Nuclear Reactions at Low Energies Matej Lipoglavek Joef

![Reaction cross section C. M. S. Energy [Me. V] A. Coc, E. Vangioni Int. Reaction cross section C. M. S. Energy [Me. V] A. Coc, E. Vangioni Int.](https://slidetodoc.com/presentation_image_h/05a1db07c32600a0b42cce3d1ea450fd/image-3.jpg)



![Thick targets 1 H(7 Li, α)4 He Target Ue [ke. V] Stoichiometry Graphite 10. Thick targets 1 H(7 Li, α)4 He Target Ue [ke. V] Stoichiometry Graphite 10.](https://slidetodoc.com/presentation_image_h/05a1db07c32600a0b42cce3d1ea450fd/image-22.jpg)
![Results Target Reaction Ue [ke. V] 7 Li+p 11 B+p 19 F+p adiabatic 0. Results Target Reaction Ue [ke. V] 7 Li+p 11 B+p 19 F+p adiabatic 0.](https://slidetodoc.com/presentation_image_h/05a1db07c32600a0b42cce3d1ea450fd/image-27.jpg)


- Slides: 32

Nuclear Reactions at Low Energies Matej Lipoglavšek Jožef Stefan Institute, Ljubljana, Slovenia Russbach, March 2019

BBN reaction network 7 Be 1 2 1 1 3 He 7 Li 9 4 He 8 4 5 1 H 1 1 n 2 2 d 7 6 3 1 0 3 t Reactions important for the production of the lightest elements (B. D. Fields, Annual Reviews of Nuclear and Particle Science 61, 2011)
![Reaction cross section C M S Energy Me V A Coc E Vangioni Int Reaction cross section C. M. S. Energy [Me. V] A. Coc, E. Vangioni Int.](https://slidetodoc.com/presentation_image_h/05a1db07c32600a0b42cce3d1ea450fd/image-3.jpg)
Reaction cross section C. M. S. Energy [Me. V] A. Coc, E. Vangioni Int. J. Mod. Phys. E 26, 1741002 (2017)

Measurements @ JSI 2 MV Tandem van de Graaf accelerator

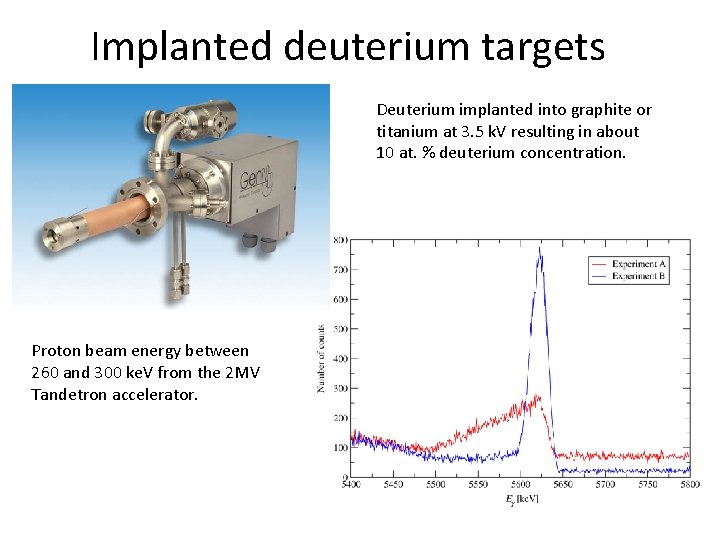
Implanted deuterium targets Deuterium implanted into graphite or titanium at 3. 5 k. V resulting in about 10 at. % deuterium concentration. Proton beam energy between 260 and 300 ke. V from the 2 MV Tandetron accelerator.

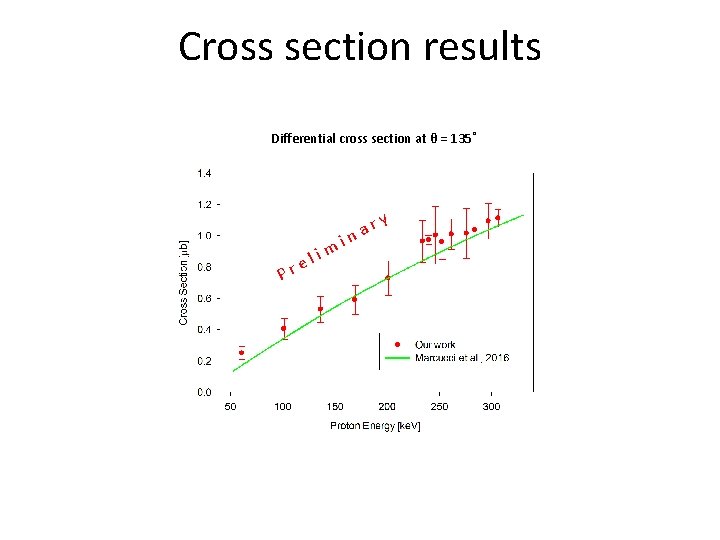
Cross section results Differential cross section at θ = 135˚ y r a n i P r m i l e

