Nuclear Reactions Alpha Beta and Gamma Decay The

Nuclear Reactions Alpha, Beta, and Gamma Decay
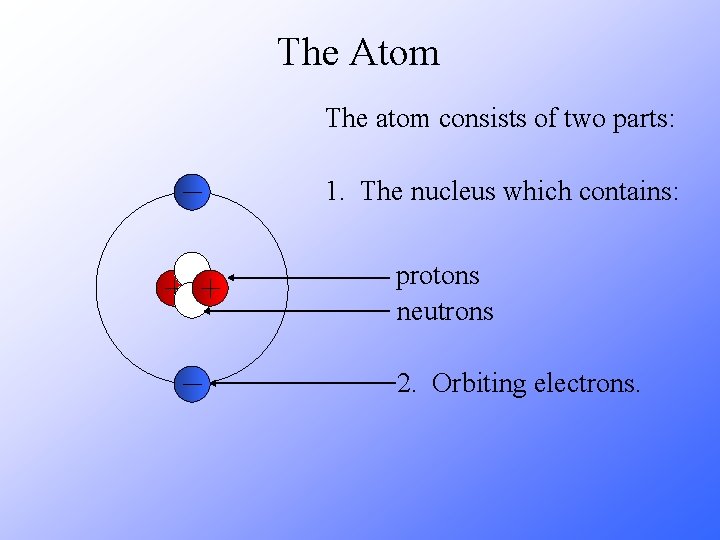
The Atom The atom consists of two parts: 1. The nucleus which contains: protons neutrons 2. Orbiting electrons.
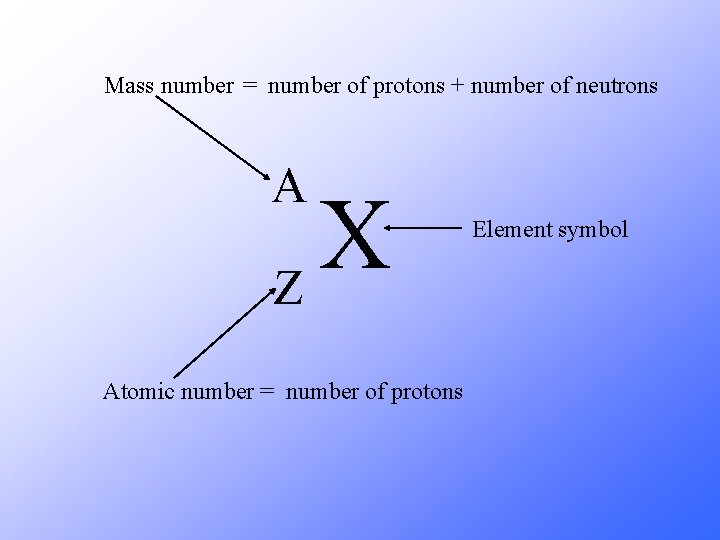
Mass number = number of protons + number of neutrons A X Z Atomic number = number of protons Element symbol

A X Z A = number of protons + number of neutrons Z = number of protons A – Z = number of neutrons Number of neutrons = Mass Number – Atomic Number

There are many types of uranium: 235 238 A A Z Z Number of protons Number of neutrons U 92

There are many types of uranium: 238 235 U 92 A 235 A 238 Z 92 Number of protons 92 Number of neutrons 143 Number of neutrons 146 Isotopes of any particular element contain the same number of protons, but different numbers of neutrons.

Most of the isotopes which occur naturally are stable. A few naturally occurring isotopes and all of the manmade isotopes are unstable. Unstable isotopes can become stable by releasing different types of particles. This process is called radioactive decay and the elements which undergo this process are called radioisotopes/radionuclides.

Radioactive Decay Radioactive decay results in the emission of either: • an alpha particle (a), • a beta particle (b), • or a gamma ray(g).
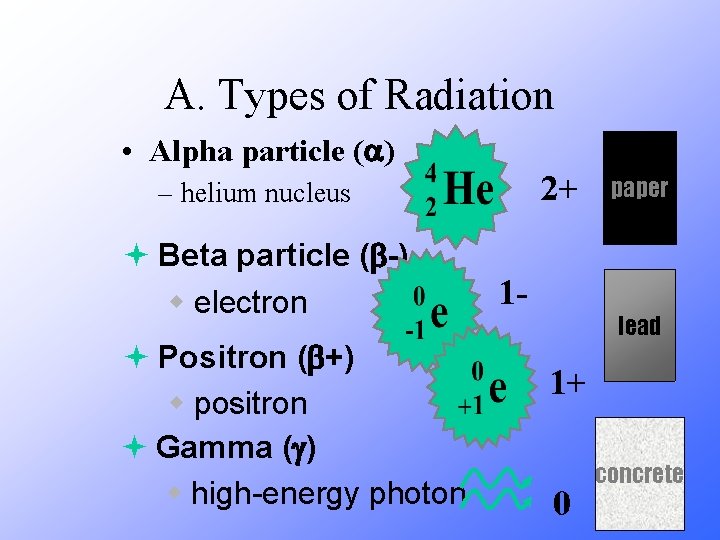
A. Types of Radiation • Alpha particle ( ) 2+ – helium nucleus ª Beta particle ( -) w electron ª Positron ( +) w positron ª Gamma ( ) w high-energy photon 1 - paper lead 1+ 0 concrete

Alpha Decay An alpha particle is identical to that of a helium nucleus. It contains two protons and two neutrons.

