Nuclear Science Merit Badge Howard Matis Lawrence Berkeley

Nuclear Science Merit Badge Howard Matis Lawrence Berkeley National Laboratory Nuclear Science Merit Badge Howard Matis - hsmatis@lbl. gov 1

Radiation gives Superhuman Powers to Spiderman Nuclear Science Merit Badge Howard Matis - hsmatis@lbl. gov 2

Radiation gives Superhuman Powers to The Hulk Nuclear Science Merit Badge Howard Matis - hsmatis@lbl. gov 3

Chernobyl Nuclear Science Merit Badge Howard Matis - hsmatis@lbl. gov 4

Radiation is n n n Plot device for fiction Scary Deadly Life saving Misunderstood Useful Nuclear Science Merit Badge Howard Matis - hsmatis@lbl. gov 5

Radiation Hazard Symbol The symbol is placed on a placard with the word CAUTION or DANGER or GRAVE DANGER centered about it. Under the symbol is the information addressing the types of hazards. Examples are: Radiation Area High Radiation Area Airborne Radioactivity Area Contaminated Area Radioactive Materials Area Nuclear Science Merit Badge Howard Matis - hsmatis@lbl. gov 60° R 1. 5 R 5 R 6

Radiation is Energy n The energy is given off by unstable (radioactive) atoms and some machines. We will be focusing on ionizing radiation and its health effects. Nuclear Science Merit Badge Howard Matis - hsmatis@lbl. gov 7

Atoms Building Blocks of Matter n n All matter is made up of atoms The nucleus is in center n n n almost all of the mass Electrons go around At this scale, electrons are at the edge of town Nuclear Science Merit Badge Howard Matis - hsmatis@lbl. gov 8

What is a Nucleus? n n n Quarks determine if proton or neutron Neutrons Protons determine chemical properties Ratio of neutrons to protons make a nucleus stable or unstable Nuclear Science Merit Badge Howard Matis - hsmatis@lbl. gov 9

Isotopes • Many elements have nuclei with the same number of protons • same name • same chemistry • but different numbers of neutrons • different masses Nuclear Science Merit Badge Howard Matis - hsmatis@lbl. gov 10

Examples - Isotopes • Hydrogen (1 H) • Helium (4 He) (a-particle) 1 proton, 0 neutrons, mass 1 • Deuterium (2 D) 2 protons, 2 neutrons, mass 4 • Helium-3 (3 He) 1 proton, 1 neutron, mass 2 2 protons, 1 neutron, mass 3 • Tritium (31 T) 1 proton, 2 neutrons, mass 3 • Uranium-238 (238 U) 92 protons, 146 neutrons, mass 238 • Uranium-235 (235 U) 92 protons, 143 neutrons, mass 235 Nuclear Science Merit Badge Howard Matis - hsmatis@lbl. gov 11

Types of Radioactivity Six Common Types Alpha Decay • Each type of radiation is ionizing Beta Decay • But different properties Gamma Decay Fission Fusion • affect the hazards they pose • the detection mechanism • shielding Cosmic Rays Nuclear Science Merit Badge Howard Matis - hsmatis@lbl. gov 12

How Does it Decay? n n n Alpha - lose an alpha particle ( - helium nucleus) Beta - emit a beta particle ( - electron or anti-electron) Gamma - emit a gamma ( or photon or light particle) Nuclear Science Merit Badge Howard Matis - hsmatis@lbl. gov 13

Alpha Decay n n n Alpha particle or helium nucleus emitted Nucleus changes mass by four units and charge by two units Common for heavy elements Changes chemical properties Alpha particle easily stopped n 4 x nucleon mass n +2 Charge n Big Nuclear Science Merit Badge Howard Matis - hsmatis@lbl. gov 14

Beta Decay n n n Beta minus - neutron converts to electron and anti-neutrino Beta plus - proton converts to a anti-electron and neutrino Nucleus changes charge but not mass number Changes chemical properties Radiation moderately penetrating n n +1 charge Small electron Nuclear Science Merit Badge Howard Matis - hsmatis@lbl. gov 15