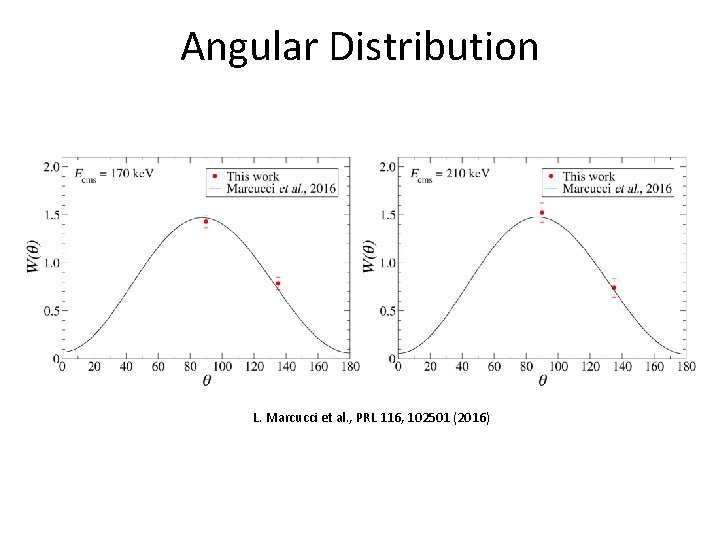
Angular Distribution L. Marcucci et al. , PRL 116, 102501 (2016)

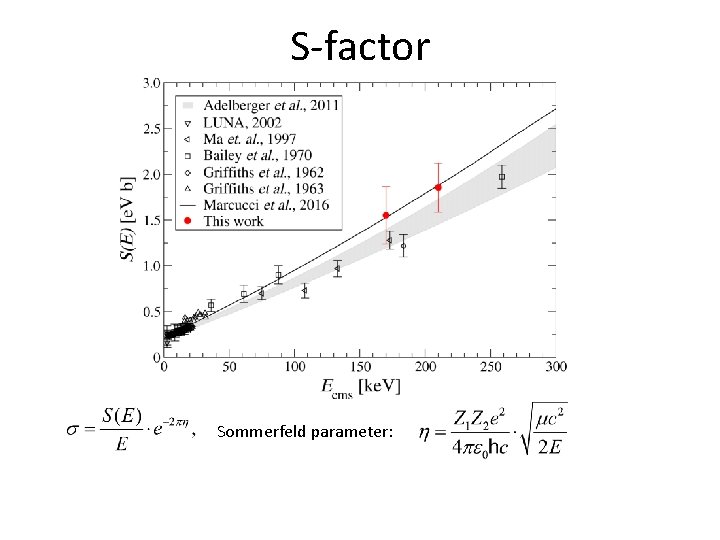
S-factor Sommerfeld parameter:

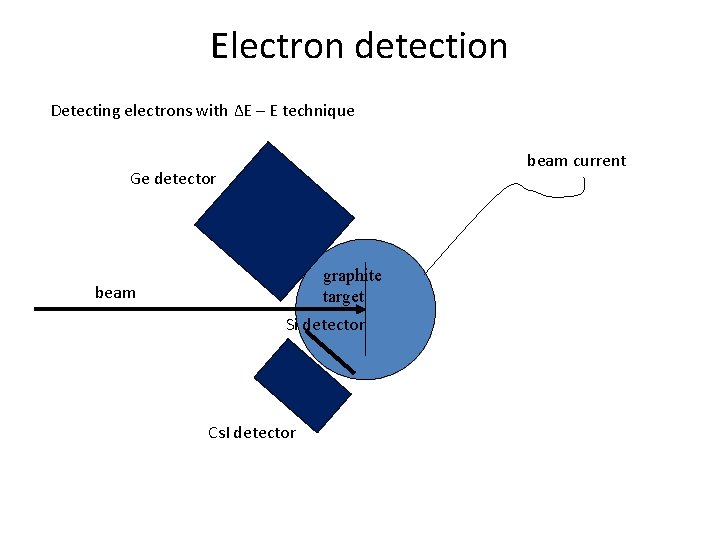
Electron detection Detecting electrons with ΔE – E technique beam current Ge detector graphite target beam Si detector Cs. I detector

