NUCLEAR MAGNETIC RESONANCE spectroscopy NMR spectroscopy Alkhair Adam




























- Slides: 50

NUCLEAR MAGNETIC RESONANCE spectroscopy NMR spectroscopy Alkhair Adam Khalil, B. Pharm. , M. Pharm. Department of Pharmaceutical Chemistry College of Pharmacy – Karary University

In this lecture we are going to discus the following: • Precessional frequency • 1 H NMR Spectrum : – – – intensity of a signal chemical shifts or the δ (delta) scale The terms upfield and downfield The reference signal at 0 on the δ scale Equivalent and non-equivalent protons • Features of 1 H NMR Spectrum : – – Number of signals Position of signals Intensity of signals Spin–spin splitting of signals

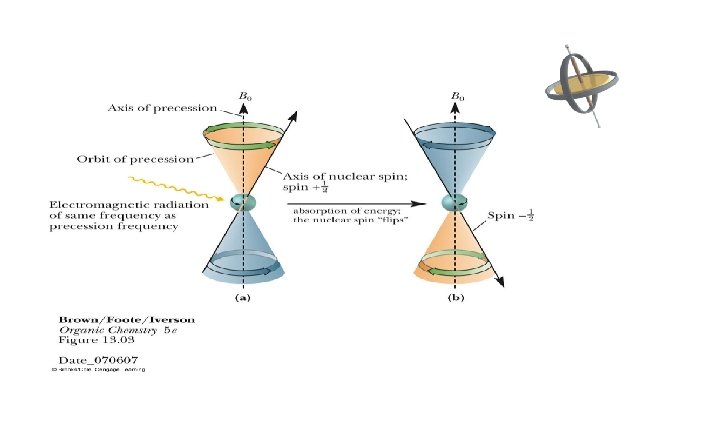
Precessional frequency: • Nuclei spin like a spinning top it will also have a slower kind of motion where the spinning axis of the top moves slowly around the vertical axis. This type is called precessional motion i. e. the top is precessing (or wobble or vibrate) around its vertical axis. 3

• If the precessing nucleus is irradiated with electromagnetic radiation of the same frequency as that frequency of precession nucleus, then – The two frequencies couple – Energy is absorbed – The nuclear spin is flipped from spin state +1/2 (with the applied field) to -1/2 (against the applied field). 4


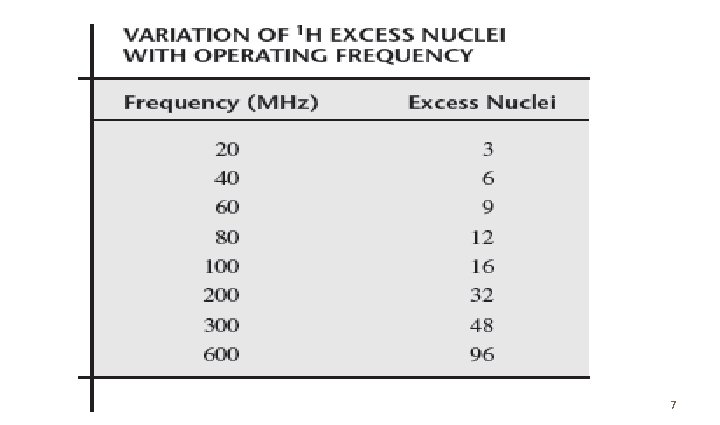
• At (60 MHz) for proton there are 1000009 nuclei in the lower (favored) spin for every 1000000 that occupy the upper spin state • If the number of lower and upper become the same we observe no signal, this called saturation. • Therefore, the very small excess of nuclei in the lower spin state is quite important to NMR spectroscopy. • Increasing the operating radio frequency increases the excess nuclei this why the modern instruments work at 300, 400 and even 600 MHz at research universities.

7

1 H NMR Spectrum

• An NMR spectrum plots the intensity of a signal against its chemical shift measured in parts per million (ppm). • The common scale of chemical shifts is called the δ (delta) scale.

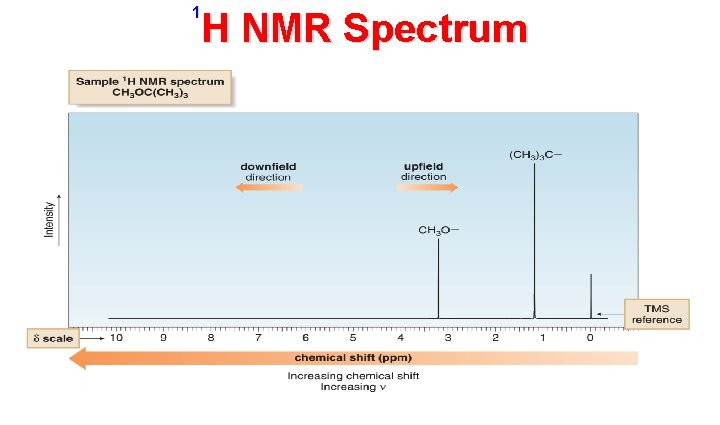
• NMR absorptions generally appear as sharp signals. • The H NMR spectrum of CH 3 OC(CH 3)3 consists of two signals: • A tall peak at 1. 2 ppm due to the (CH 3)3 C – group, and, • A smaller peak at 3. 2 ppm due to the CH 3 O – group. • Increasing chemical shift is plotted from right to left. • Most protons absorb somewhere from 0– 12 ppm. 1

