Nuclear Masses and Binding Energy Lesson 3 Nuclear

Nuclear Masses and Binding Energy Lesson 3

Nuclear Masses • Nuclear masses and atomic masses Because Belectron(Z)is so small, it is neglected in most situations.

Mass Changes in Beta Decay • β- decay • β+ decay

Mass Changes in Beta Decay • EC decay Conclusion: All calculations can be done with atomic masses

Nomenclature • Sign convention: Q=(massesreactants-massesproducts)c 2 Q has the opposite sign as ΔH Q=+ exothermic Q=- endothermic
![Nomenclature • Total binding energy, Btot(A, Z)=[Z(M(1 H))+(A-Z)M(n)-M(A, Z)]c 2 • Binding energy per Nomenclature • Total binding energy, Btot(A, Z)=[Z(M(1 H))+(A-Z)M(n)-M(A, Z)]c 2 • Binding energy per](http://slidetodoc.com/presentation_image_h/935e4a66df047914e864ed85f804f91d/image-6.jpg)
Nomenclature • Total binding energy, Btot(A, Z)=[Z(M(1 H))+(A-Z)M(n)-M(A, Z)]c 2 • Binding energy per nucleon Bave(A, Z)= Btot(A, Z)/A • Mass excess (Δ) M(A, Z)-A See appendix of book for mass tables
![Nomenclature • Packing fraction (M-A)/A • Separation energy, S Sn=[M(A-1, Z)+M(n)-M(A, Z)]c 2 Sp=[M(A-1, Nomenclature • Packing fraction (M-A)/A • Separation energy, S Sn=[M(A-1, Z)+M(n)-M(A, Z)]c 2 Sp=[M(A-1,](http://slidetodoc.com/presentation_image_h/935e4a66df047914e864ed85f804f91d/image-7.jpg)
Nomenclature • Packing fraction (M-A)/A • Separation energy, S Sn=[M(A-1, Z)+M(n)-M(A, Z)]c 2 Sp=[M(A-1, Z-1)+M(1 H)-M(A, Z)]c 2
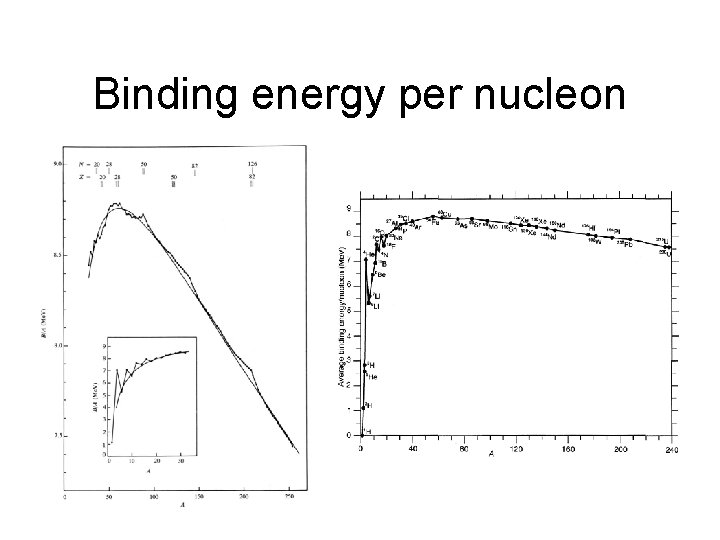
Binding energy per nucleon
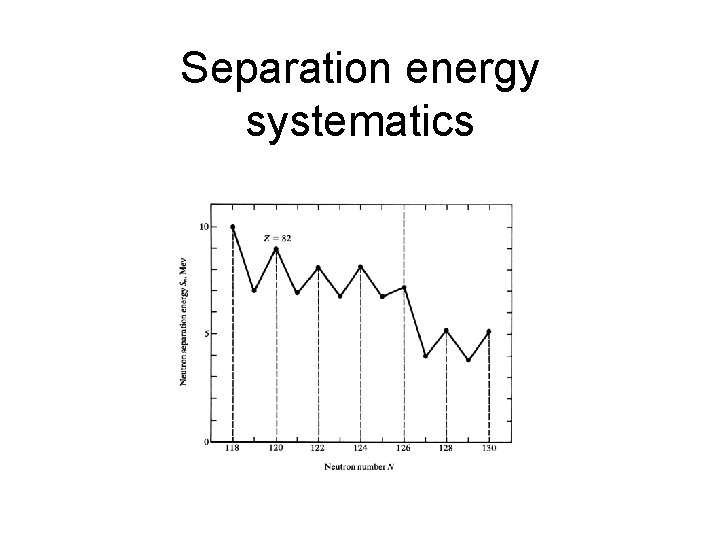
Separation energy systematics
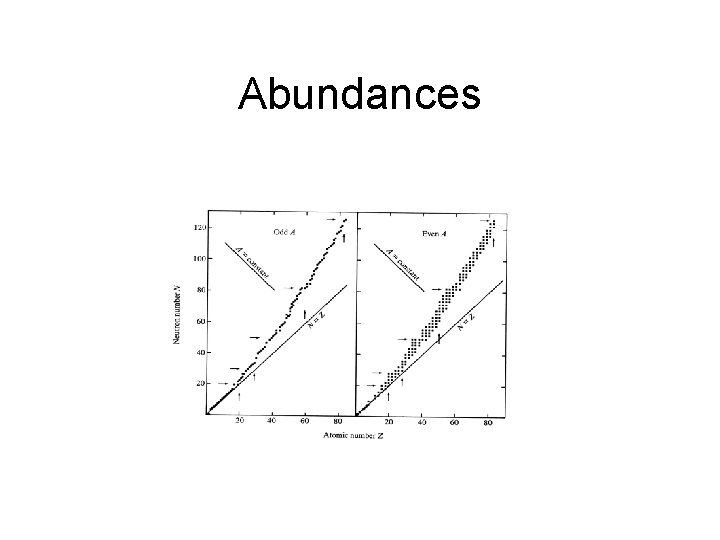
Abundances