Alpha Decay A X Z A - 4 4 Y He + Z - 2 2 unstable atom alpha particle more stable atom
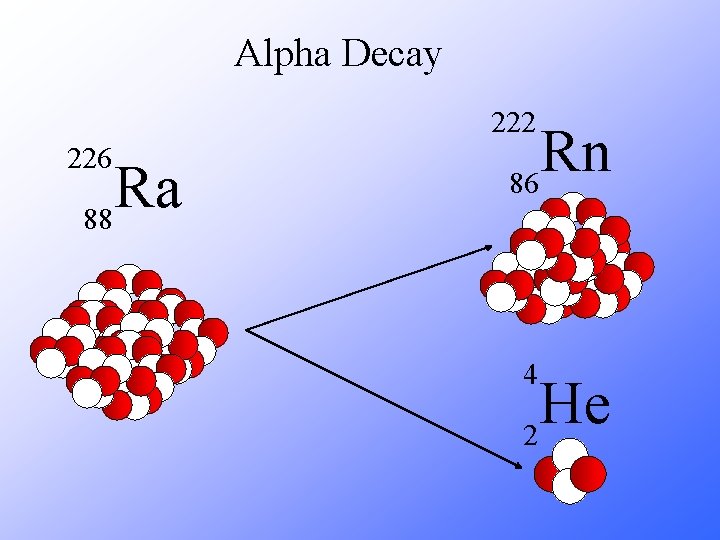
Alpha Decay 222 226 Ra 88 Rn 86 4 He 2

Alpha Decay A A - 4 4 226 222 4 X Z Ra 88 Y + Z - 2 Rn + 86 He 2

Alpha Decay 222 A 222 218 Rn 86 4 Y He + Z 2 Po + 84 4 He 2

Alpha Decay A 230 4 X Z U 92 Th He + 90 2 Th He + 90 2

Alpha Decay 230 A 4 230 226 4 Th 90 Y He + Z 2 Ra He + 88 2

Alpha Decay A 214 4 218 214 4 X Z Po 84 Pb He + 82 2 Pb He + 82 2

Beta Decay A beta particle is a fast moving electron which is emitted from the nucleus of an atom undergoing radioactive decay. Beta decay occurs when a neutron changes into a proton and an electron.

Beta Decay As a result of beta decay, the nucleus has one less neutron, but one extra proton. The atomic number, Z, increases by 1 and the mass number, A, stays the same.
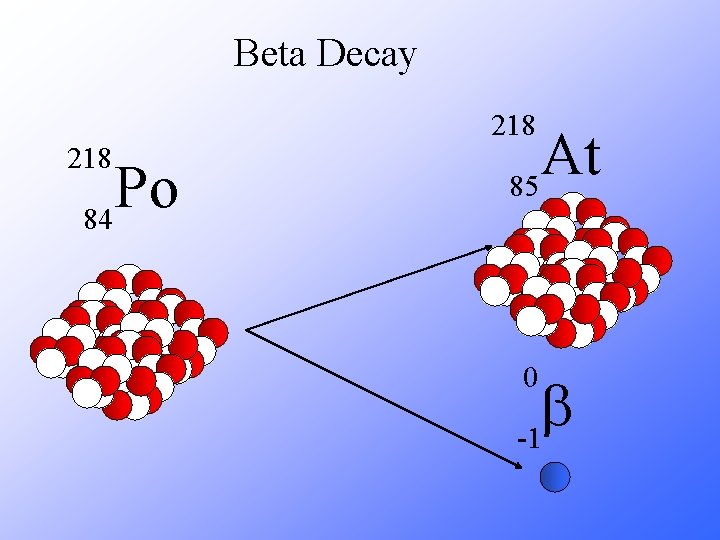
Beta Decay 218 Po 84 At 85 0 b -1

