57 Fe - Charge state Symmetry Magnetism Binding prop. . .
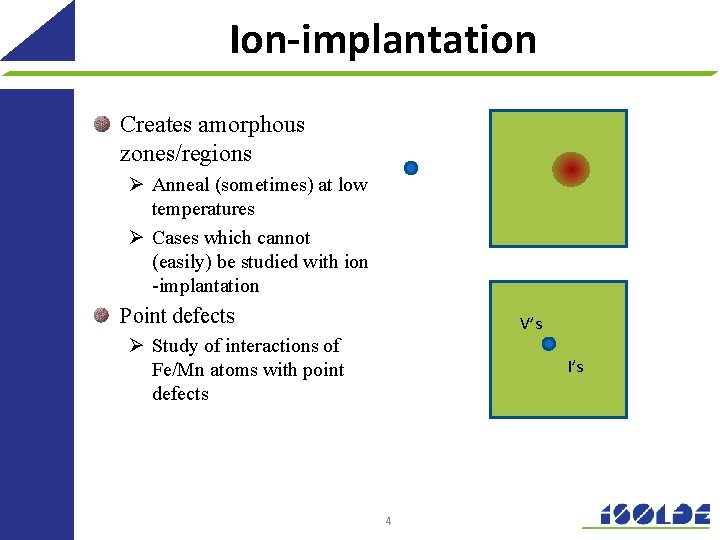
Ion-implantation Creates amorphous zones/regions Ø Anneal (sometimes) at low temperatures Ø Cases which cannot (easily) be studied with ion -implantation Point defects V’s Ø Study of interactions of Fe/Mn atoms with point defects I’s 4

Make materials with the probe Surface physics Ø Used successfully in ASPIC/VITO Ø Implantation into catcher transfer MBE growth Ø Measurements with Perturbed Angular Correlation (PAC) spectroscopy Ø Dictated by the life-time of the radioactive probe Ø Many fundamental papers on surface physics in the 1990’s Biophysics Ø Used successfully for different isotopes (111 m. Cd, 199 Hg, . . . ) all with T½ > 30 minutes Ø Implantation into ice thawing reactions Ø Measurements with PAC 5
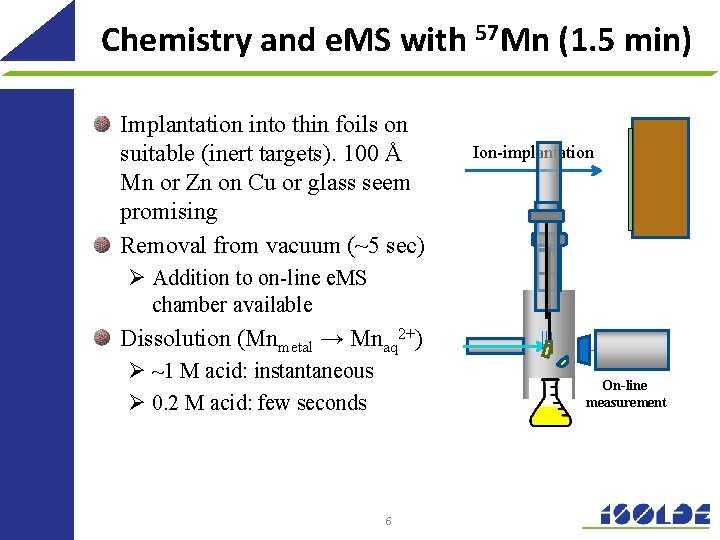
Chemistry and e. MS with 57 Mn (1. 5 min) Implantation into thin foils on suitable (inert targets). 100 Å Mn or Zn on Cu or glass seem promising Removal from vacuum (~5 sec) Ion-implantation Ø Addition to on-line e. MS chamber available Dissolution (Mnmetal → Mnaq 2+) Ø ~1 M acid: instantaneous Ø 0. 2 M acid: few seconds On-line measurement 6
Chemistry with 57 Mn (cont) Chemical reactions Ø 1 -300 seconds Transfer to a cold finger Off-line measurements (77 K – 273 K) Variables: Ø Acid strength Ø Reaction time/temperature Ø Measurement temperature Ø. . . Chemical reaction (1 -300 s) 7

Risk involved Radioactivity Ø ~27 µSv/h at 30 cm distance Ø ~50 µSv (total dose during ~5 day experiments) Ø Transfer of (radiactive) acids in confined spaces Protocols have to be made that ensure the safety at all levels of measurements Ø Practice-practice, . . Risk assessment Ø ~25 min wait time in case of spills (radioactive) Ø Water to thin out chemical spills? No long lived impurities (MR-TOF) Ø No radioactive waste (only chemical) 8
Physics & e. MS Mössbauer spectrum: d | (0)|2 DEQ Ø charge/spin state of the atom Area DEQ VZZ Ø ~ coordination d Area f(T, q. D) Ø ~ binding properties 9

Nuclear chemical synthesis Fe. O 2 does not exist (Fe as Fe 4+) However, Mn. O 2 exists Ø Make 57 Mn. O 2 Ø Wait for 57 Fe. O 2 10

High charge states of Fe “Natural” charge states: Ø Fe: 2+, 3+, 4+, 5+, 6+ Ø Mn: 2+, 3+, 4+, 5+, 6+, 7+, 8+ Oxidation involving Mn 7+ Ø 2 Mn 2+ + 5 Na. Bi. O 3 + 14 H+ = 2 Mn. O 4 - + 5 Na+ + 5 Bi 3+ + 7 H 2 O Freezing and wait for the decay. . . (b-) The 100 ns lifetime of 57*Fe, should not change the local configuration i. e. Fe 7+ should be measured All supported by theoretical calculations 11

Future. . . Isotopic exchange Ø 57 Mn 2+ + 55 Mn. O 4 - = 55 Mn 2+ + 57 Mn. O 4 - Precipitation of Mn. O 2 Ø + comparison to direct implantation into MO 2 substrates Biological role: Mn 2+ function as cofactors for a large variety of enzymes and are essential in detoxification of superoxide free radicals in organisms. This project may provide a new analytical method within this field of study 12

The e. MS collaboration The Mössbauer collaboration at ISOLDE/CERN, >30 active members Involved in 6 experiments at CERN Organize the WEMS conference series Year Rev. publications 2017 (2) 2016 5 2015 2 2014 4 2013 2 2012 10 2016 Key people: H. P. Gunnlaugsson, Copenhagen: e. MS S. K. Dedushenko, Moscow : Chemistry & MS L. Hemmingsen, Copenhagen: Biophysics & PAC S. P. A. Sauer, Copenhagen: Chemistry, simulations J. Bendix, Copenhagen, Calculations 13

Beam request For each reaction (See tables), one needs to: Ø vary reaction time/temperature Ø strength (acid) Ø possibly repeat to get sufficient statistics Ø Measurement temperature Ø. . Each measurement: ~10 min. Thanks for your attention Ø +20% calibration Ø +20% contingency 14 shifts requested for the next 3 operation years 14
- Slides: 14