NUCLEAR PHYSICS BINDING ENERGY RADIOACTIVITY Sri Balaji Sankar

NUCLEAR PHYSICS BINDING ENERGY & RADIOACTIVITY Sri Balaji Sankar Panda Department of Physics Khemundi college, Digapahandi

Nuclear Components • • • Nucleus contains nucleons: protons and neutrons Atomic number Z = number of protons Neutron number N = number of neutrons Mass number A = number of nucleons = Z + N Each element has unique Z value Isotopes of element have same Z, but different N and A values Notation:

Nucleus Charge and Mass Particle Charge Mass (kg) Mass (u) Mass (Me. V/c 2) Proton +e 1. 672 6 E− 27 1. 007 276 938. 28 Neutron 0 1. 675 0 E− 27 1. 008 665 939. 57 Electron −e 9. 109 5. 486 E− 4 0. 511 E− 31 • Unified mass unit, u, defined using Carbon 12 • Mass of 1 atom of 12 C ≡ 12 u

Nuclei Sizes • Scattering experiments determine size • Measured in femtometers (aka fermis) • All nuclei have nearly the same density Fig. 29. 2, p. 959
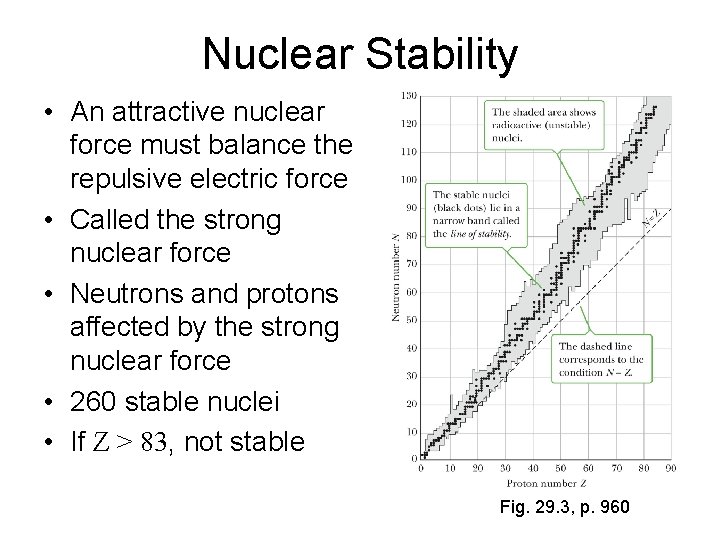
Nuclear Stability • An attractive nuclear force must balance the repulsive electric force • Called the strong nuclear force • Neutrons and protons affected by the strong nuclear force • 260 stable nuclei • If Z > 83, not stable Fig. 29. 3, p. 960
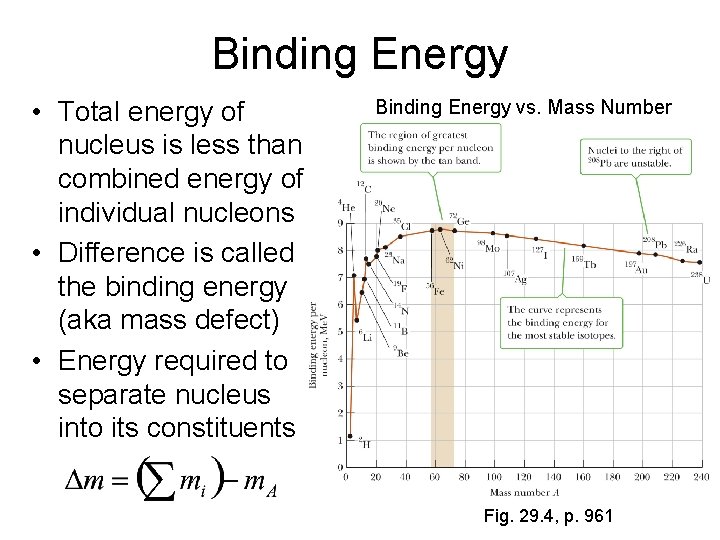
Binding Energy • Total energy of nucleus is less than combined energy of individual nucleons • Difference is called the binding energy (aka mass defect) • Energy required to separate nucleus into its constituents Binding Energy vs. Mass Number Fig. 29. 4, p. 961