Beta Decay A X Z 218 Po 84 A Y + Z + 1 218 At + 85 0 b -1

Beta Decay 234 Th 90 A Y + Z 234 Pa + 91 0 b -1

Beta Decay A 210 210 X Z Tl 81 Pb + 82 0 b -1

Beta Decay 210 Bi 83 A Y + Z 210 Po + 84 0 b -1

Beta Decay A 214 214 X Z Pb 82 Bi + 83 0 b -1

Gamma Decay Gamma rays are not charged particles like a and b particles. Gamma rays are electromagnetic radiation with high frequency. When atoms decay by emitting a or b particles to form a new atom, the nuclei of the new atom formed may still have too much energy to be completely stable. This excess energy is emitted as gamma rays (gamma ray photons have energies of ~ 1 x 10 -12 J).
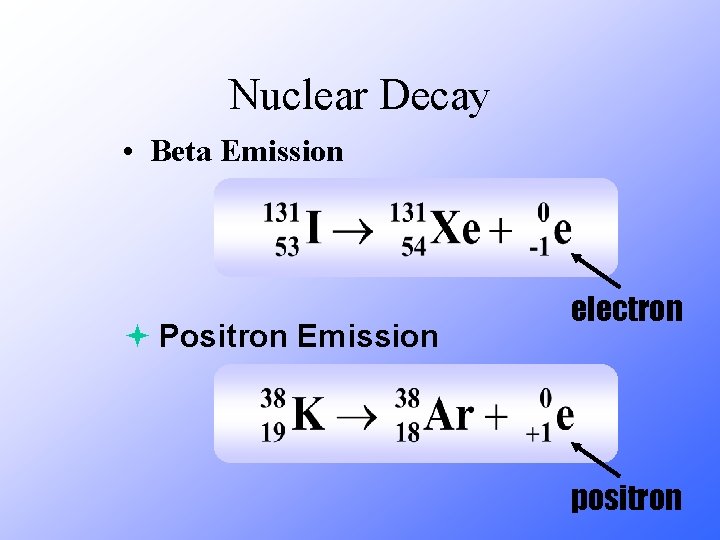
Nuclear Decay • Beta Emission ª Positron Emission electron positron

Nuclear Decay • Electron Capture electron ª Gamma Emission w Usually follows other types of decay. w Transmutation w One element becomes another.
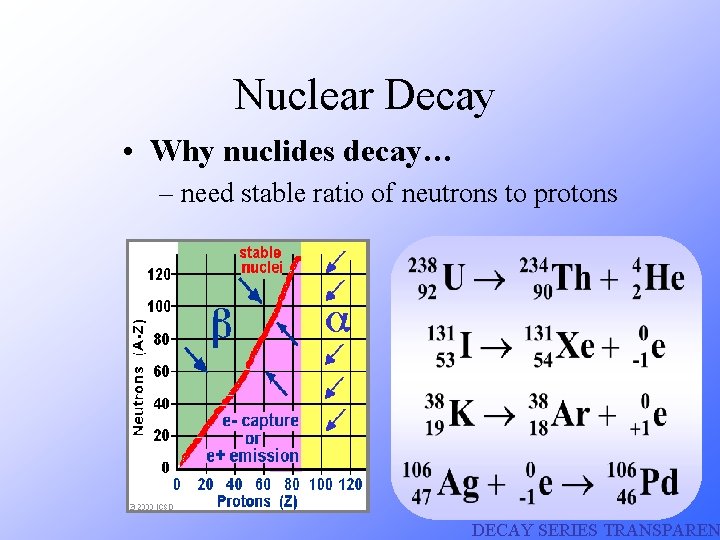
Nuclear Decay • Why nuclides decay… – need stable ratio of neutrons to protons DECAY SERIES TRANSPAREN

