Nuclear Radioactive Decay ATOMS NUCLEAR INTERACTIONS Nuclear Stability

Nuclear Radioactive Decay ATOMS: NUCLEAR INTERACTIONS

Nuclear Stability �The nucleus consists of protons and neutrons that are held together by the nuclear force – a strong force of attraction between nucleons. �Nucleus stability is determined by the ratio between the neutrons and protons. �For elements up to Z=20, the ratio between neutrons and protons is between 1: 1 and 1. 1: 1 �Above Z=20, the ratio increases to 1. 5: 1 �There are no stable nuclides above element 83
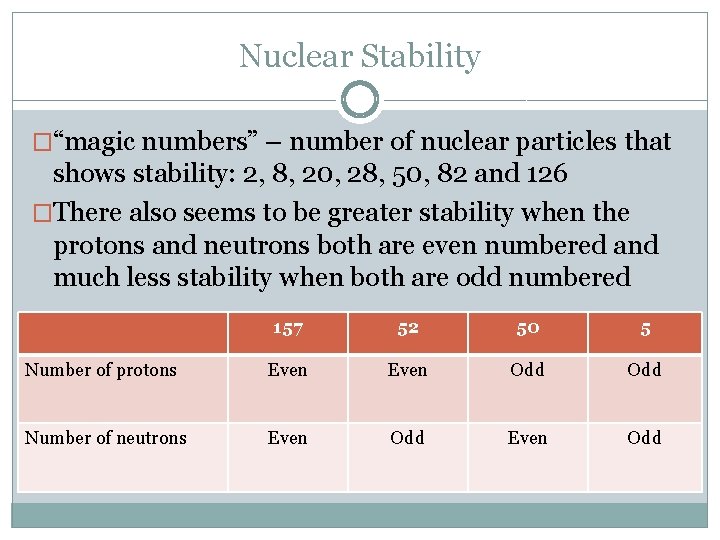
Nuclear Stability �“magic numbers” – number of nuclear particles that shows stability: 2, 8, 20, 28, 50, 82 and 126 �There also seems to be greater stability when the protons and neutrons both are even numbered and much less stability when both are odd numbered 157 52 50 5 Number of protons Even Odd Number of neutrons Even Odd

Nuclear Stability �Calculate the stability (possible radioactivity) of the following isotopes: 39 20 Neutrons 19 K The ratio = Protons = 19 = 1. 05: 1 Stable 14 8 Neutrons 6 C The ratio = Protons = 6 = 1. 33: 1 Unstable 208 84 Po The element is greater than Z = 83 Unstable

Natural Radioactive Decay �There are 5 main types of radioactive decay: 4 � 1. Alpha Emission (α) – the emission of a He nucleus 2 This occurs for all elements Z > 83 226 4 222 Ra → He + Rn 88 2 86 Conservation of Matter: 2 + 86 = 88 and 4 + 222 = 226 234 4 230 Th → Ra He + 90 2 88

Natural Radioactive Decay � 2. Beta Emission (β or 0 e) – emission of a high speed -1 electron This occurs when the N/Z (neutron/proton) ratio is too large 14 0 14 C → e + N 6 -1 7 �Notice the conservation of matter again: 6 = -1 + 7 �The ratio for carbon-14 should be 1: 1 since it is Z < 20, but the ratio is 8: 6 or 1. 33: 1 making it unstable – the final ratio of nitrogen-14 is 1: 1, making it stable

Natural Radioactive Decay 0 � 3. Positron Emission or e) – emission of positron 1 This occurs when the N/Z ration is too small 95 0 95 Tc → e + Mo 43 1 42 11 = 0 + ? 11 0 11 C → e + B 6 1 5 6 = 1 + ? �Notice the ratio of carbon-11 is 5: 6 or 0. 8: 1 which is smaller than it should be, making it unstable (β+

Natural Radioactive Decay � 4. Electron Capture (EC) – unstable nucleus captures an electron In essence a proton is changed into a neutron 1 p + 0 e → 1 n 1 -1 0 � 5. Gamma Emission (γ) – emission of a gamma photon from an excited nucleus (metastable) 99 m 99 0 Tc → Tc + γ 43 43 0
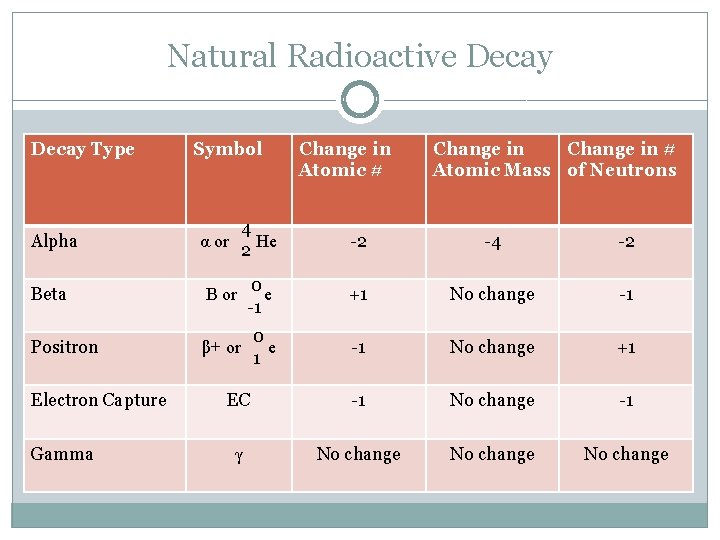
Natural Radioactive Decay Type Symbol Change in Atomic # Change in # Atomic Mass of Neutrons Alpha α or 4 He 2 -2 -4 -2 Beta Β or 0 e -1 +1 No change -1 Positron β+ or 0 e 1 -1 No change +1 EC -1 No change -1 γ No change Electron Capture Gamma

Natural Radioactive Decay �The penetrating ability of alpha (α), beta (β) and gamma (γ) rays. Gamma rays are the most penetrating; alpha particles are the least. �Alpha (α) is stopped by paper �Beta (β) is stopped by aluminum �Gamma (γ) is stopped by lead sheet or concrete block
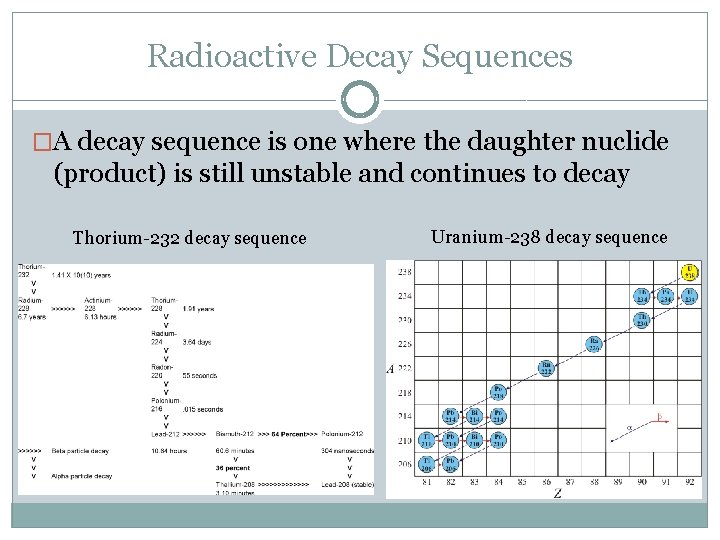
Radioactive Decay Sequences �A decay sequence is one where the daughter nuclide (product) is still unstable and continues to decay Thorium-232 decay sequence Uranium-238 decay sequence
- Slides: 11