Nuclear physics 1 Structure At the center of








- Slides: 14

Nuclear physics 1. Structure • • At the center of an atom there is a tiny but massive nucleus. Nuclei are made up of protons and neutrons Proton has a positive charge with the same magnitude as electron (+e=1. 6 x 10 -19 C) and a mass mp = 1. 67262 x 10 -27 kg Neutron is electrically neutral (q=0) and has a mass mn = 1. 67493 x 10 -27 kg that is very slightly lager the mass of the proton Nucleons: protons and neutrons. Different nuclei are referred as nuclides 2. Terminology Z - the atomic number or proton number (the number of protons in the nucleus) N - the neutron number (the number of neutrons in the nucleus) A - the atomic mass number (the number of nucleons in the nucleus) A=Z+N X - chemical symbol for the element; it contains the same information as Z but in a more easily recognizable form Standard notation: Isotopes – nuclei that contain the same number of protons but different number of neutrons 1

3. Atomic mass units Masses of atoms are measured with reference to the carbon-12 atom A neutral atom is given the precise value 12. 000000 u (unified atomic mass units) 1 u = 1. 6605∙ 10 -27 kg = 931. 5 Me. V/c 2 Proton: mp = 1. 67262∙ 10 -27 kg = 1. 007276 u = 938. 27 Me. V/c 2 Neutron: mn = 1. 67493∙ 10 -27 kg = 1. 008665 u = 939. 57 Me. V/c 2 1 H atom: m. H = 1. 67353∙ 10 -27 kg = 1. 007825 u = 938. 78 Me. V/c 2 (proton +electron) Electron: me = 9. 10938∙ 10 -31 kg = 0. 00054858 u =0. 51100 Me. V/c 2 What it is Me. V/c 2 ? 2

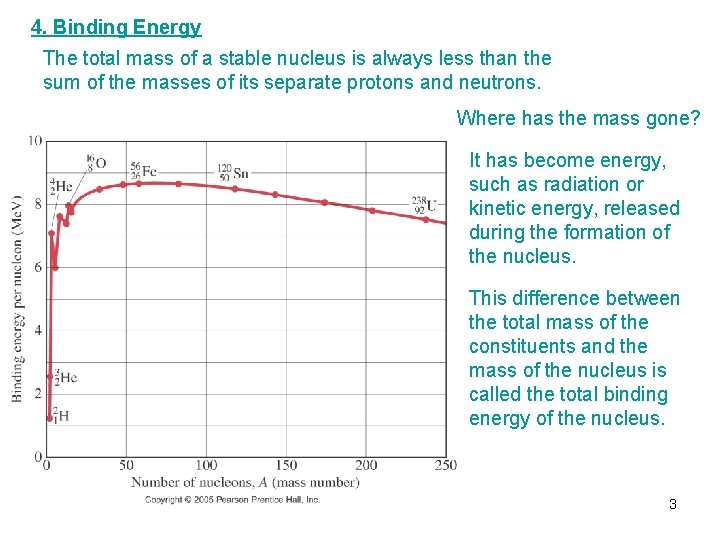
4. Binding Energy The total mass of a stable nucleus is always less than the sum of the masses of its separate protons and neutrons. Where has the mass gone? It has become energy, such as radiation or kinetic energy, released during the formation of the nucleus. This difference between the total mass of the constituents and the mass of the nucleus is called the total binding energy of the nucleus. 3