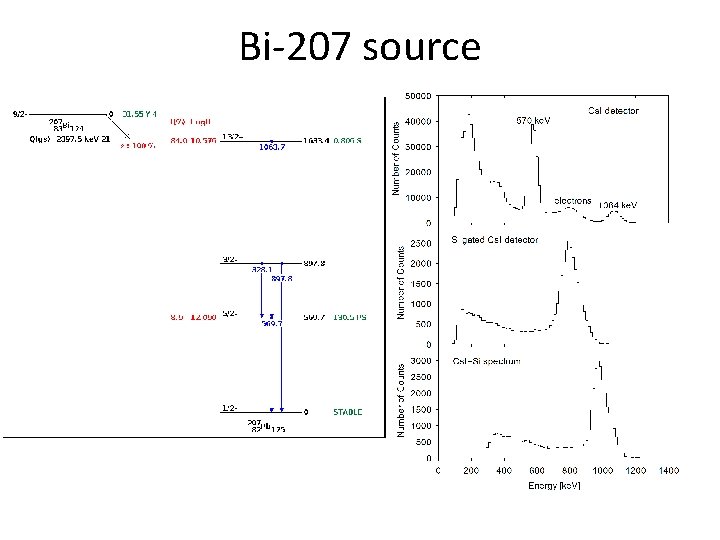
Bi-207 source

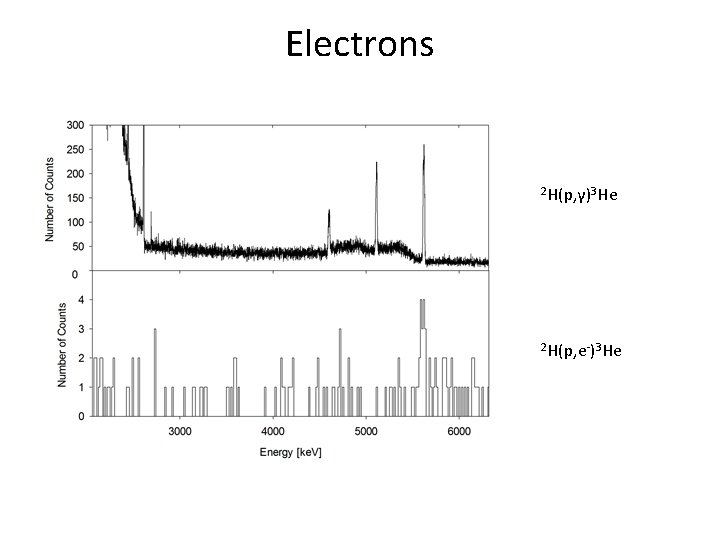
Electrons 2 H(p, γ)3 He 2 H(p, e-)3 He

Results Beam energy at half target thickness: 243 ke. V Cross section: 2 H(p, γ)3 He, σ=1. 0(2) μb 2 H(p, e-)3 He, σ=0. 3(1) nb Our We can only say that in at least 7% of the screened reactions an electron is emitted instead of a γ ray.

Catalysis of nuclear reactions by muons Instead of 2 H(p, γ)3 He Alvarez measured 2 H(p, μ)3 He

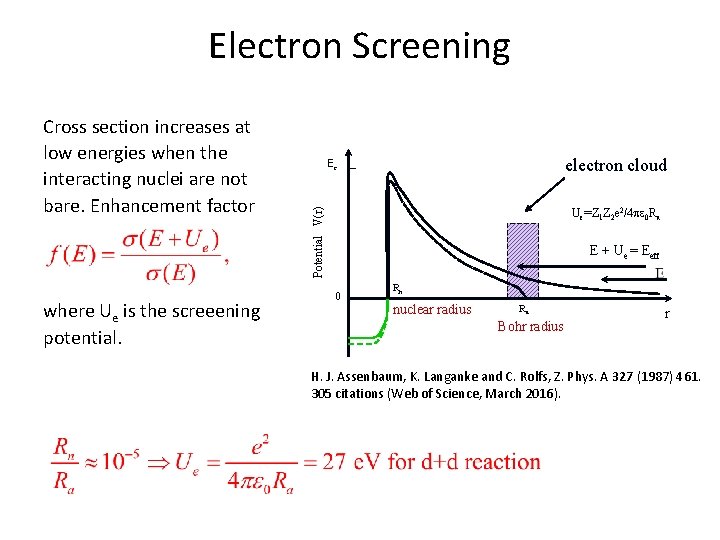
Electron Screening where Ue is the screeening potential. electron cloud Ec Ue=Z 1 Z 2 e 2/4 pe 0 Ra Potential V(r) Cross section increases at low energies when the interacting nuclei are not bare. Enhancement factor E + Ue = Eeff 0 Rn nuclear radius Ra Bohr radius r H. J. Assenbaum, K. Langanke and C. Rolfs, Z. Phys. A 327 (1987) 461. 305 citations (Web of Science, March 2016).

