Nuclear Symbols Mass number Atomic number 12 6

Nuclear Symbols Mass number Atomic number 12 6 C Carbon-12

Nuclear Reactions • Unstable nuclei undergo spontaneous changes that change their number of protons and neutrons. • A nuclear reaction is a reaction that affects the nucleus of an atom.
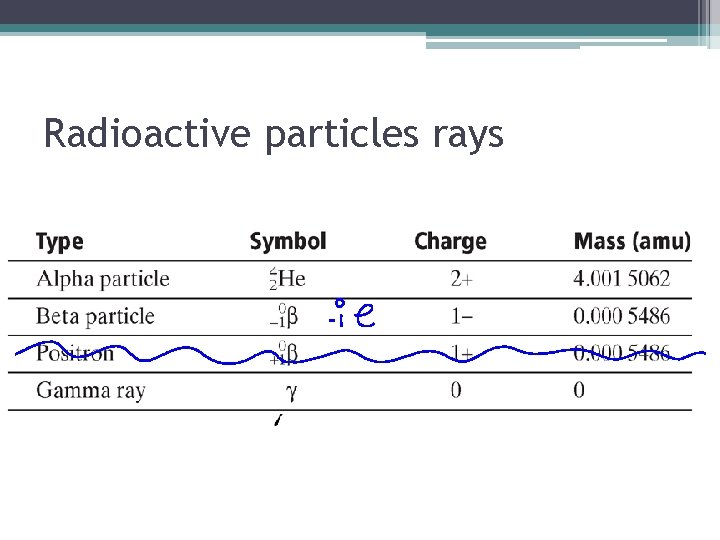
Radioactive particles rays
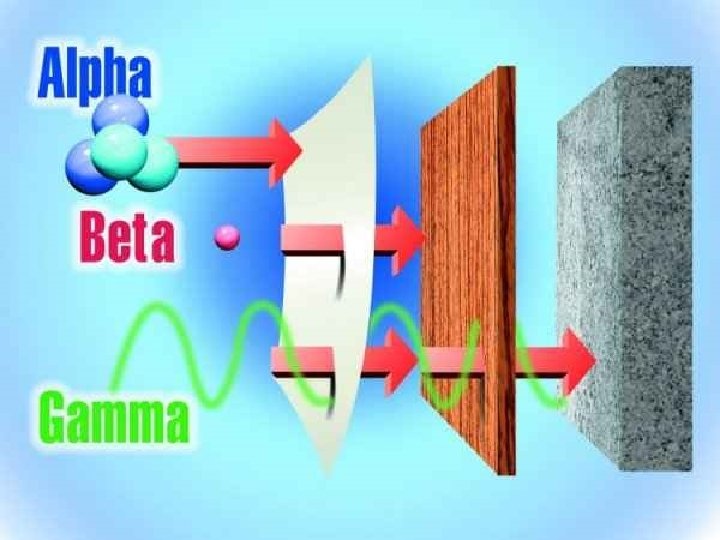

Section 2: Radioactive Decay • Radioactive Decay: is the spontaneous disintegration of a nucleus into a slightly lighter nucleus. This leads to more stable nucleons. • 3 types of radioactive decay • Alpha Emission • Beta Emission • Gamma Emission •

Alpha Emission • An alpha particle (α) is a helium nucleus. • Lowest penetrating power. • Alpha emission is restricted almost entirely to very heavy nuclei. 210 84 Po 206 82 Pb + 4 2 He

Beta Emission ▫ A beta particle (β or e) is an electron emitted from the nucleus during some kinds of radioactive decay. ▫ To decrease the number of neutrons, a neutron can be converted into a proton and an electron. 14 6 C 14 7 N + 0 -1 e
Gamma Emission Gamma rays ( ) are high-energy electromagnetic waves emitted from a nucleus as it changes from an excited state to a ground energy state. -Gamma rays don’t actually change the nucleus of an atom.

Write the Nuclear Reaction for: The a decay of Americum-245

The b decay of Thorium-236

The a decay of Iodine - 125

Uranium-234 to Thorium-230

Half-life ▫ Half-life, t 1/2, is the time required for half the atoms of a radioactive nuclide to decay. ▫ Each radioactive nuclide has its own half-life. ▫ More-stable nuclides decay slowly and have longer half-lives.
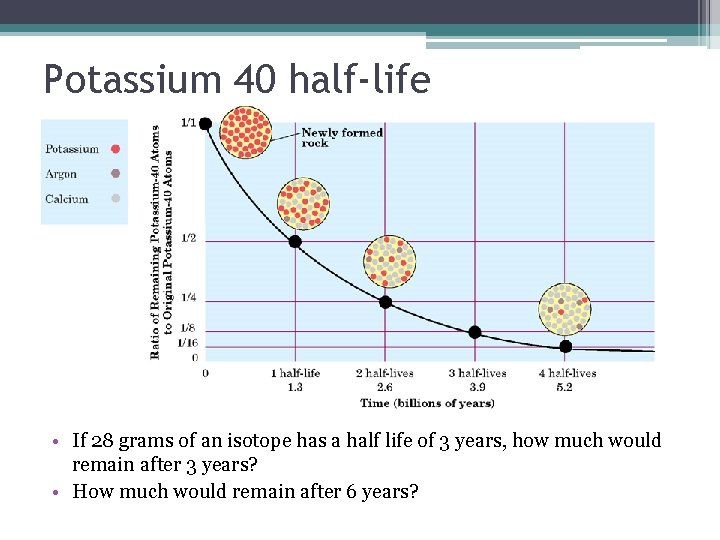
Potassium 40 half-life • If 28 grams of an isotope has a half life of 3 years, how much would remain after 3 years? • How much would remain after 6 years?
- Slides: 14