Nuclear Reactions Alpha Beta and Gamma Decay CS

Nuclear Reactions Alpha, Beta, and Gamma Decay

CS 4. 2 State what is meant by alpha, beta and gamma decay of radionuclides. CS 4. 3 Identify the processes occurring in nuclear reactions written in symbolic form.

Warm-up 9/23 • • What is an isotope? What is the same in isotopes? What is different in isotopes? Write down two or three sentences about what you think a nuclear reaction is.
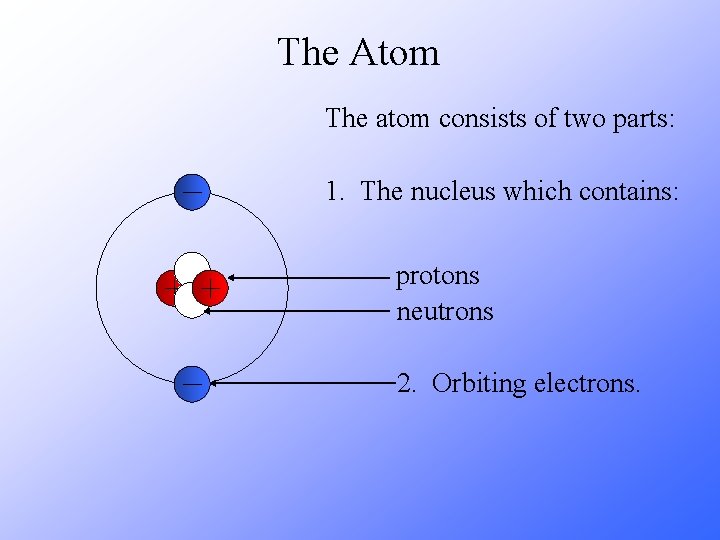
The Atom The atom consists of two parts: 1. The nucleus which contains: protons neutrons 2. Orbiting electrons.

The Atom All matter is made up of elements (e. g. carbon, hydrogen, etc. ). The smallest part of an element is called an atom. Atom of different elements contain different numbers of protons. The mass of an atom is almost entirely due to the number of protons and neutrons.
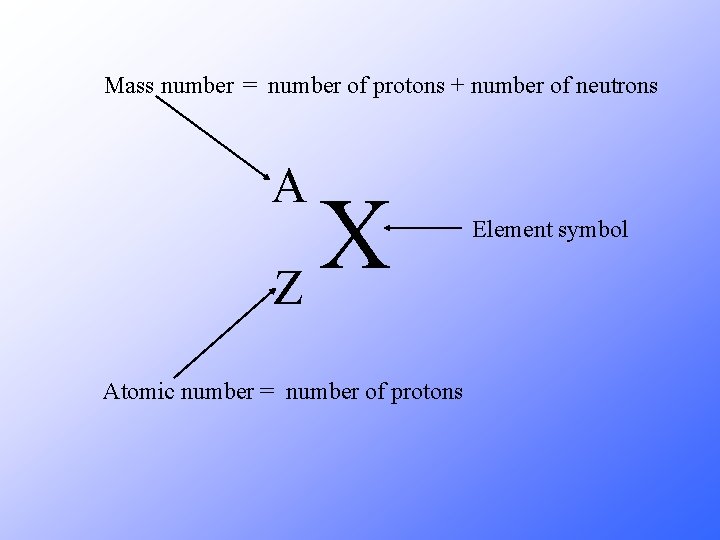
Mass number = number of protons + number of neutrons A X Z Atomic number = number of protons Element symbol

A X Z A = number of protons + number of neutrons Z = number of protons A – Z = number of neutrons Number of neutrons = Mass Number – Atomic Number

There are many types of uranium: 235 238 A A Z Z Number of protons Number of neutrons U 92

There are many types of uranium: 235 238 U 92 A 235 A 238 Z 92 Number of protons 92 Number of neutrons 143 Number of neutrons 146 Isotopes of any particular element contain the same number of protons, but different numbers of neutrons.

Most of the isotopes which occur naturally are stable. A few naturally occurring isotopes and all of the manmade isotopes are unstable. Video A stable by releasing Unstable isotopes can become different types of particles. This process is called radioactive decay and the elements which undergo this process are called radioisotopes/radionuclides.