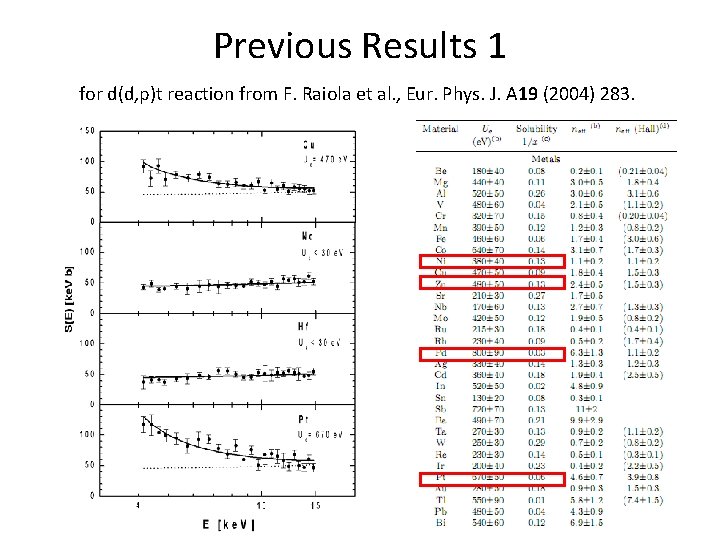
Previous Results 1 for d(d, p)t reaction from F. Raiola et al. , Eur. Phys. J. A 19 (2004) 283.

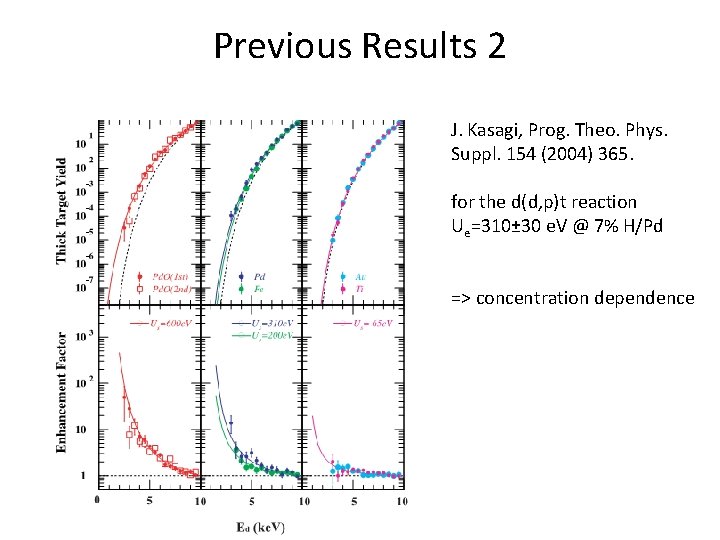
Previous Results 2 J. Kasagi, Prog. Theo. Phys. Suppl. 154 (2004) 365. for the d(d, p)t reaction Ue=310± 30 e. V @ 7% H/Pd => concentration dependence