Gamma Decay n Nucleus changes energy level n n Nucleus stays the same n n Emits gamma ray or photon No change in chemical properties Very penetrating n n Almost no size Neutral Nuclear Science Merit Badge Howard Matis - hsmatis@lbl. gov 16
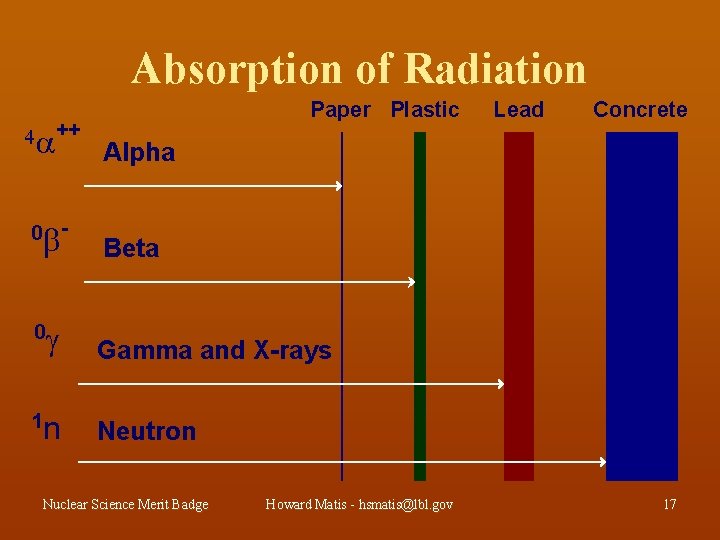
Absorption of Radiation ++ 0 - Lead Concrete Alpha Beta Gamma and X-rays n Neutron 0 1 Paper Plastic Nuclear Science Merit Badge Howard Matis - hsmatis@lbl. gov 17

Fission Sometimes a very heavy nucleus will fall apart before it can emit an alpha particle. The heavy parent nucleus fissions … … into two lighter (radioactive) fission fragment nuclei plus some left over neutrons Fission can release an enormous amount of energy and is utilized in power plants and fission bombs (A-bomb). Nuclear Science Merit Badge Howard Matis - hsmatis@lbl. gov 18

Fusion n n When two nuclei collide and stick together Process that powers the sun and stars All life arises from it Not usually found in every day experience on Earth Component of the H-bomb Nuclear Science Merit Badge Howard Matis - hsmatis@lbl. gov 19

How Unstable Is It? n The “Half-life” describes how quickly Radioactive Material decays away with time. It is the time required for half of the unstable atoms to decay. n Some Examples: n n n Some natural isotopes (like uranium and thorium) have half-lives that are billions of years Since Earth is about 5 billion years old, short lived naturally produced isotopes gone Most medical isotopes (like 99 m. Tc) last only a few days Nuclear Science Merit Badge Howard Matis - hsmatis@lbl. gov 20

Half-Life Experiment Guess the number I am thinking from 1 to 4 Nuclear Science Merit Badge Howard Matis - hsmatis@lbl. gov 21

Some Isotopes & Their Half Lives ISOTOPE HALFLIFE APPLICATIONS 238 U – Uranium billions Natural uranium is comprised of several different isotopes. of years When enriched in the isotope of 235 U, it’s used to power nuclear reactor or nuclear weapons. 14 C – Carbon 5730 y Found in nature from cosmic interactions, used to “carbon date” items and as radiolabel for detection of tumors. 137 Ce – Cesium 30. 2 y Blood irradiators, tumor treatment through external exposure. Also used for industrial radiography. 3 H – Tritium 12. 3 y Labeling biological tracers. 192 Ir – Iridium 74 d Implants or "seeds" for treatment of cancer. Also used for industrial radiography. 99 Mo – 66 h Parent for 99 m. Tc generator. 6 h Brain, heart, liver (gastroenterology), lungs, bones, thyroid, and kidney imaging, regional cerebral blood flow, etc. Molybdenum 99 m. Tc – Technetium Nuclear Science Merit Badge Howard Matis - hsmatis@lbl. gov 22

How do we Measure the Amount of Radiation? Nuclear Science Merit Badge Howard Matis - hsmatis@lbl. gov 23

Radiation Quantities and Units Radiation Absorbed Dose Qty: Dose Unit: rad (Gray) 1 rad = 1000 mrad 1 rad = 100 erg/gram 1 Gy =100 rad Radioactivity Qty: Activity Unit: Curie (Bequerel) 1 Ci = 1000 m. Ci 1 Bq = 1 disintegration/sec 1 Ci = 3. 7 1010 Bq Nuclear Science Merit Badge n e Radiation Risk ntg lent e Ro iva Qty: Dose Equivalent u eq n Unit: rem (Sievert) ma 1 rem = 1000 mrem 1 Sv=100 rem Howard Matis - hsmatis@lbl. gov 24