Half-Life • is the required for half time Half-life of a radioisotope’s nuclei to decay into its products. # of ½ lives % Remaining • For any radioisotope, 0 1 100% 50% 2 3 4 5 6 25% 12. 5% 6. 25% 3. 125% 1. 5625%
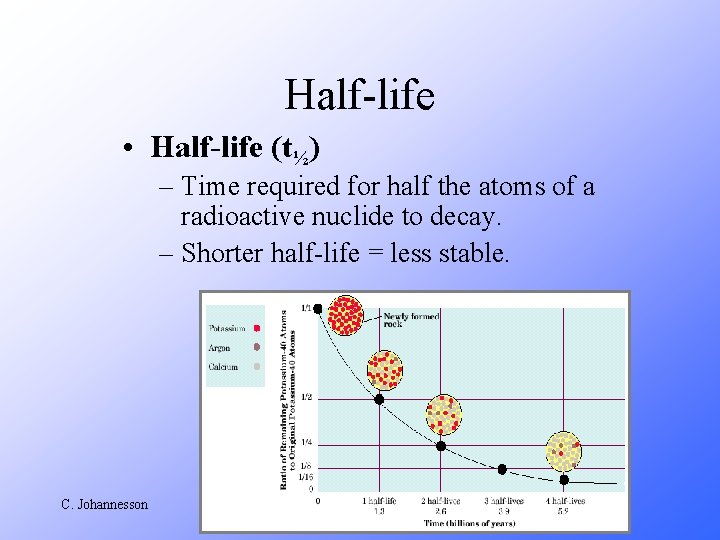
Half-life • Half-life (t½) – Time required for half the atoms of a radioactive nuclide to decay. – Shorter half-life = less stable. C. Johannesson
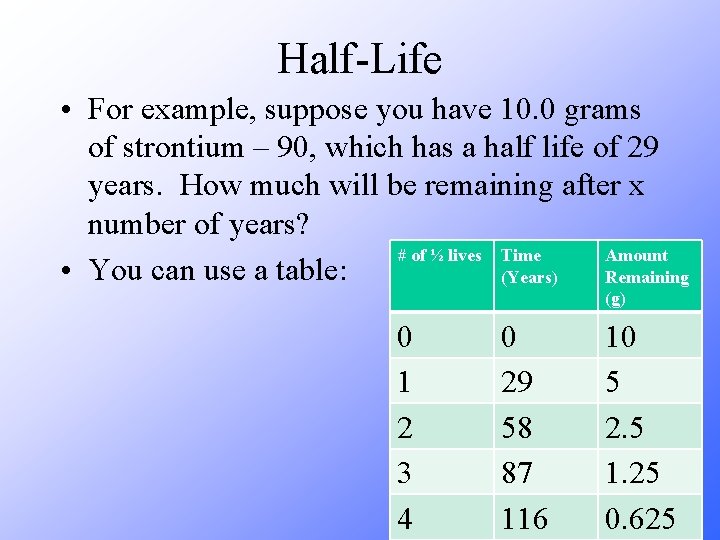
Half-Life • For example, suppose you have 10. 0 grams of strontium – 90, which has a half life of 29 years. How much will be remaining after x number of years? # of ½ lives Time Amount • You can use a table: (Years) Remaining (g) 0 1 2 3 4 0 29 58 87 116 10 5 2. 5 1. 25 0. 625
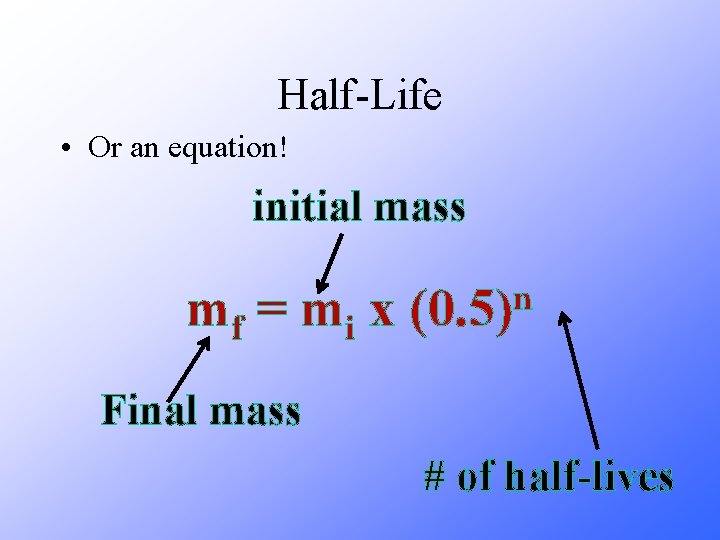
Half-Life • Or an equation! initial mass mf = m i x n (0. 5) Final mass # of half-lives

Half-life ª Fluorine-21 has a half-life of 5. 0 seconds. If you start with 25 g of fluorine-21, how many grams would remain after 60. 0 s? GIVEN: WORK: t½ = 5. 0 s mf = mi (½)n mi = 25 g mf = (25 g)(0. 5)12 mf = ? mf = 0. 0061 g total time = 60. 0 s n = 60. 0 s ÷ 5. 0 s =12 C. Johannesson
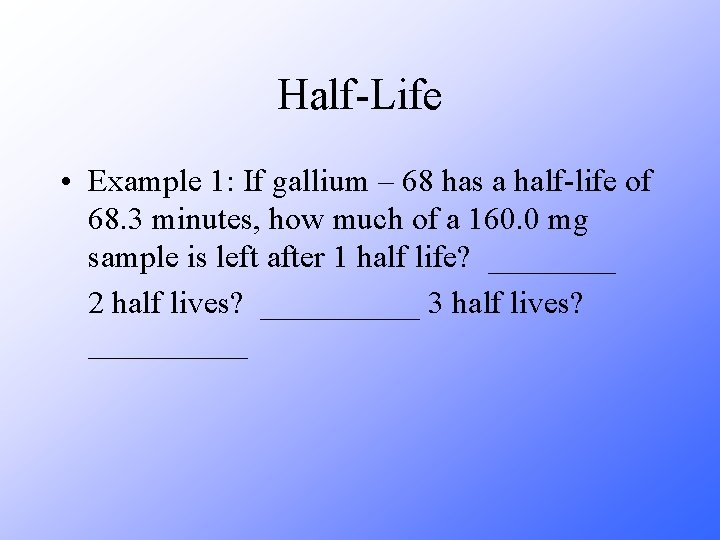
Half-Life • Example 1: If gallium – 68 has a half-life of 68. 3 minutes, how much of a 160. 0 mg sample is left after 1 half life? ____ 2 half lives? _____ 3 half lives? _____