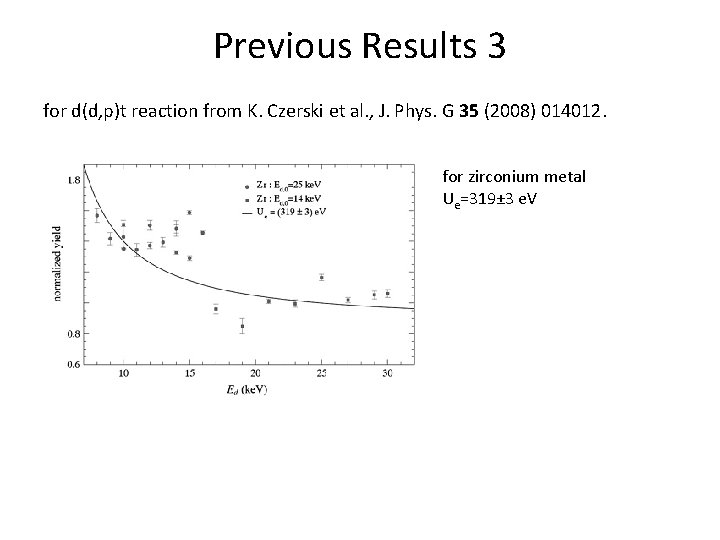
Previous Results 3 for d(d, p)t reaction from K. Czerski et al. , J. Phys. G 35 (2008) 014012. for zirconium metal Ue=319± 3 e. V

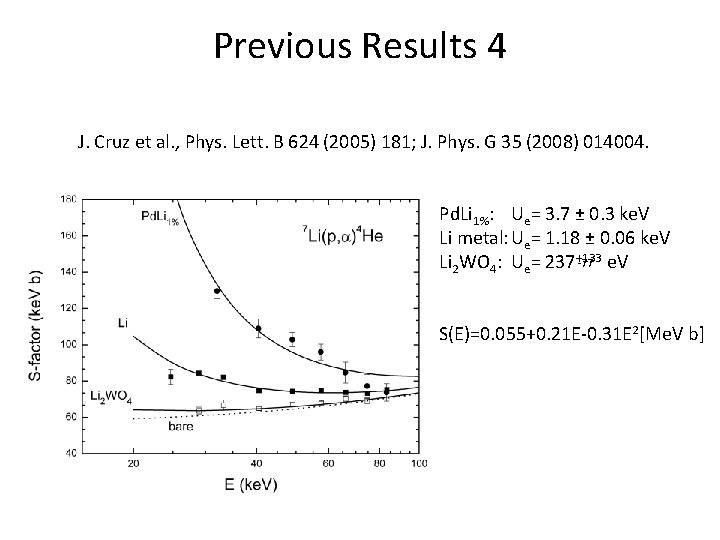
Previous Results 4 J. Cruz et al. , Phys. Lett. B 624 (2005) 181; J. Phys. G 35 (2008) 014004. Pd. Li 1%: Ue= 3. 7 ± 0. 3 ke. V Li metal: Ue= 1. 18 ± 0. 06 ke. V -77 e. V Li 2 WO 4: Ue= 237+133 S(E)=0. 055+0. 21 E-0. 31 E 2[Me. V b]

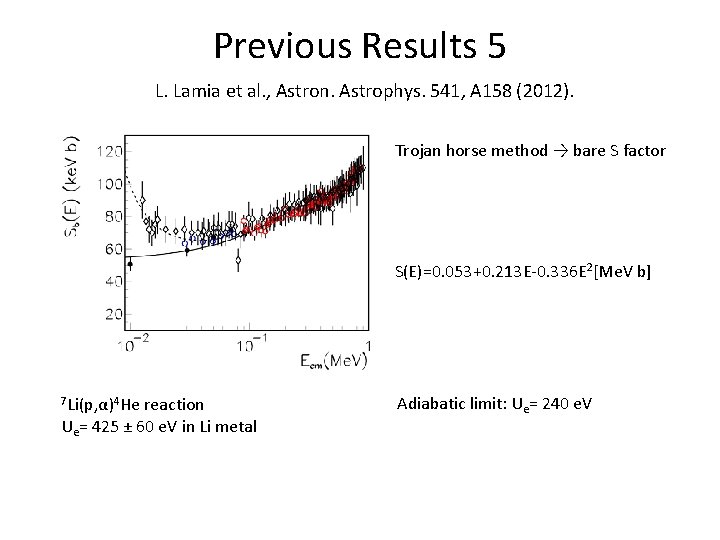
Previous Results 5 L. Lamia et al. , Astron. Astrophys. 541, A 158 (2012). Trojan horse method → bare S factor S(E)=0. 053+0. 213 E-0. 336 E 2[Me. V b] 7 Li(p, α)4 He reaction Ue= 425 ± 60 e. V in Li metal Adiabatic limit: Ue= 240 e. V