Semi-empirical mass equation Terms • Volume • Surface • Coulomb av. A -as. A 2/3 -ac. Z 2/A 1/3

Asymmetry term To make AZ from Z=N=A/2, need to move q protons qΔ in energy, thus the work involved is q 2Δ=(N-Z)2Δ/4. If we add that Δ=1/A, we are done.
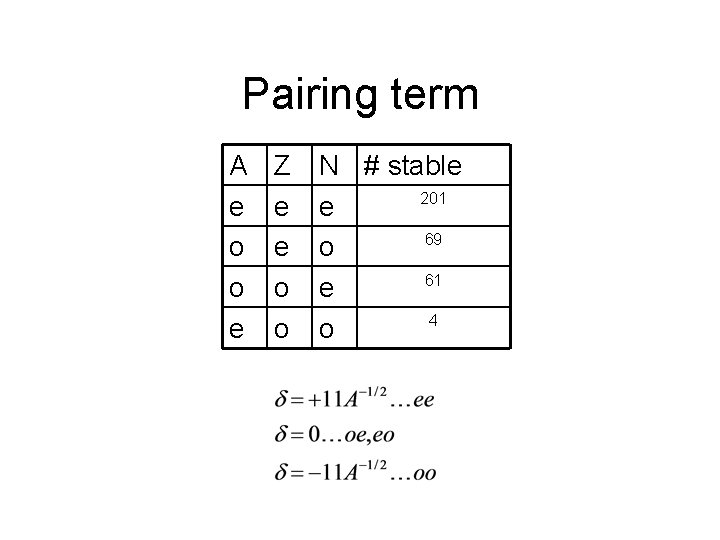
Pairing term A e o o e Z e e o o N # stable 201 e 69 o 61 e 4 o
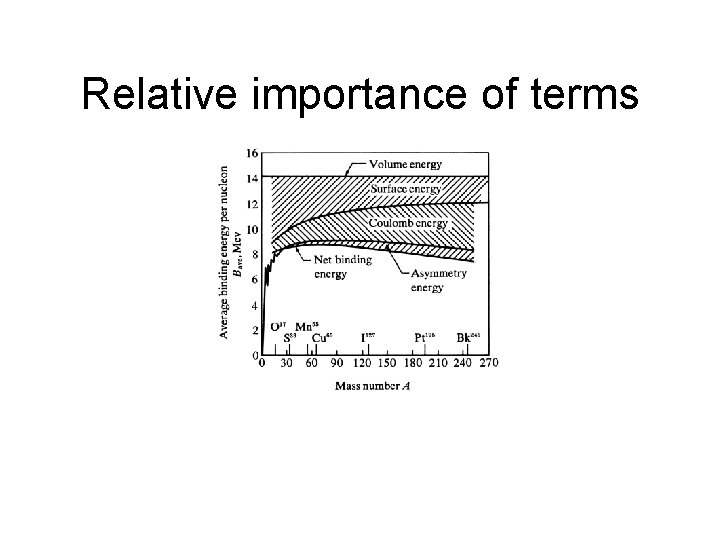
Relative importance of terms