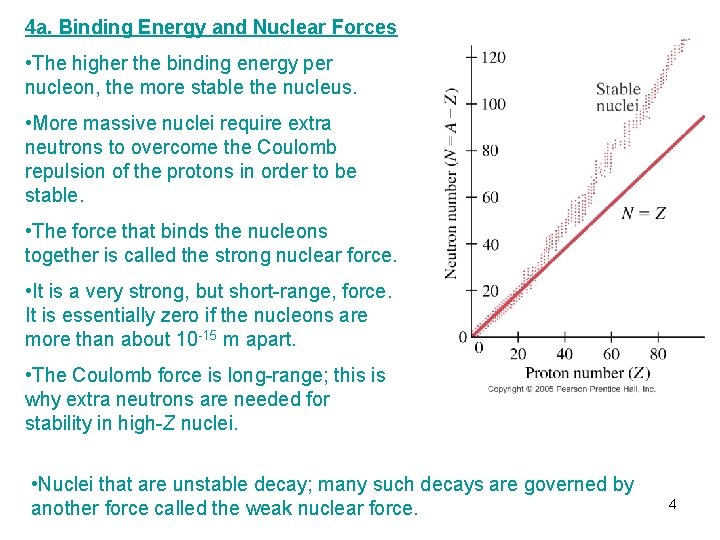
4 a. Binding Energy and Nuclear Forces • The higher the binding energy per nucleon, the more stable the nucleus. • More massive nuclei require extra neutrons to overcome the Coulomb repulsion of the protons in order to be stable. • The force that binds the nucleons together is called the strong nuclear force. • It is a very strong, but short-range, force. It is essentially zero if the nucleons are more than about 10 -15 m apart. • The Coulomb force is long-range; this is why extra neutrons are needed for stability in high-Z nuclei. • Nuclei that are unstable decay; many such decays are governed by another force called the weak nuclear force. 4

5. Nuclear Reactions and the Transmutation of Elements A nuclear reaction takes place when a nucleus is struck by another nucleus or particle. If the original nucleus is transformed into another, this is called transmutation. Example: Laws of conservation: • total number of nucleons • total charge • energy • momentum must be conserved in nuclear reactions. 5

6. Nuclear Fission. Nuclear Reactors Neutrons are very effective in nuclear reactions, as they have no charge and therefore are not repelled by the nucleus. After absorbing a neutron, a uranium-235 nucleus will split into two roughly equal parts. One way to visualize this is to view the nucleus as a kind of liquid drop. The mass distribution of the fragments shows that the two pieces are large, but usually unequal. The energy release in a fission reaction is quite large. 6

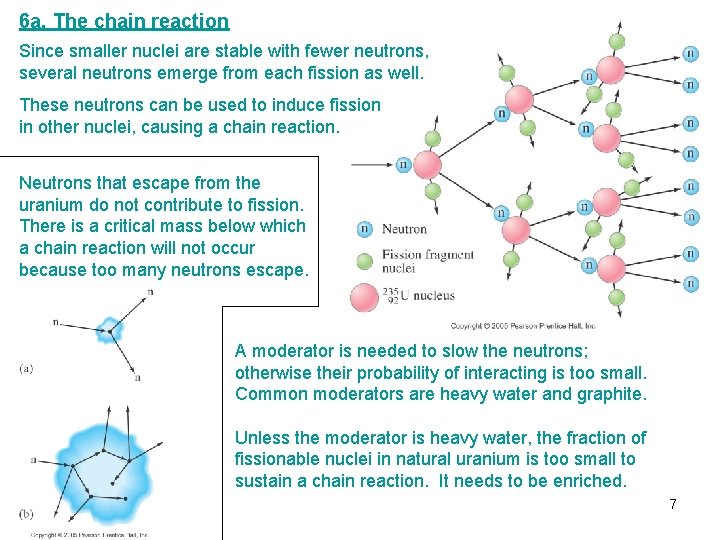
6 a. The chain reaction Since smaller nuclei are stable with fewer neutrons, several neutrons emerge from each fission as well. These neutrons can be used to induce fission in other nuclei, causing a chain reaction. Neutrons that escape from the uranium do not contribute to fission. There is a critical mass below which a chain reaction will not occur because too many neutrons escape. A moderator is needed to slow the neutrons; otherwise their probability of interacting is too small. Common moderators are heavy water and graphite. Unless the moderator is heavy water, the fraction of fissionable nuclei in natural uranium is too small to sustain a chain reaction. It needs to be enriched. 7