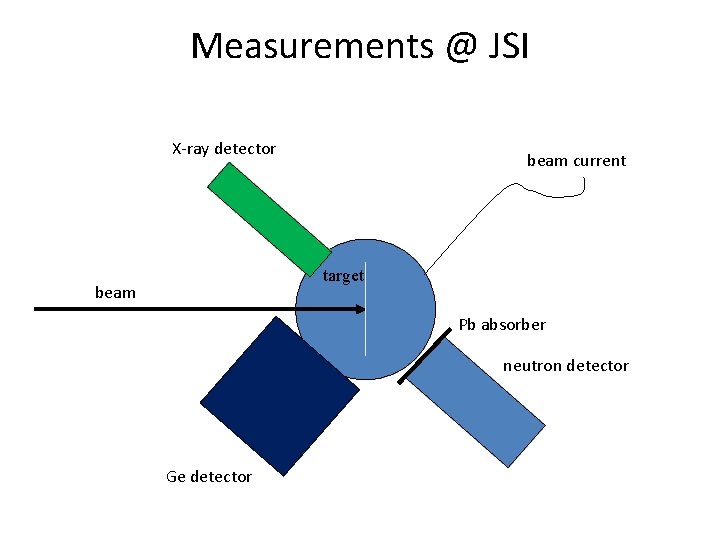
Measurements @ JSI X-ray detector beam current target beam Pb absorber neutron detector Ge detector

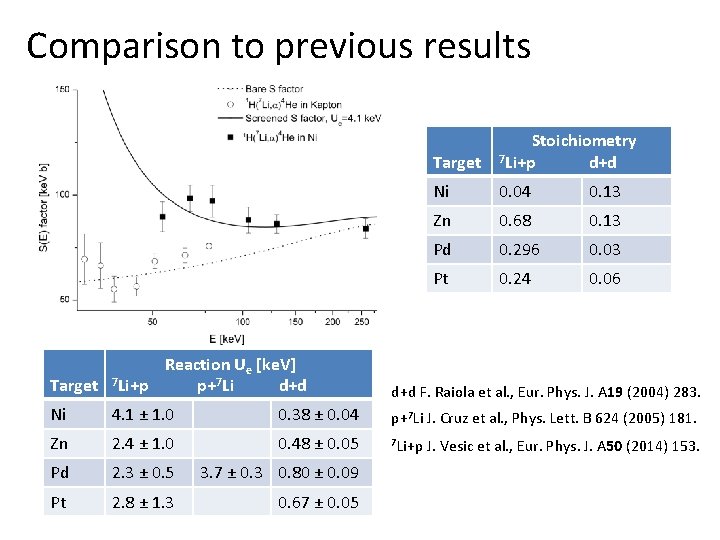
Comparison to previous results Target Stoichiometry 7 Li+p d+d Ni 0. 04 0. 13 Zn 0. 68 0. 13 Pd 0. 296 0. 03 Pt 0. 24 0. 06 Reaction Ue [ke. V] Target 7 Li+p p+7 Li d+d F. Raiola et al. , Eur. Phys. J. A 19 (2004) 283. Ni 4. 1 ± 1. 0 0. 38 ± 0. 04 p+7 Li J. Cruz et al. , Phys. Lett. B 624 (2005) 181. Zn 2. 4 ± 1. 0 0. 48 ± 0. 05 7 Li+p J. Vesic et al. , Eur. Phys. J. A 50 (2014) 153. Pd 2. 3 ± 0. 5 3. 7 ± 0. 3 0. 80 ± 0. 09 Pt 2. 8 ± 1. 3 0. 67 ± 0. 05
![Thick targets 1 H7 Li α4 He Target Ue ke V Stoichiometry Graphite 10 Thick targets 1 H(7 Li, α)4 He Target Ue [ke. V] Stoichiometry Graphite 10.](https://slidetodoc.com/presentation_image_h/05a1db07c32600a0b42cce3d1ea450fd/image-22.jpg)
Thick targets 1 H(7 Li, α)4 He Target Ue [ke. V] Stoichiometry Graphite 10. 3 ± 0. 4 0. 059± 0. 003 Pd 3. 6 ± 0. 7 0. 21± 0. 01 Ti. H 3. 9 ± 0. 4 1. 03± 0. 04 W 5. 9 ± 0. 9 0. 042± 0. 003 Adiabatic limit: Ue= 0. 24 ke. V