Radioactivity • Unstable nuclei decay to more stable nuclei • Can emit 3 types of radiation in the process A positron (e+) is the antiparticle of the electron (e−) Fig. 29. 5, p. 962
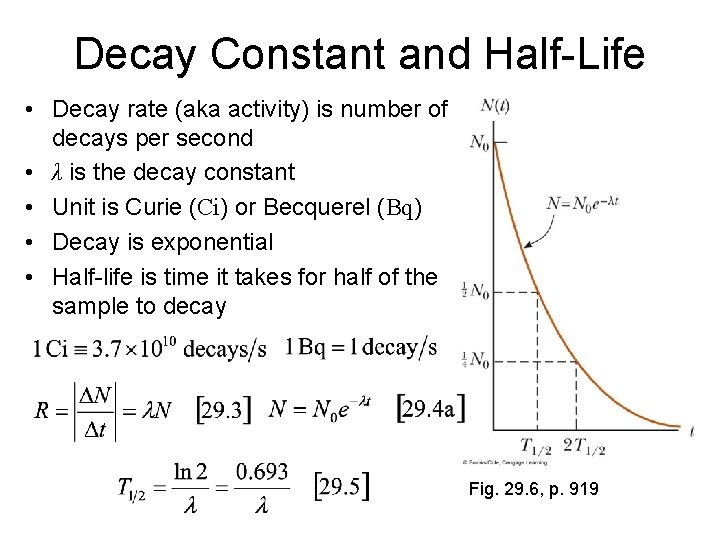
Decay Constant and Half-Life • Decay rate (aka activity) is number of decays per second • λ is the decay constant • Unit is Curie (Ci) or Becquerel (Bq) • Decay is exponential • Half-life is time it takes for half of the sample to decay Fig. 29. 6, p. 919

Alpha Decay • Unstable nucleus emits particle (i. e. , a helium nucleus) spontaneously • Mass of parent is greater than mass of daughter plus particle • Most of KE carried away by particle Fig 29. 7, p. 966
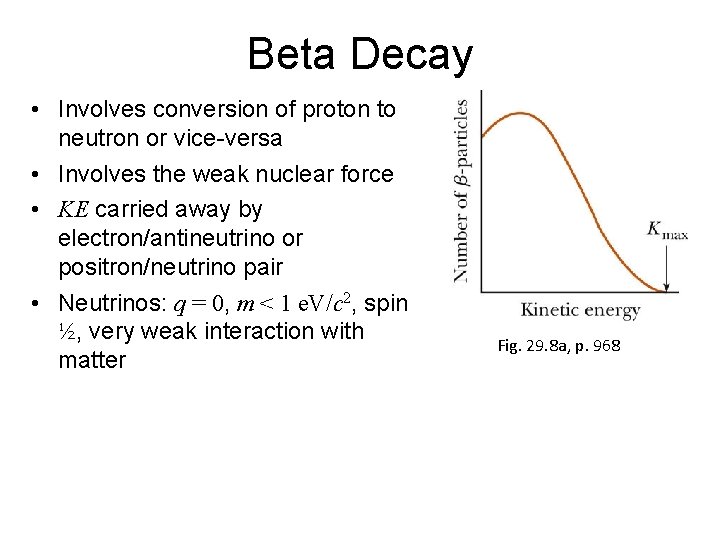
Beta Decay • Involves conversion of proton to neutron or vice-versa • Involves the weak nuclear force • KE carried away by electron/antineutrino or positron/neutrino pair • Neutrinos: q = 0, m < 1 e. V/c 2, spin ½, very weak interaction with matter Fig. 29. 8 a, p. 968

Gamma (γ) Decay • Following radioactive decay, nucleus may be left in an excited state • Undergoes nuclear de-excitation: protons/neutrons move to lower energy level • Nucleus emits high energy photons (γ rays) • No change in A or Z results

THANK YOU
- Slides: 12