Radioactivity • Spontaneous emission of energy from unstable nuclei in order for an atom to increase its stability. • Atoms within the band of stability are not radioactive. • Creation of a new element (changing the atomic #) through radioactive decay is called transmutation.
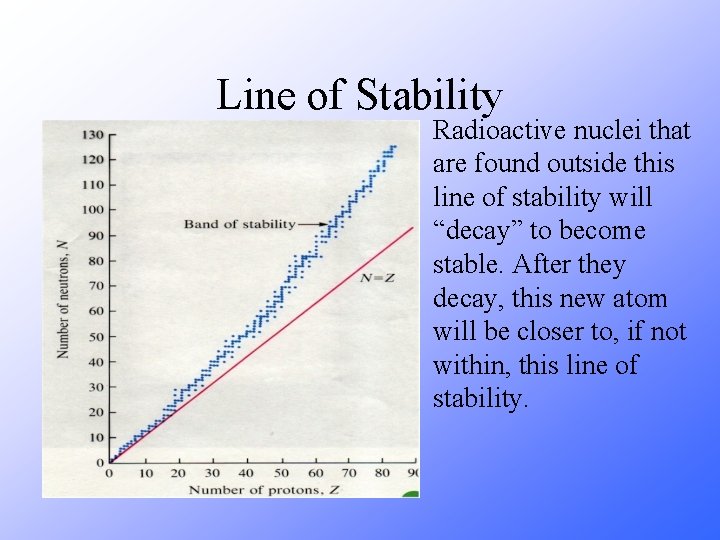
Line of Stability Radioactive nuclei that are found outside this line of stability will “decay” to become stable. After they decay, this new atom will be closer to, if not within, this line of stability.

Why is energy released? • Energy is released because there is a slight mass difference between the nucleus before radioactive decay and afterward. • That mass difference is called a mass defect. • Einstein in his Theory of Relativity, stated that mass defect is converted into pure energy. • The equation for that energy release is E=mc 2, where E in energy, m is mass, and c is the speed of light.

Modes of Decay Type Symbol Composition Result Alpha (a) 4 He Helium nucleus, Nucleus 2 neutrons, 2 loses mass protons Beta (b) b- or -1 e Electron Gamma 0 g Pure energy; Nucleus gives off extra energy or g Strength Heaviest, therefore the weakest Neutron into Lighter, proton stronger Strongest

Radioactive Decay Radioactive decay results in the emission of either: • an alpha particle (a), • a beta particle (b), • or a gamma ray(g).

Alpha Decay An alpha particle is identical to that of a helium nucleus. It contains two protons and two neutrons.

Alpha Decay A X Z A-4 4 Y He + Z-2 2 unstable atom alpha particle more stable atom
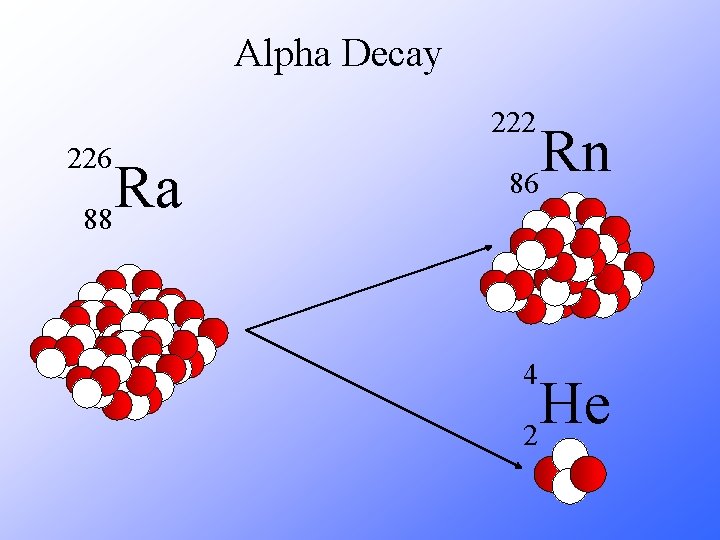
Alpha Decay 222 226 Ra 88 Rn 86 4 He 2

Alpha Decay A A-4 4 226 222 4 X Z Ra 88 Y + Z-2 Rn + 86 He 2

Alpha Decay 222 Rn 86 A 4 Y He + Z 2 218 Po + 84 4 He 2

Alpha Decay A 230 4 X Z U 92 Th He + 90 2

Alpha Decay 230 Th 90 A 4 226 4 Y He + Z 2 Ra He + 88 2

Alpha Decay A 214 4 218 214 4 X Z Po 84 Pb He + 82 2

Beta Decay A beta particle is a fast moving electron which is emitted from the nucleus of an atom undergoing radioactive decay. Beta decay occurs when a neutron changes into a proton and an electron.