Half-Life • Example 2: Cobalt – 60, with a half-life of 5 years, is used in cancer radiation treatments. If a hospital purchases a supply of 30. 0 g, how much would be left after 15 years? _______
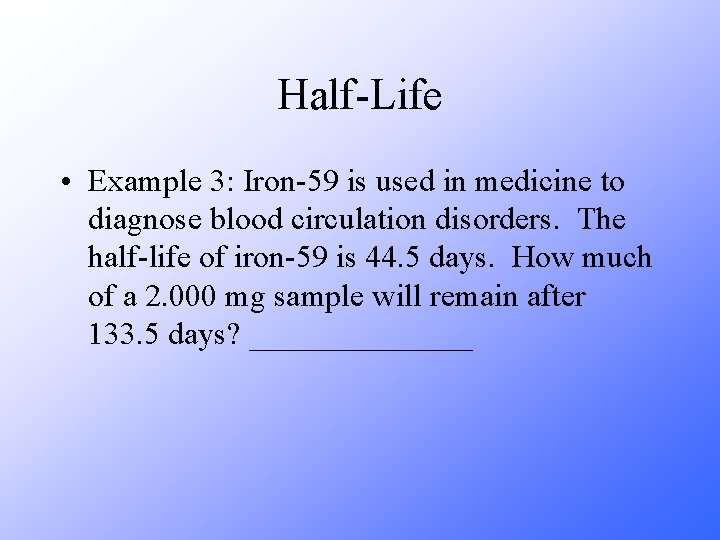
Half-Life • Example 3: Iron-59 is used in medicine to diagnose blood circulation disorders. The half-life of iron-59 is 44. 5 days. How much of a 2. 000 mg sample will remain after 133. 5 days? _______

Half-Life • Example 4: The half-life of polonium-218 is 3. 0 minutes. If you start with 20. 0 g, how long will it take before only 1. 25 g remains? _______

Half-Life • Example 5: A sample initially contains 150. 0 mg of radon-222. After 11. 4 days, the sample contains 18. 75 mg of radon-222. Calculate the half-life.

Nuclear Fission and Fusion

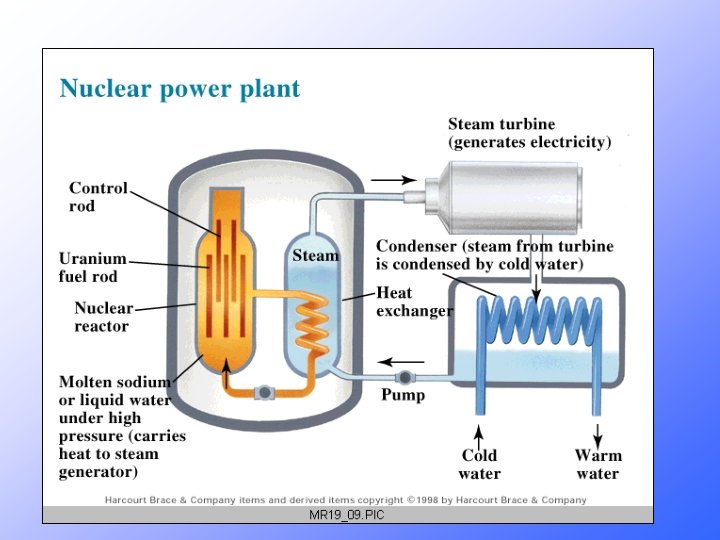
Nuclear power • Power can be obtained two ways. • Fission Splitting atoms • Get energy if the nucleus is big. • The smaller ones are more stable. • What we do in nuclear reactors. • Fusion Joining atoms • Get energy if the nuclei are small. • The larger one is more stable. • This is how the sun works.

NUCLEAR FISSION A reaction in which an atomic nucleus of a radioactive element splits by bombardment from an external source, with simultaneous release of large amounts of energy, used for electric power generation


Nuclear Fission is the splitting of atoms These are usually very large, so that they are not as stable Fission chain has three general steps: 1. Initiation. Reaction of a single atom starts the chain (e. g. , 235 U + neutron) 2. Propagation. 236 U fission releases neutrons that initiate other fissions 3. Termination.