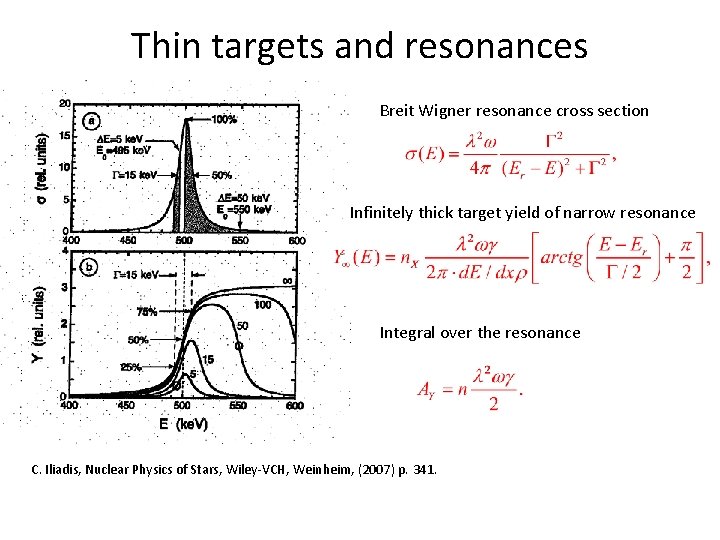
Thin targets and resonances Breit Wigner resonance cross section Infinitely thick target yield of narrow resonance Integral over the resonance C. Iliadis, Nuclear Physics of Stars, Wiley-VCH, Weinheim, (2007) p. 341.

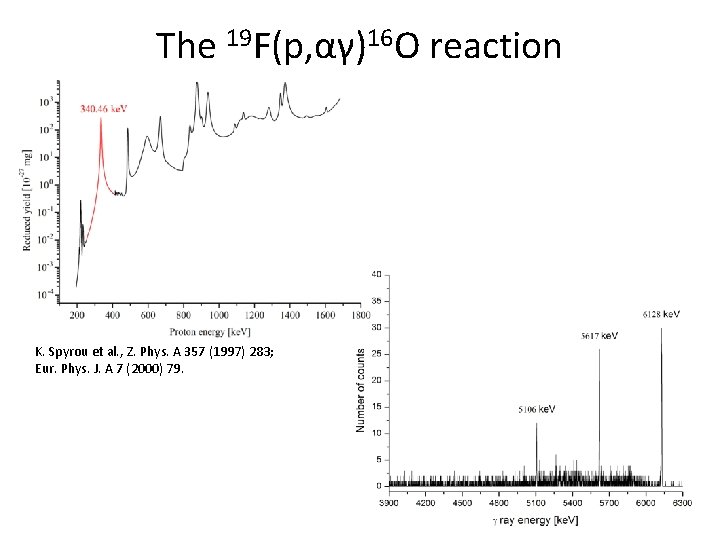
The 19 F(p, αγ)16 O reaction K. Spyrou et al. , Z. Phys. A 357 (1997) 283; Eur. Phys. J. A 7 (2000) 79.

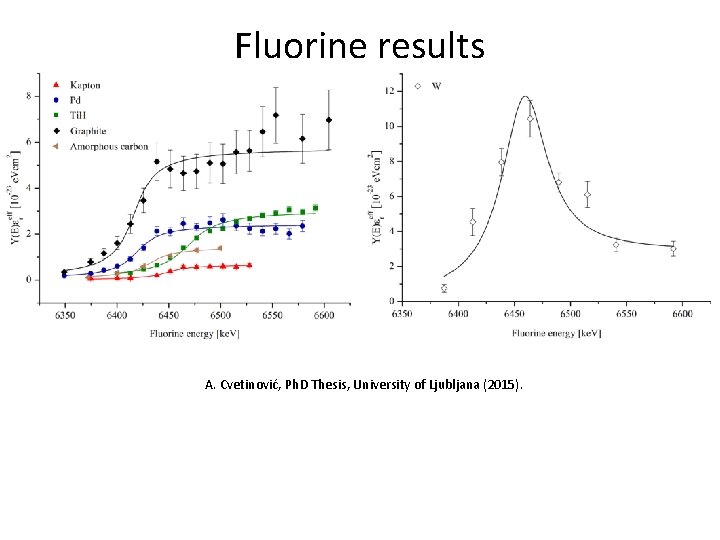
Fluorine results A. Cvetinović, Ph. D Thesis, University of Ljubljana (2015).

