Nuclear Radiation www assignmentpoint com Nuclear Radiation 1



























- Slides: 31

Nuclear Radiation www. assignmentpoint. com

Nuclear Radiation 1. 2. 3. 4. 5. 6. Nuclear Emissions Nuclear Equations Producing Radioactive Isotopes Half-Life Nuclear Fission and Fusion Uses & Effects www. assignmentpoint. com

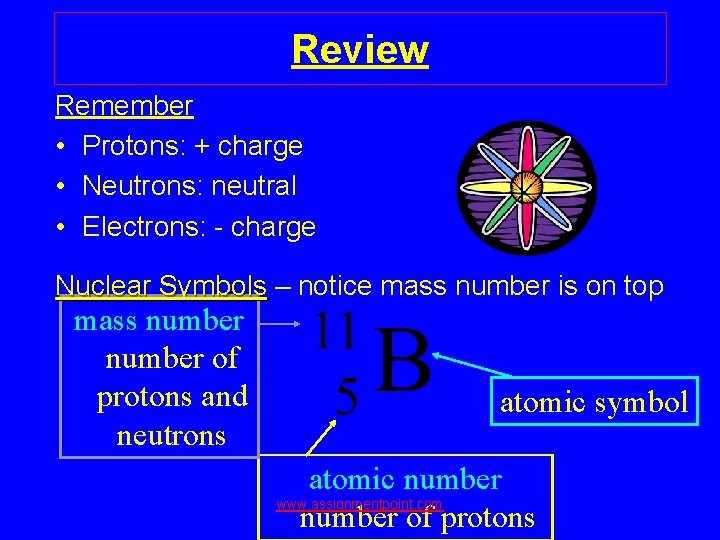
Review Remember • Protons: + charge • Neutrons: neutral • Electrons: - charge Nuclear Symbols – notice mass number is on top mass number of protons and neutrons atomic symbol atomic number www. assignmentpoint. com number of protons

1. Nuclear Emissions • Radiation comes from the nucleus of an atom. • Unstable nucleus emits a particle or energy alpha beta gamma • non-radioactive particle = neutron www. assignmentpoint. com

Alpha Particle Same as a helium nucleus (He) 4 2 He or Two protons Two neutrons www. assignmentpoint. com

Beta Particle An electron emitted from the nucleus 0 e or 1 A neutron in the nucleus breaks down 1 1 0 n H + e 0 1 -1 www. assignmentpoint. com

Gamma Radiation • Pure radiation • Like an X-ray but comes from the nucleus www. assignmentpoint. com

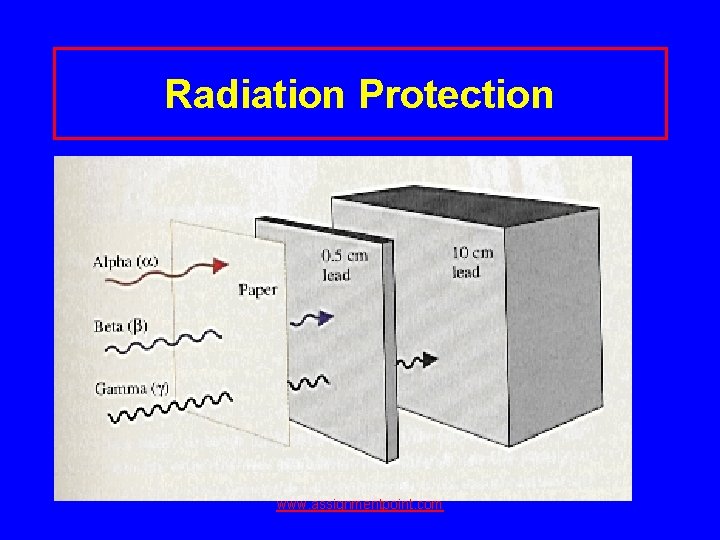
Radiation Protection • Shielding alpha – paper, clothing beta – lab coat, gloves gamma- lead, thick concrete • Limit time exposed • Keep distance from source www. assignmentpoint. com

Radiation Protection www. assignmentpoint. com

2. Nuclear Equations In the reactants and products Atomic numbers must balance and Mass numbers must balance www. assignmentpoint. com

Alpha decay www. assignmentpoint. com

Beta decay 234 Th 90 ® 234 Pa 91 + 0 e 1 beta particle www. assignmentpoint. com

Gamma radiation No change in atomic or mass number 11 B 5 5 + boron atom in a high-energy state www. assignmentpoint. com 0 0

Learning Check • To predict the product, simply remember that the mass number and atomic number are conserved • What is the identity of X? www. assignmentpoint. com

Learning Check Write the nuclear equation for the beta emitter Co-60. www. assignmentpoint. com

Solutions 239 Np 93 60 Co 60 Ni 27 28 + www. assignmentpoint. com 0 e -1

3. Producing Radioactive Isotopes Bombardment of atoms produces radioisotopes = 60 59 Co = 60 + 1 n 27 56 Mn 0 25 = 27 cobalt atom + 4 H e 2 = 27 neutron manganese alpha radioisotope particle www. assignmentpoint. com

Learning Check What radioactive isotope is produced in the following bombardment of boron? 10 B 5 + 4 He ? + 2 0 www. assignmentpoint. com 1 n