Nuclear Fission • A very heavy nucleus splits into more stable nuclei of intermediate mass. • The mass of the products is less than the mass of the reactants. • Missing mass is converted to energy
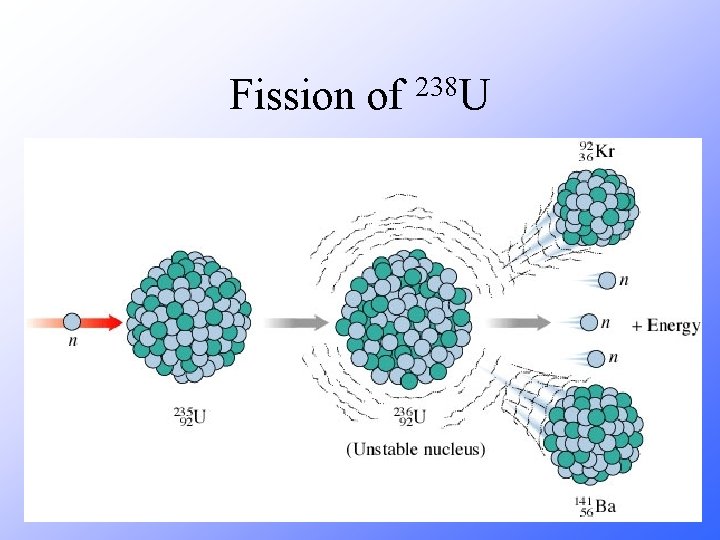
238 Fission of U
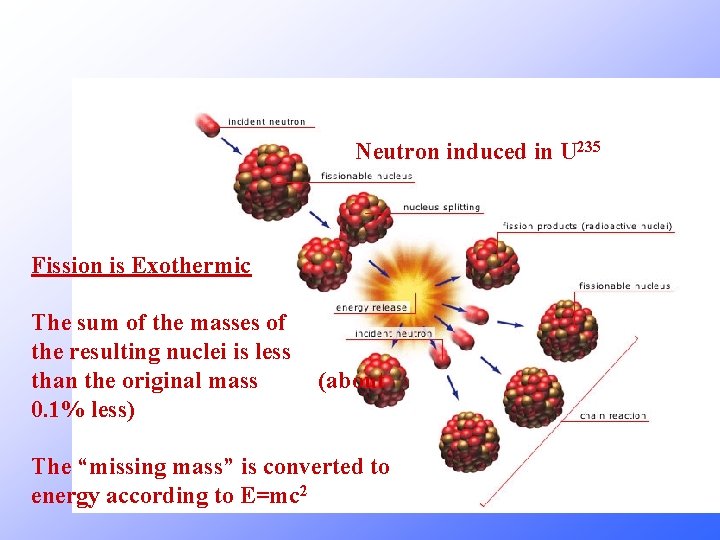
Neutron induced in U 235 Fission is Exothermic The sum of the masses of the resulting nuclei is less than the original mass 0. 1% less) (about The “missing mass” is converted to energy according to E=mc 2

Neutrons may: 1 - Cause another fission by colliding with a U 235 nucleus • Creates two smaller nuclides and free neutrons • The free neutrons potentially collide with nearby U 235 nuclei • May cause the nuclide to split as well Each split (fission) is accompanied by a large quantity of E-N-E-R-G-Y 2 - Be absorbed in other material 3 - Lost in the system If sufficient neutrons are present, we may achieve a chain reaction

Uranium Isotopes Naturally occurring Uranium contains two major isotopes l Uranium-238 (99. 3%) l Uranium-235 (0. 7%) l As it turns out the only isotope of Uranium that undergoes fission is Uranium-235 l