Values of coefficients

Modern version of semiempirical mass equation (Myers and Swiatecki)


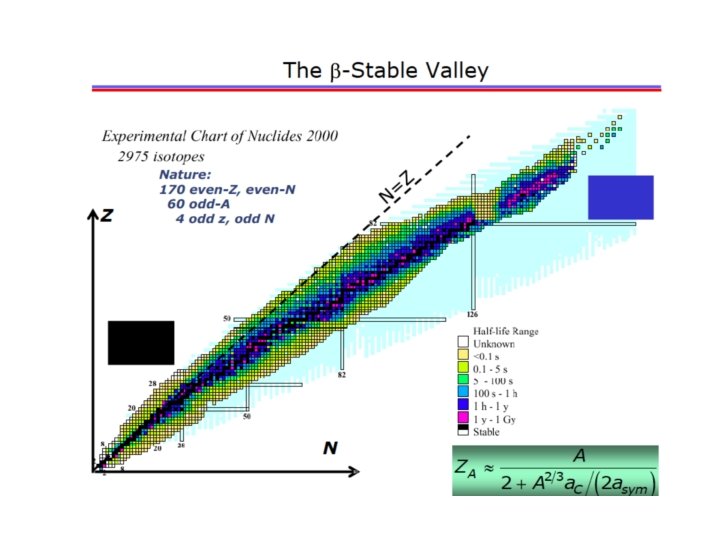
Mass parabolas and Valley of beta stability This is the equation of a parabola, a+b. Z+c. Z 2




Where is the minimum of the parabolas?
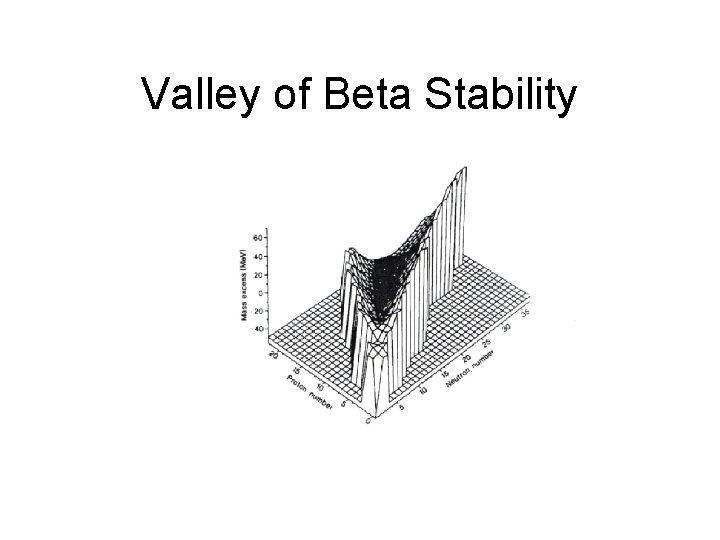
Valley of Beta Stability
- Slides: 24