Radioactivity (Discovered in 1896 by Henry Becquerel) Most of the known nuclides are unstable (radioactive). A radioactive nuclide spontaneously emits particles (radioactive rays), transforming itself into a different nuclide. Radioactive rays were observed to be of three types: 1. Alpha rays, which could barely penetrate a piece of paper 2. Beta rays, which could penetrate 3 mm of aluminum 3. Gamma rays, which could penetrate several centimeters of lead Alpha and beta rays are bent in opposite directions in a magnetic field, while gamma rays are not bent at all. 1. Alpha rays are helium nuclei 2. Beta rays are electrons 3. Gamma rays are electromagnetic radiation 8

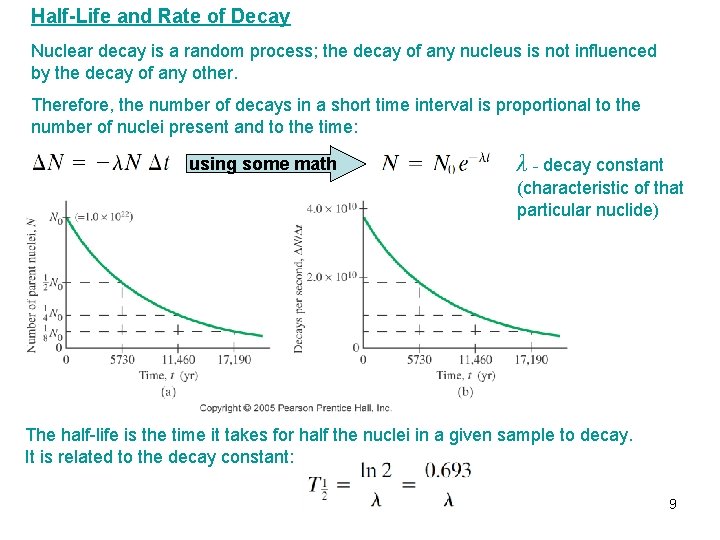
Half-Life and Rate of Decay Nuclear decay is a random process; the decay of any nucleus is not influenced by the decay of any other. Therefore, the number of decays in a short time interval is proportional to the number of nuclei present and to the time: using some math λ - decay constant (characteristic of that particular nuclide) The half-life is the time it takes for half the nuclei in a given sample to decay. It is related to the decay constant: 9

Radioactive Dating • Radioactive dating can be done by analyzing the fraction of carbon-14 in organic material. • The ratio of the carbon-14 atoms to carbon-12 atoms in the atmosphere has been roughly constant over thousands of years. This ratio is about 1. 3 x 10 -12. • A living plant or tree will be constantly exchanging carbon with the atmosphere, and will have the same carbon ratio in its tissues. • When the plant dies, this exchange stops. Carbon-14 has a half-life of about 5730 years; it gradually decays away and becomes a smaller and smaller fraction of the total carbon in the plant tissue. • This fraction can be measured, and the age of the tissue deduced. • Objects older than about 60, 000 years cannot be dated this way – there is too little carbon-14 left. • Other isotopes are useful for geologic time scale dating. • Uranium-238 has a half-life of 4. 5 x 109 years, and has been used to date the oldest rocks on Earth as about 4 billion years old. 10