235 U l l l l 235 U 92 Fission + 10 n 23692 U* and 10 -14 seconds later. . . 236 U* 92 Kr + 141 Ba + 3 1 n + ENERGY 92 36 56 0 50 possible sets of fission products (sum of atomic numbers = 92) 3 neutrons released for ONE 23592 U each neutron can split another 23592 U CHAIN REACTION POSSIBLE If amount of 23592 U is sufficient (CRITICAL MASS) then the number of neutrons generated is high enough to result in a nuclear explosion )
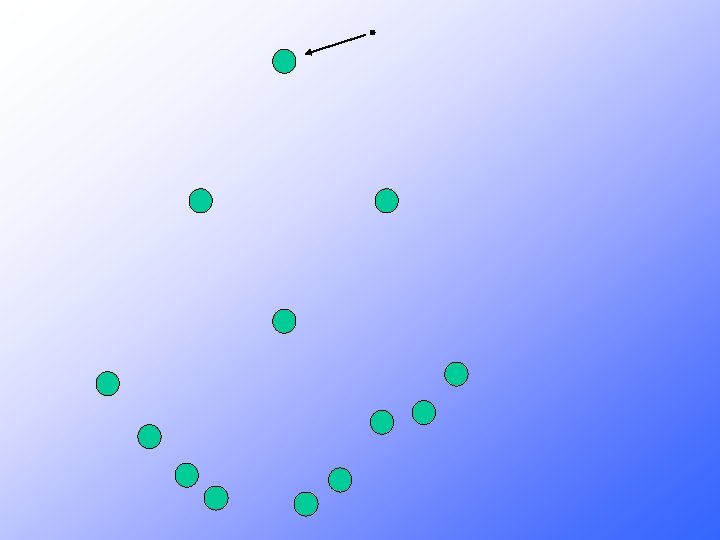
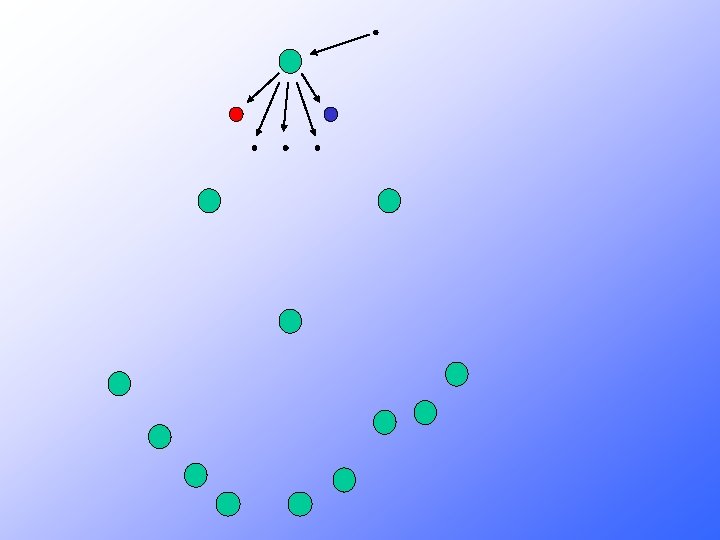
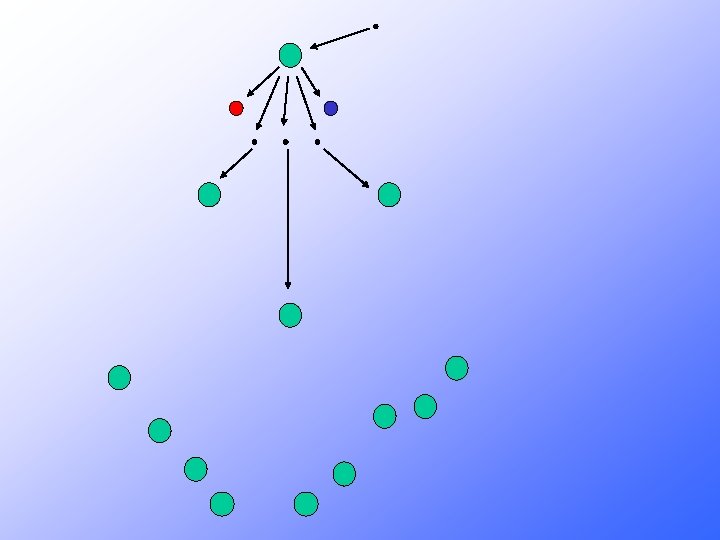
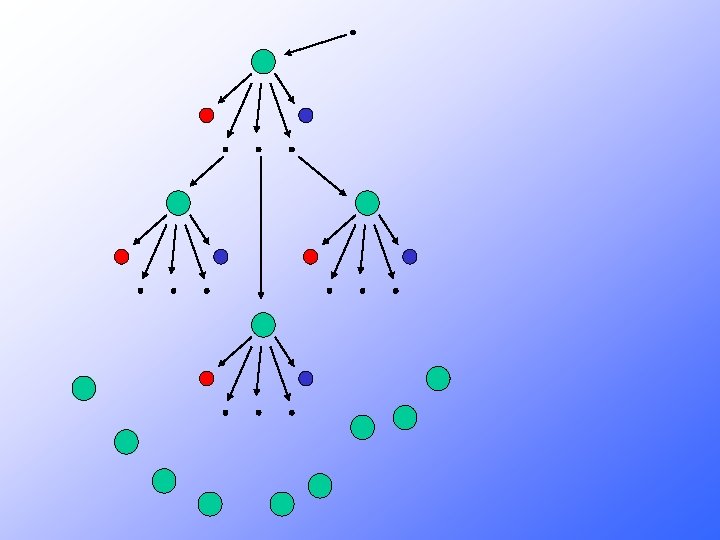
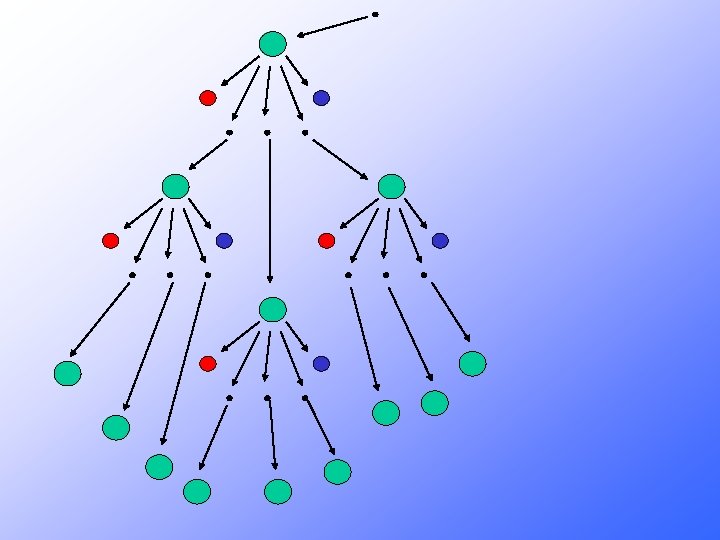