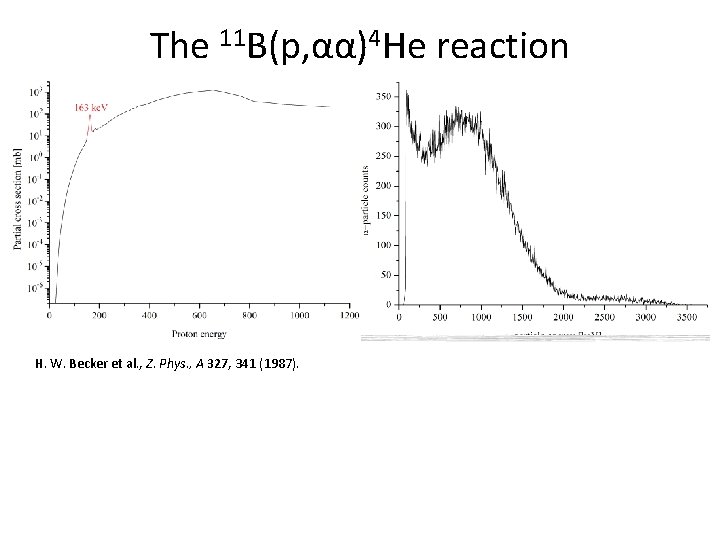
The 11 B(p, αα)4 He reaction H. W. Becker et al. , Z. Phys. , A 327, 341 (1987).
![Results Target Reaction Ue ke V 7 Lip 11 Bp 19 Fp adiabatic 0 Results Target Reaction Ue [ke. V] 7 Li+p 11 B+p 19 F+p adiabatic 0.](https://slidetodoc.com/presentation_image_h/05a1db07c32600a0b42cce3d1ea450fd/image-27.jpg)
Results Target Reaction Ue [ke. V] 7 Li+p 11 B+p 19 F+p adiabatic 0. 24 0. 68 2. 19 Graphite 10. 3 ± 0. 4 32 ± 4 115 ± 8 Pd 3. 6 ± 0. 7 8. 0 ± 1. 9 63 ± 6 Ti. H 3. 9 ± 0. 4 6. 7 ± 1. 8 62 ± 6 W 5. 9 ± 0. 9 - 75 ± 15 Graphite/ adiabatic 42. 9 ± 1. 7 47. 1 ± 5. 9 52. 5 ± 3. 6 A. Cvetinović, Phys. Rev. C 92 (2015) 065801.

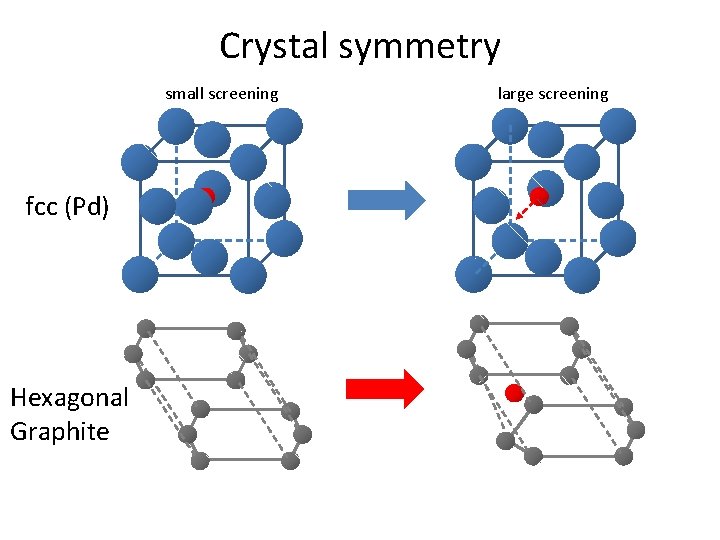
Crystal symmetry small screening large screening fcc (Pd) Hexagonal Graphite

Hydrogen molecular ion

Hydrogen molecular ion

Measurement setup In addition to 2 H(p, γ)3 He we looked for electrons from the 2 H(p, e-)3 He reaction Proton beam energy: 260 ke. V, intensity: 7 μA, 8 days Target: graphite implanted with deuterium at 3 ke. V once a day for ½ hour at 0. 8 m. A resulted in 8 at. % of deuterium per carbon atom down to a depth of about 270 nm Cs. I-Si timestamped Expectations:

Conclusions • During the electron screening process the electrons come close to the nucleus. • A substantial number of fusion reactions in the Sun proceeds with electron emission. • Electron screening could help in lithium abundance problem. • Different screening potentials are due to different proportions of target nuclei on active and inactive sites. • For stellar plasma we really need to understand what happens in the laboratory experiments.