Nuclear Reactions Radioactive Decay Unstable nuclei lose energy

Nuclear Reactions

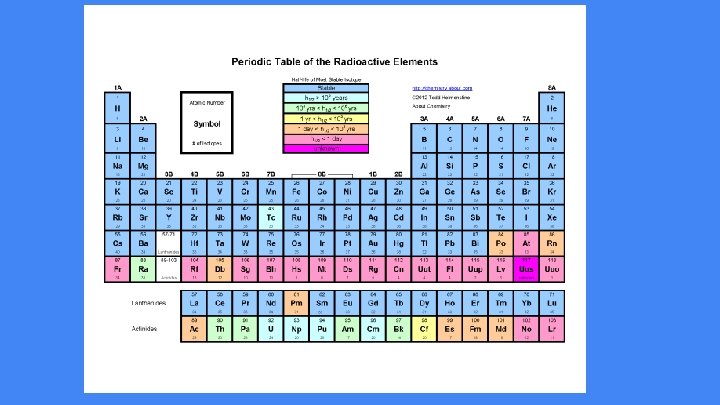
Radioactive Decay Unstable nuclei lose energy by emitting radiation. Occurs in isotopes when the ratio of p+ to n 0 is off causing instability. Decay occurs spontaneously Radioactive Decay attempts to fix the instability of the nucleus

3 Types of Nuclear Reactions Alpha Decay ( Symbol �� is alpha) Beta Decay (Symbol �� is beta) Beta Capture

Alpha Decay An �� particle is released. An �� particle contain 2 protons and 2 neutrons. During. Essentially �� decay: Atomic # ↓ 2 Mass # ↓ 4 a nucleus releases a 4 He 2

Alpha Decay Sample Problems: #1 23892 U → #2 21183 Bi→

Alpha Decay Sample Problems: #1 23892 U → #2 21183 Bi→ 4 He 2 + 23490 Th + 20781 Ti
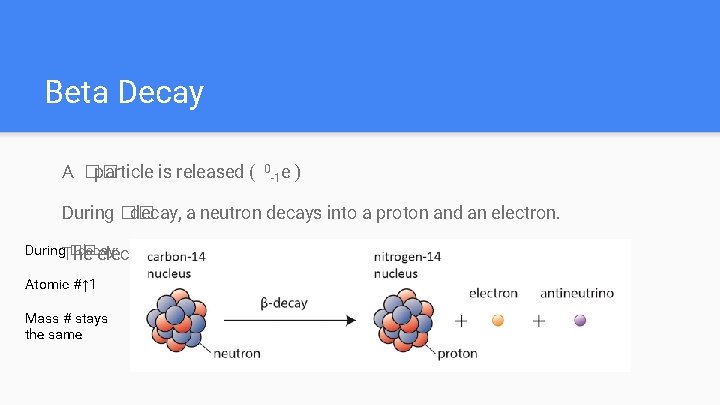
Beta Decay A �� particle is released ( 0 e -1 ) During �� decay, a neutron decays into a proton and an electron. During. The �� decay: electron Atomic #↑ 1 Mass # stays the same is emitted but the proton stays in the nucleus

Beta Decay Examples: #1: 20180 Hg→ #2 6832 Ge→

Beta Decay Examples: #1: 20180 Hg→ #2 6832 Ge→ 0 e -1 + 22790 Th + 6831 Ga

Beta Capture A �� particle combines with a proton to form a neutron During Beta Capture: Atomic # 1 Mass # does not change

Beta Capture Examples: #1. 201 Hg 80 + 0 -1 e #2. 6832 Ge + 0 -1 e

Beta Capture Examples: #1. 201 Hg 80 + 0 -1 e #2. 6832 Ge + 0 -1 e 201 Au 79 68 Ga 31

Law Cons. of Energy & Half Life Law of Conservation of Energy- Energy cannot be created nor destroyed -Energy is conserved Half Life- How long it takes for ½ of a radioactive sample to decay into new elements

Half Life Example: #1. Sr-90 has a half life of 29 years. If I had 10. 0 g of Sr-90, how much would be left after 29 years? After 116 years?

Half Life Example: #1. Sr-90 has a half life of 29 years. If I had 10. 0 g of Sr-90, how much would be left after 29 years? After 116 years? After 29 years? 5. 0 g After 116 years? 1. 25 g

Half Life Example: #2. Rn-222 has a half life of 3. 8 days. How many days would it take for 15. 0 g of Rn-222 to decay to 0. 234 g?

Half Life Example: Rn-222 has a half life of 3. 8 days. How many days would it take for 15. 0 g of Rn -222 to decay to 0. 234 g? 15 g 7. 5 g 3. 75 g 6 half lives = 22. 8 days 1. 88 g 0. 938 g 0. 469 g 0. 234 g

Radioactive Dating- based on the ½ life of C-14 Since all organisms contain Carbon, they all contain a certain percent of C-14 (radioactive). When they die, no new C-14 is absorbed so the C-14 already contained continues to decay 14 C 6 0 e -1 + 147 N By measuring the percent of C-14 and comparing it to the amount in Earth’s

C-14 Example: #1. A fossil was found that is 28, 500 years old and contains 10 g of C-14. How many grams of C-14 did the specimen have when it was alive? Half life of C-14 is 5, 700 years. 28, 500/5, 700 = 5 half lives have passed. Work backwards to get original amount. 10 g → 20 g → 40 g → 80 g → 160 g → 320 g
- Slides: 20