Convert from Curies to Rad n Curie is the number of decays/s n n 1 Curie = 3. 7 1010 decays/s (exactly) Rad is the absorbed dose or physical dose n Amount of energy deposited in unit mass n n human tissue or other media 1 Rad = 100 erg/g n Often use gray n n 1 J/kg 1 gray = 100 rad Nuclear Science Merit Badge Howard Matis - hsmatis@lbl. gov 25

Need Biological Dose – REM • To convert from rad to rem multiply by appropriate value of Q rad Q = rem • Q is the Quality Factor • Q reflects the damage Q Gamma x-ray 1 Beta 1 Neutron 3 -20 Alpha Nuclear Science Merit Badge 20 Howard Matis - hsmatis@lbl. gov 26

Radiation and Health Does radiation affect you? Nuclear Science Merit Badge Howard Matis - hsmatis@lbl. gov 27

Ionizing Radiation can Damage DNA Ionizing radiation has the ability to ionize* atoms and molecules, possibly altering structure and function. * ionize = produce positive and negative electrical charge Nuclear Science Merit Badge Howard Matis - hsmatis@lbl. gov 28

Alpha Radiation Is Only a Hazard When Inside Your Body (Internal Hazard) Your skin will stop it can’t penetrate skin internal hazard stopped by paper found in soil, radon and other radioactive materials Nuclear Science Merit Badge Howard Matis - hsmatis@lbl. gov 29
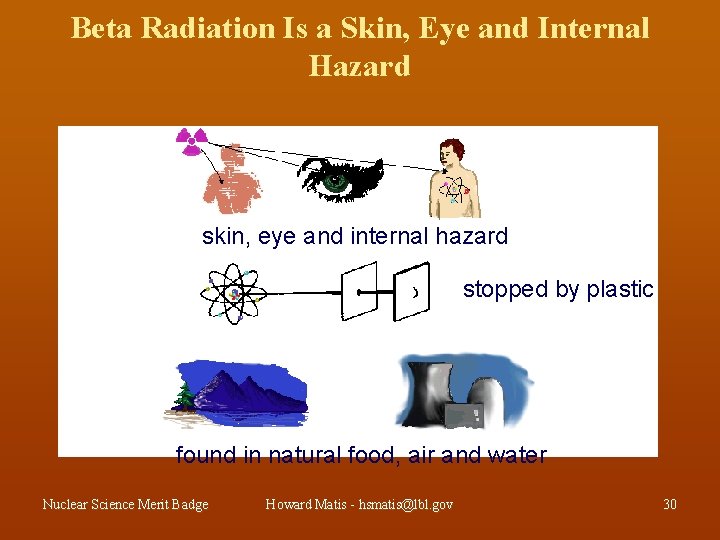
Beta Radiation Is a Skin, Eye and Internal Hazard skin, eye and internal hazard stopped by plastic found in natural food, air and water Nuclear Science Merit Badge Howard Matis - hsmatis@lbl. gov 30

X-ray and Gamma Radiation Are Penetrating Radiation and an External Hazard stopped by lead found in medical uses naturally present in soil and cosmic radiation Nuclear Science Merit Badge Howard Matis - hsmatis@lbl. gov 31

How does Radiation Injure Cells? - • High energy radiation breaks chemical bonds. • This creates free radicals, like those produced by other insults + as well as by normal cellular processes in the body. • The free radicals can change chemicals in the body. • These changes can disrupt cell function and may kill cells. Nuclear Science Merit Badge Howard Matis - hsmatis@lbl. gov 32

Types of Exposure & Health Effects n Acute Dose - Deterministic n n Large radiation dose in a short period of time Large doses may result in observable health effects n Early: Nausea & vomiting n Hair loss, fatigue, & medical complications n Burns and wounds heal slowly Examples: medical exposures and accidental exposure to sealed sources Chronic Dose - Stochastic n n Radiation dose received over a long period of time Body more easily repairs damage from chronic doses Does not usually result in observable effects Examples: Background Radiation and Inhalation Internal Deposition Nuclear Science Merit Badge Howard Matis - hsmatis@lbl. gov 33