Where does all this energy come from?
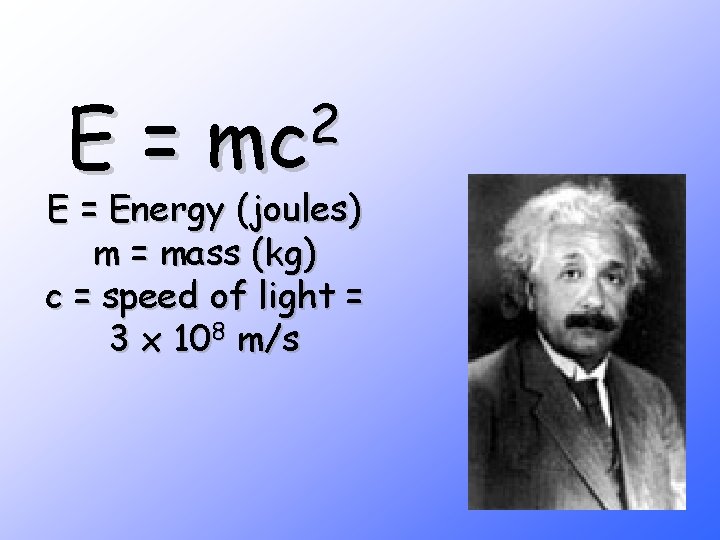
E= 2 mc E = Energy (joules) m = mass (kg) c = speed of light = 3 x 108 m/s

Nuclear Fission & POWER • Currently about 103 nuclear power plants in the U. S. and about 435 worldwide. • 17% of the world’s energy comes from nuclear.

Fusion • Light-mass nuclei combine to form a heavier, more stable nucleus. • More energetic than fission reactions • Source of energy for the H-bomb • Origin of the elements
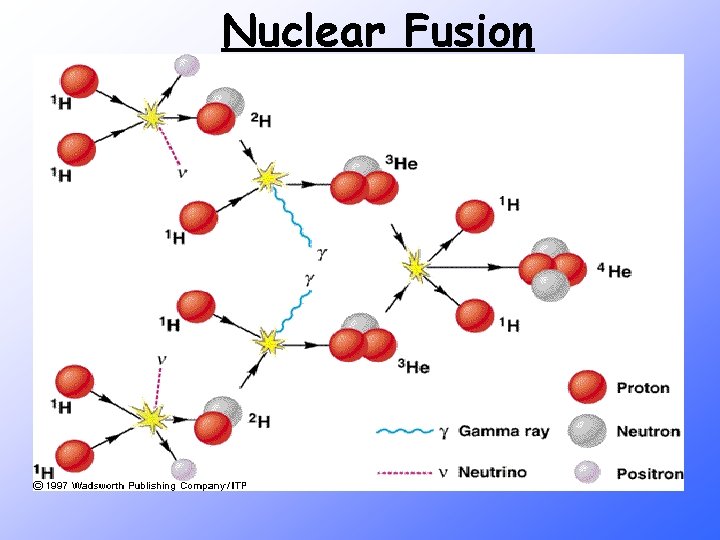
Nuclear Fusion
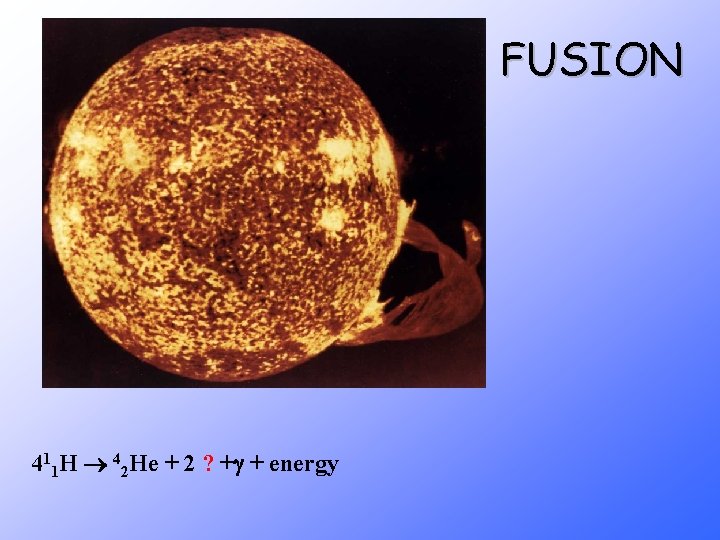
FUSION 411 H 42 He + 2 ? + + energy

Stars energy is produced through fusion reactions Fusion occurs until Fe is produced because less energy is released than required to fuse Fe nuclei = Star ____ burns ____ out _____

The most destructive force on the planet H-bombs 1000 s of times more powerful than A-bombs

Cold Fusion: Efforts are being made to start and sustain a fusion reaction at lower temperatures, in other words with a lower amount of input energy
- Slides: 66