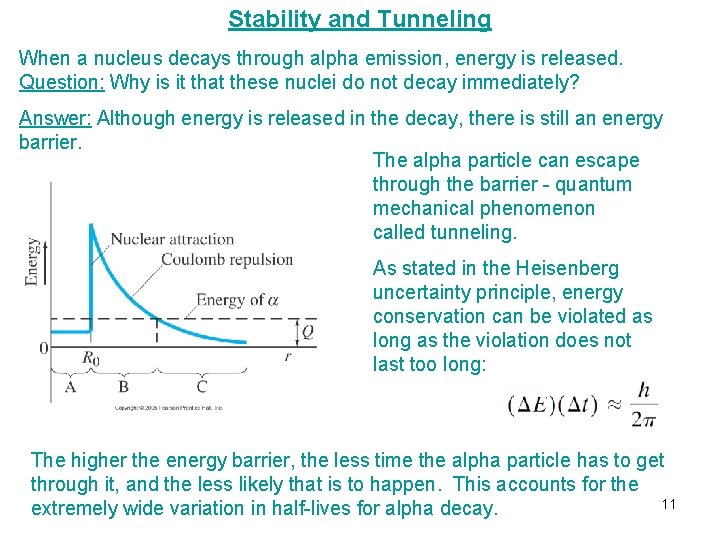
Stability and Tunneling When a nucleus decays through alpha emission, energy is released. Question: Why is it that these nuclei do not decay immediately? Answer: Although energy is released in the decay, there is still an energy barrier. The alpha particle can escape through the barrier - quantum mechanical phenomenon called tunneling. As stated in the Heisenberg uncertainty principle, energy conservation can be violated as long as the violation does not last too long: The higher the energy barrier, the less time the alpha particle has to get through it, and the less likely that is to happen. This accounts for the 11 extremely wide variation in half-lives for alpha decay.

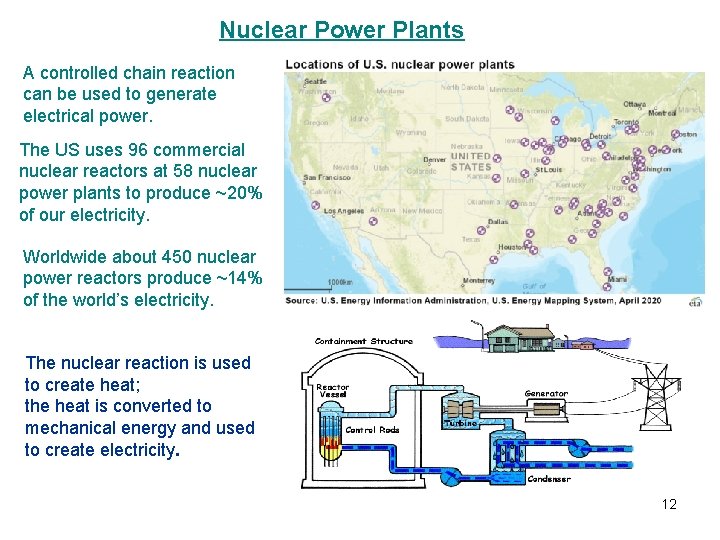
Nuclear Power Plants A controlled chain reaction can be used to generate electrical power. The US uses 96 commercial nuclear reactors at 58 nuclear power plants to produce ~20% of our electricity. Worldwide about 450 nuclear power reactors produce ~14% of the world’s electricity. The nuclear reaction is used to create heat; the heat is converted to mechanical energy and used to create electricity. 12

Three Mile Island shows US nuclear risks and rewards The Three Mile Island (Pennsylvania) accident (March 28, 1979) was a partial meltdown of a reactor, and subsequent radiation. It was the most significant accident in U. S. commercial nuclear power plant history. Unit 1 was shutdown at on September 20, 2019. Unit 2 has been dormant since the accident in 1979 Unlike the Chernobyl disaster in Ukraine (April 26, 1986) no one died at Three Mile Island. But critics of atomic power raise concerns over potential terrorist threats to plants and say science has yet to provide an adequate solution for highly toxic nuclear waste. eia. gov/energyexplained/nuclear/us-nuclear-industry. php https: //en. wikipedia. org/wiki/Three_Mile_Island_Nuclear_Generating_Station 13

Lecture outline Nuclear physics • Structure (protons and neutrons) • • • Terminology (A = Z + N ), , isotopes Atomic mass units and Me. V/c 2 Binding energy and nuclear forces Nuclear reactions and the transmutation Laws of conservation: • Nuclear Fission. Nuclear Reactors • The chain reaction • Radioactivity • Half-life and rate of decay • Radioactive dating • Stability and tunneling • Nuclear power plants 14