n n . At HIGH Doses, We KNOW Radiation Causes Harm High Dose effects seen in n Radium dial painters n Early radiologists n Atomic bomb survivors n Populations near Chernobyl n Medical treatments n Criticality Accidents Cancer n Leukemia (A-bomb data) n Thyroid (Chernobyl data) n Bone and other solid cancers (A-bomb data) Birth defects (A-bomb data) Genetic effects (only animal data) Nuclear Science Merit Badge Howard Matis - hsmatis@lbl. gov 34
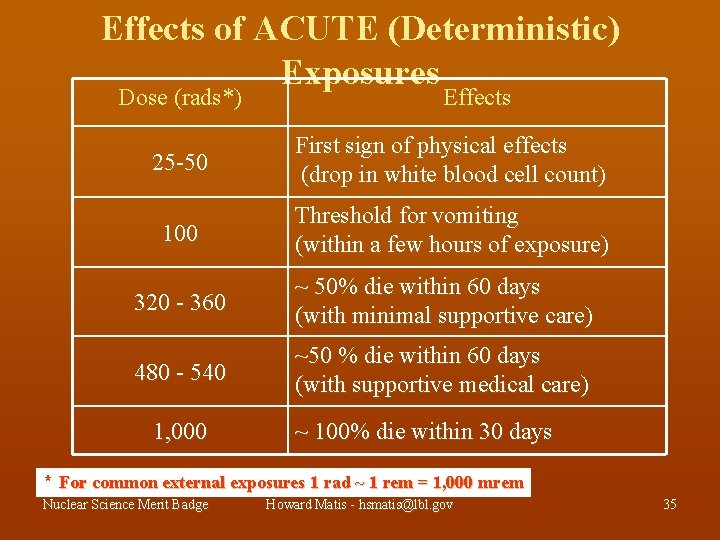
Effects of ACUTE (Deterministic) Exposures Dose (rads*) Effects 25 -50 First sign of physical effects (drop in white blood cell count) 100 Threshold for vomiting (within a few hours of exposure) 320 - 360 ~ 50% die within 60 days (with minimal supportive care) 480 - 540 ~50 % die within 60 days (with supportive medical care) 1, 000 ~ 100% die within 30 days * For common external exposures 1 rad ~ 1 rem = 1, 000 mrem Nuclear Science Merit Badge Howard Matis - hsmatis@lbl. gov 35

At LOW Doses, We PRESUME Radiation Causes Harm n No physical effects have been observed The Bad News: Radiation is a carcinogen and a mutagen The Good News: Radiation is a very weak carcinogen and mutagen! Very Small DOSE = Very Small RISK Nuclear Science Merit Badge Howard Matis - hsmatis@lbl. gov 36
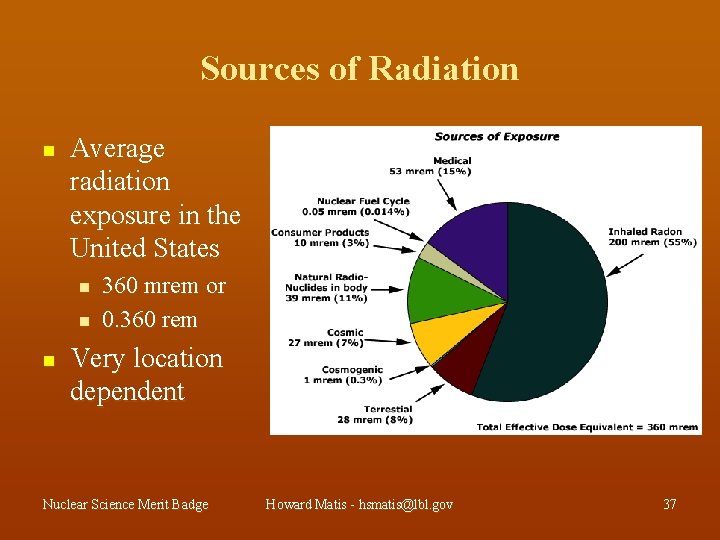
Sources of Radiation n Average radiation exposure in the United States n n n 360 mrem or 0. 360 rem Very location dependent Nuclear Science Merit Badge Howard Matis - hsmatis@lbl. gov 37

Manufactured Sources of Radiation Contribute an Average of 60 mrem/year cigarette smoking - 1300 mrem lung dose medical - 53 mrem building materials - 3. 6 mrem smoke detectors - 0. 0001 mrem Nuclear Science Merit Badge Howard Matis - hsmatis@lbl. gov 38

Risks in Perspective n n n n 1 in 1 million chance of fatality 40 tablespoon peanut butter (aflotoxin) 2 days in New York City (air quality) 3 mrem radiation (cancer) 1 mile on motorcycle (collision) 300 miles in car (collision) 10 charbroiled steaks Smoking 1 cigarette Nuclear Science Merit Badge Howard Matis - hsmatis@lbl. gov 39