Solution What radioactive isotope is produced in the following bombardment of boron? 10 B 5 + 4 He 2 13 N 7 nitrogen radioisotope www. assignmentpoint. com + 0 1 n

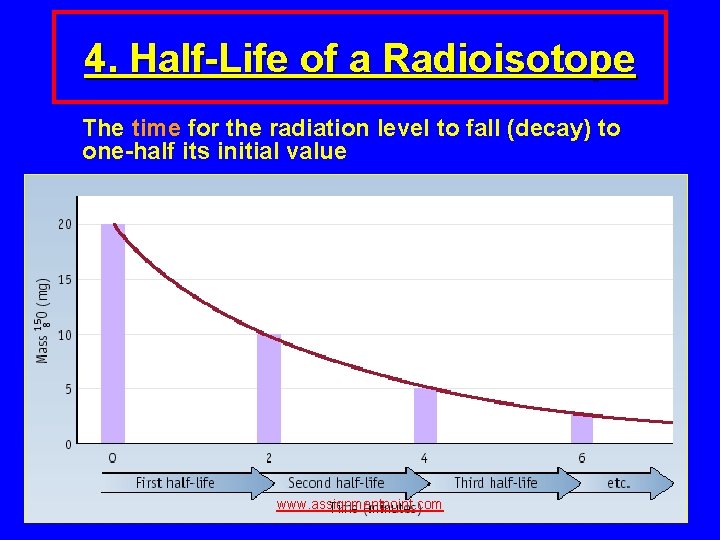
4. Half-Life of a Radioisotope The time for the radiation level to fall (decay) to one-half its initial value www. assignmentpoint. com

Examples of Half-Life Isotope C-15 Ra-224 Ra-223 I-125 C-14 U-235 Half life 2. 4 sec 3. 6 days 12 days 60 days 5700 years 710 000 years www. assignmentpoint. com

Learning Check The half life of I-123 is 13 hr. How much of a 64 mg sample of I-123 is left after 26 hours? www. assignmentpoint. com

Solution t 1/2 = 13 hrs 26 hours = 2 x t 1/2 Amount initial = 64 mg Amount remaining = 64 mg x ½ = 16 mg www. assignmentpoint. com

5. Fission VS Fusion Fission large nuclei break up 235 U 92 + 1 n 0 139 Ba 56 + 94 Kr 36 www. assignmentpoint. com + 3 1 n + 0 Energy

Fission of Uranium-235 • Chain reaction – the reaction sustains itself by producing more neutrons www. assignmentpoint. com

Nuclear Fusion small nuclei combine 2 H 1 + 3 H 4 He 1 2 + 1 n + 0 Occurs in the sun and other stars • Excessive heat can not be contained • Attempts at “cold” fusion have FAILED. • “Hot” fusion is difficult to contain www. assignmentpoint. com Energy

Learning Check Indicate if each of the following are (1) Fission (2) fusion A. B. C. D. Nucleus splits Large amounts of energy released Small nuclei form larger nuclei Hydrogen nuclei react www. assignmentpoint. com Energy

Solution Indicate if each of the following are (1) Fission (2) fusion A. B. C. D. 1 1+2 2 2 Nucleus splits Large amounts of energy released Small nuclei form larger nuclei Hydrogen nuclei react www. assignmentpoint. com

6. Uses & Effects • Radiation - treatment of cancer • Nuclear medicine - use of radioisotopes in diagnosis – Tracers – small amounts of radioactive substances used as probes to study internal organs – Nuclear imaging – medical techniques involving tracers • Food Irradiation – Food - irradiated with g rays from 60 Co or 137 Cs. – Irradiated milk - shelf life of 3 mo. w/o cooling. – USDA - approved irradiation of meats and eggs www. assignmentpoint. com

www. assignmentpoint. com

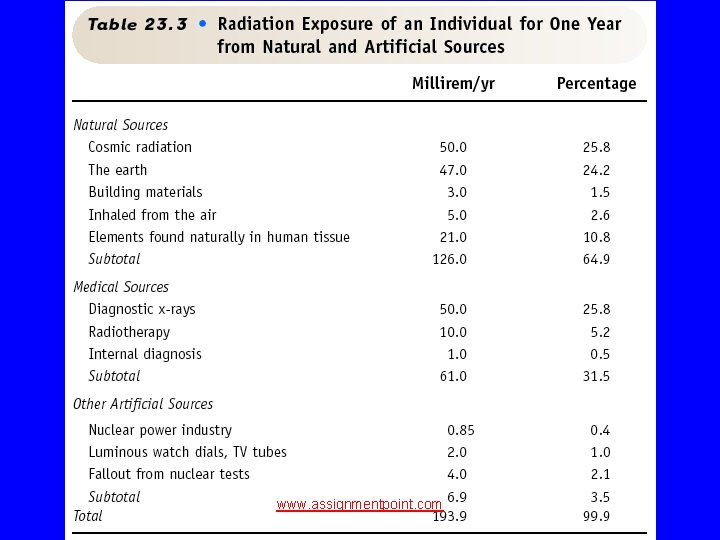
Exposure Sources www. assignmentpoint. com