Beta Decay As a result of beta decay, the nucleus has one less neutron, but one extra proton. The atomic number, Z, increases by 1 and the mass number, A, stays the same.
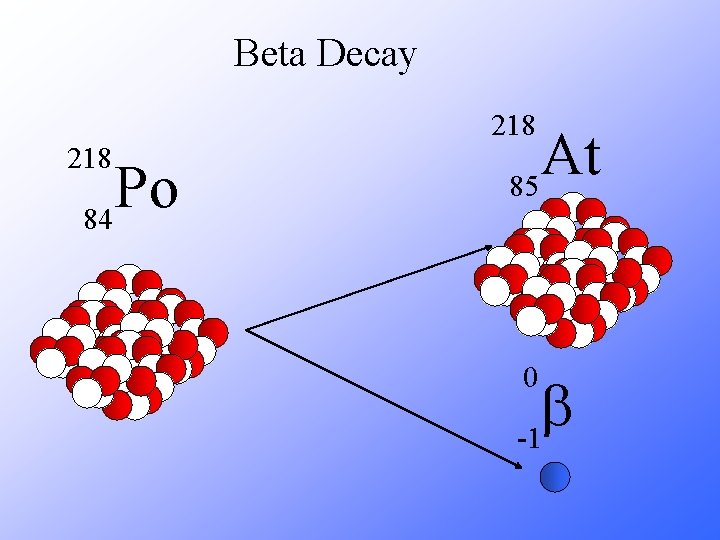
Beta Decay 218 Po 84 At 85 0 b -1

Beta Decay A X Z 218 Po 84 A Y + Z+1 218 Rn + 85 0 b -1

Beta Decay 234 A 234 Th 90 Y + Z Pa + 91 0 b -1

Beta Decay A 210 b -1 210 0 X Z Tl 81 Pb + 82 0 b -1

Beta Decay 210 A 210 Bi 83 Y + Z Po + 84 0 b -1

Beta Decay A 214 b -1 214 0 X Z Pb 82 Bi + 83 0 b -1

Gamma Decay Gamma rays are not charged particles like a and b particles. Gamma rays are electromagnetic radiation with high frequency. When atoms decay by emitting a or b particles to form a new atom, the nuclei of the new atom formed may still have too much energy to be completely stable. This excess energy is emitted as gamma rays (gamma ray photons have energies of ~ 1 x 10 -12 J).

Penetrating Power of Radiation • The most dangerous form of radiation is gamma radiation. The least dangerous is alpha radiation.

Fission and Fusion • Fission is a special type of nuclear reaction in which a large nucleus is split into smaller nuclei, releasing tremendous amounts of energy. • Fission powers nuclear power plants and bombs. • 235 U + 1 n 90 Sr + 143 Xe + 3 1 n • Fusion is the case where small nuclei are combined into larger ones. • This also releases large amounts of energy. This is what powers the sun and what fuels hydrogen bombs. • 2 H + 3 H 4 He + 1 n

Half-Life • Whether a radioactive nucleus will decay depends on a function of probability. • The amount of time in which a nucleus has a 50% chance of decaying is called half-life. • If you have a large amount of a radioactive substance, half of it will decay in the period of a half-life.

Half-life Equation • The amount of a substance left during a half -life can be calculated from the following equation. • Initial Amount Left = ------------2 n n = number of half-lives
Half-Life Example • The amount of Carbon-14 in a substance can be used to determine the age of a fossil. • Today’s plants contain 0. 25% Carbon-14. A fossil is found to have 0. 031% Carbon-14. If the half-life of Carbon-14 is 5730 years, how old is the fossil?

Solution • • Initial Amount Left = ------------2 n 0. 25% 0. 031% = -------2 n • 2 n = 8 ; n = 3 Three half-lives • One half-life = 5730 yr. 3 x 5730 = 17190 years. • The fossil is 17190 years old.
- Slides: 38