Nuclear Reactions Balancing Nuclear Equations n n Conservation

Nuclear Reactions

Balancing Nuclear Equations n n Conservation of Atomic Number (subscript) Conservation of Atomic Mass (superscript)

Balancing Nuclear Equations 16 N 7 0 e -1 + 16 O 8 Conservation of mass number: 16 = 0 + 16 Conservation of atomic number: 7 = -1 + 8

Natural Transmutation n 1 term on the reactant side n n Original isotope 2 terms on the product side Emitted Particle n New Isotope n Happens all by itself. Not affected by anything in the environment.
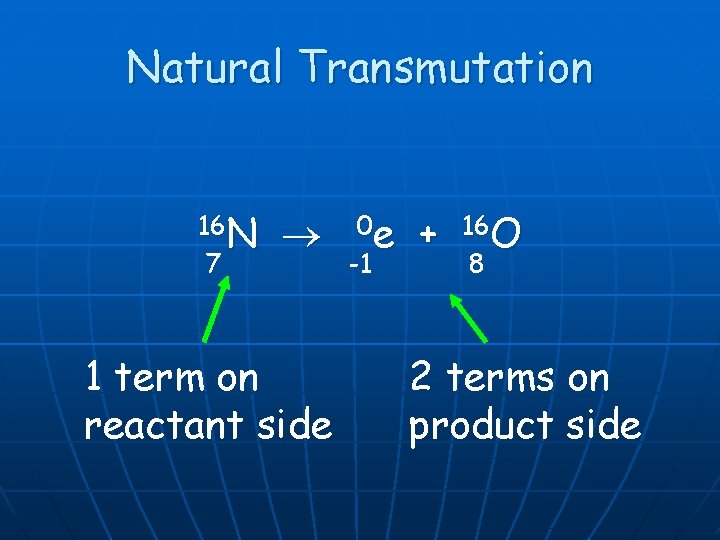
Natural Transmutation 16 N 7 1 term on reactant side 0 e -1 + 16 O 8 2 terms on product side

Artificial Transmutation n n We cause it to happen by smashing particles into one another. 2 terms on the reactant side. Original Isotope n Particle that hit it: neutron, proton, or -particle n n Product side usually has 2 terms.

Artificial Transmutation 27 Al 13 + 4 He 30 P + 1 n 2 Original isotope or target nucleus 15 0 “Bullet”

Bombarding with Protons, n n Protons and -particles have positive charge and mass. They can do some damage when they hit the target nucleus. Protons and -particles have to be accelerated to high speeds to overcome repulsive forces. (The nucleus they are aiming for is also positive. ) Use magnetic and electric fields to accelerate them.

What is an accelerator? n n An accelerator consists of a vacuum chamber, usually a long pipe, surrounded by vacuum pumps, magnets, radiofrequency cavities, high voltage instruments and electronic circuits. Inside the pipe, particles are accelerated to very high speeds & smashed into each other.

Neutron Capture n n n Neutrons don’t have to be accelerated. They’re neutral so they aren’t repelled by a positive nucleus. Don’t need high K. E. to overcome repulsive forces. It’s a good thing – we can’t accelerate neutrons.

Artificial Transmutation 27 Al 13 14 N 7 + 4 He 30 P + 1 n 2 15 0 + 42 He 178 O + 11 H 75 As 33 37 Cl 17 1 n + 24 He 78 Br + 35 0 + 1 n 38 Cl 17 0 All of these equations have 2 reactants!
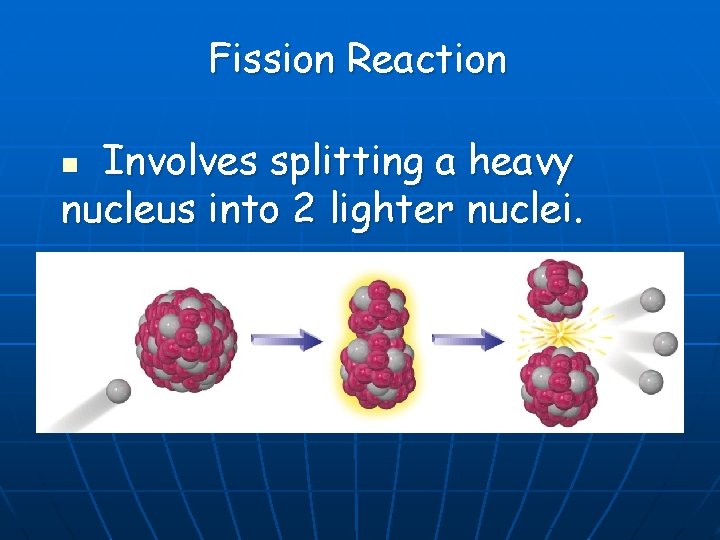
Fission Reaction Involves splitting a heavy nucleus into 2 lighter nuclei. n