ALARA stands for As Low As Reasonably Achievable Reduce radiation dose by using: Time n Distance n Shielding n Nuclear Science Merit Badge Howard Matis - hsmatis@lbl. gov 40

Reduce Time Spend as short as time as necessary to complete the task Nuclear Science Merit Badge Howard Matis - hsmatis@lbl. gov 41

Activity Demonstrate Time – (t) Nuclear Science Merit Badge Howard Matis - hsmatis@lbl. gov 42
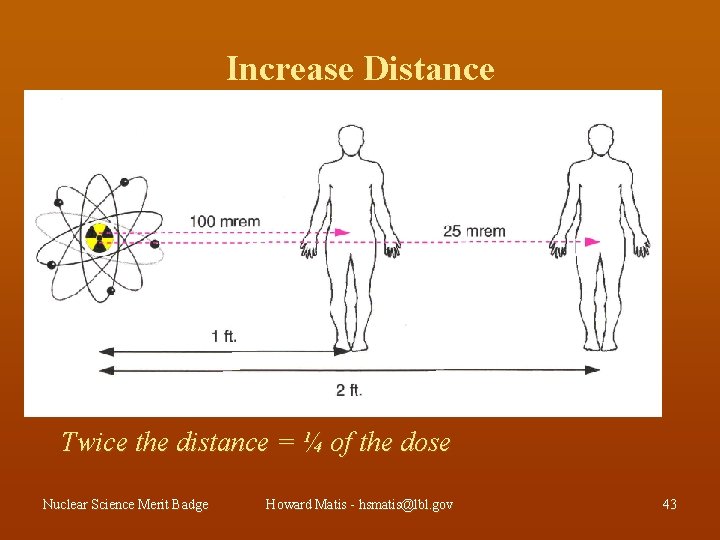
Increase Distance Twice the distance = ¼ of the dose Nuclear Science Merit Badge Howard Matis - hsmatis@lbl. gov 43

Activity Demonstrate Distance – (d) Nuclear Science Merit Badge Howard Matis - hsmatis@lbl. gov 44
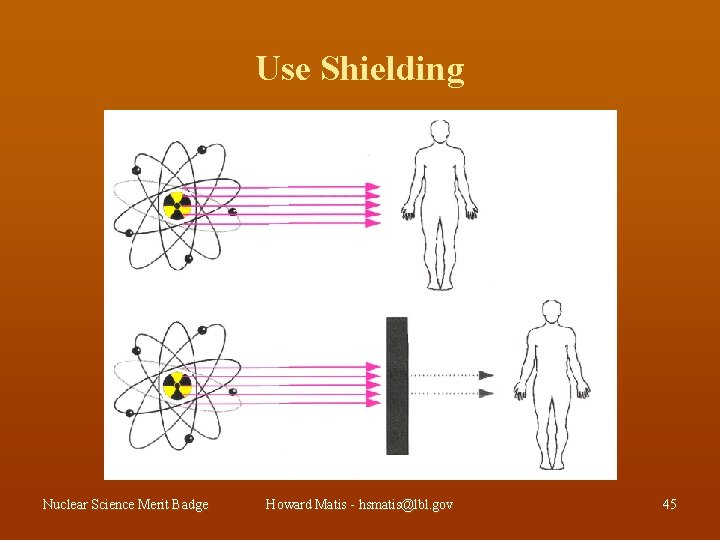
Use Shielding Nuclear Science Merit Badge Howard Matis - hsmatis@lbl. gov 45

Activity Demonstrate Shielding – (s) Nuclear Science Merit Badge Howard Matis - hsmatis@lbl. gov 46

Reactors Glow in the Dark n n n Reactor core emits electrons Electrons move faster than the speed of light in water At that speed they emit blue light n n Cherenkov Radiation Similar to sonic boom or wake of a boat Nuclear Science Merit Badge Howard Matis - hsmatis@lbl. gov 47

Can You Glow in the Dark? n Ingest a very hot radiation source n n n Do not swallow n n n Unless your midriff is exposed Or inject source into your blood Don’t be in direct light n n Beta needed Short half life Usually too faint for sunlight Try it for Halloween? Nuclear Science Merit Badge Howard Matis - hsmatis@lbl. gov 48

Summary n Radiation is part n n n Health effects n n n Our natural environment Technology Known for high doses Unknown for low You deal with it regularly Nuclear Science Merit Badge Howard Matis - hsmatis@lbl. gov 49

The End Nuclear Science Merit Badge Howard Matis - hsmatis@lbl. gov 50
- Slides: 50