Fission n n Involves splitting a heavy nucleus into 2 lighter nuclei. Reactant side has 2 terms: 1 heavy isotope, U-235 or Pu-239 n Bombarding particle – usually a neutron n n Product side has at least 2 terms: 2 medium-weight isotopes n 1 or more neutrons n Huge amount of energy is released. n n Fission = Division

Fission 235 U 92 + 1 n 91 Kr + 142 Ba + 31 n + energy 56 0 235 U 92 1 n + energy + 1 n 72 Zn + 160 Sm + 4 62 0 0 36 30 0 More than 200 different product isotopes from the fission of U-235 have been identified. A small amount of mass is converted to energy by E = mc 2.
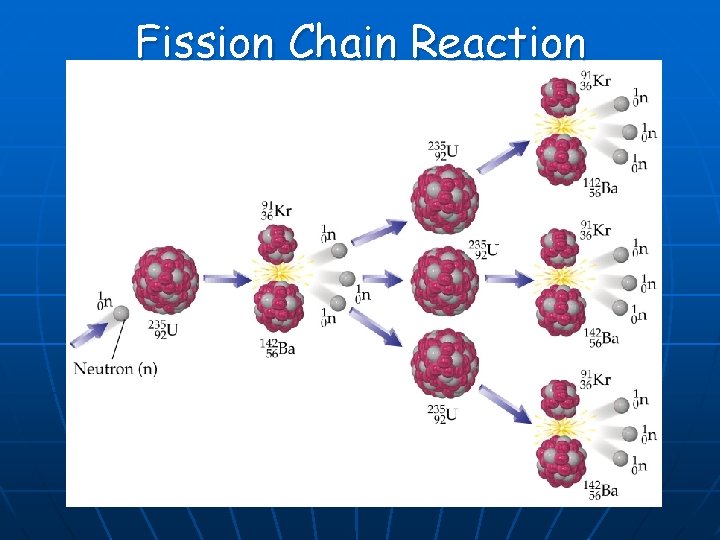
Fission Chain Reaction

Fission Chain Reaction n n Requires a critical mass of fissionable isotope. Controlled – nuclear reactor. Animation of nuclear reactor n Uncontrolled – bomb.

Fusion n n Reactant side has 2 small nuclei – H + H or H + He or He + He. Product side has 1 (still pretty small nucleus) and maybe a particle. Source of sun’s energy. 2 nuclei unite. 2 H 1 + 31 H 42 He + 01 n + energy


Inertial Confinement - Fusion
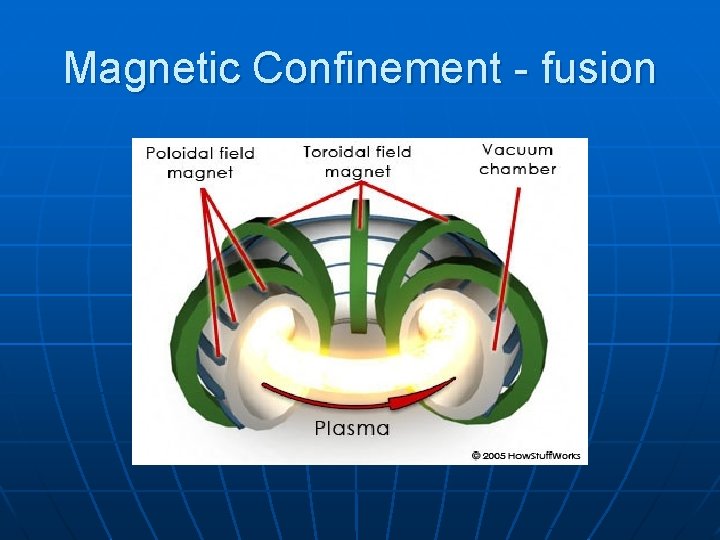
Magnetic Confinement - fusion

Fermi. Lab 4 miles in circumference!

CERN 27 kilometer ring. Particles travel at just below the speed of light. In 10 hrs, the particles make 400 million revolutions of the ring.

SLAC LBL


- Slides: 25