• The terms upfield and downfield describe the relative location of signals. • Upfield means to the right. • The (CH ) C – peak is upfield from CH O • Downfield means to the left. • The CH O – peak is downfield from the ((CH ) C –) peak. 3 3 3

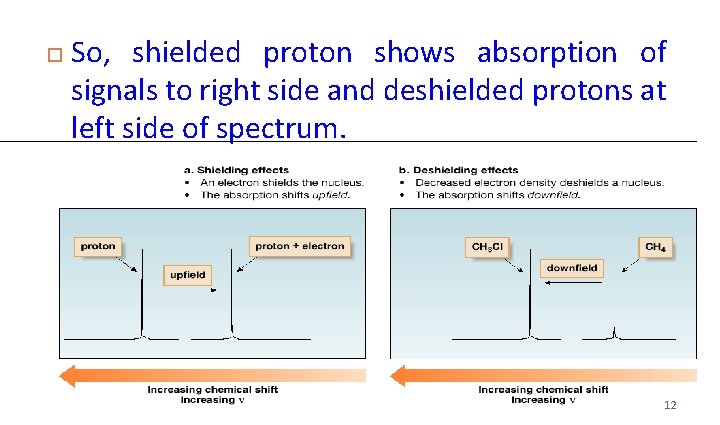
So, shielded proton shows absorption of signals to right side and deshielded protons at left side of spectrum. 12

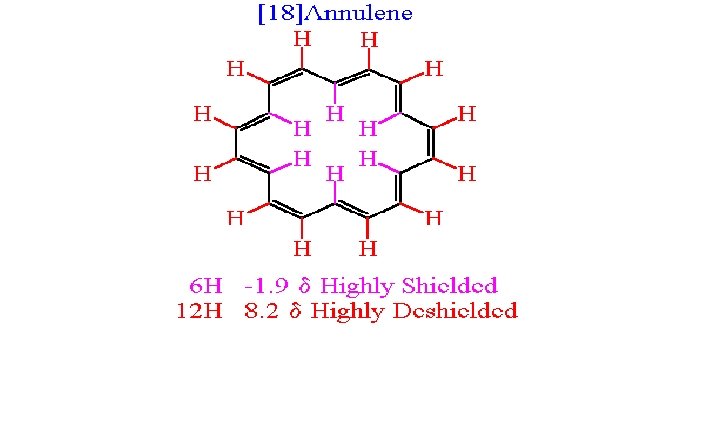
• NMR absorptions are measured relative to the position of a reference signal at 0 ppm on the δ scale due to Tetra-Methyl-Silane (TMS) ((CH ) Si ). • TMS is a volatile and inert compound that gives a single peak upfield from most organic compounds in NMR spectra. • A very small number of organic compound’s absorptions occur upfield from the TMS signal, which is defined as the negative direction of the δ scale (e. g. 18 -Annulene ). 3 4


The chemical shift • The chemical shift on the x axis gives the position of an NMR signal, measured in ppm, according to the following equation:

• A chemical shift gives absorptions as a fraction of the NMR operating frequency, making it independent of the spectrometer used to record a spectrum. • Because the frequency of the radiation required for resonance is proportional to the strength of the applied magnetic field, B , reporting NMR absorptions in frequency is meaningless, unless the value of B is also reported. • By reporting the absorption as a fraction of the NMR operating frequency, though, we get units (ppm) that are independent of the spectrometer. 0 0

Example: • Calculate the chemical shift of an absorption that occurs at 1500 Hz downfield from TMS using a 300 MHz NMR spectrometer.

Problems • The H NMR spectrum of CH OH recorded on a 500 MHz NMR spectrometer consists of two signals, one due to the CH protons at 1715 Hz and one due to the OH proton at 1830 Hz, both measured downfield from TMS. • (a) Calculate the chemical shift of each absorption. • (b) Do the CH protons absorb upfield or downfield from the OH proton? 1 3 3 3

Problems • The H NMR spectrum of 1, 2 -dimethoxyethane (CH OCH ) recorded on a 300 MHz NMR spectrometer consists of signals at 1017 Hz and 1065 Hz downfield from TMS. • (a) Calculate the chemical shift of each absorption. • (b) At what frequency would each absorption occur if the spectrum were recorded on a 500 MHz NMR spectrometer? 1 3 2 2 3

Features of H NMR 1 1 H NMR spectrum provide information about a compound’s structure: • [1] Number of signals • [2] Position of signals • [3] Intensity of signals • [4] Spin–spin splitting of signals

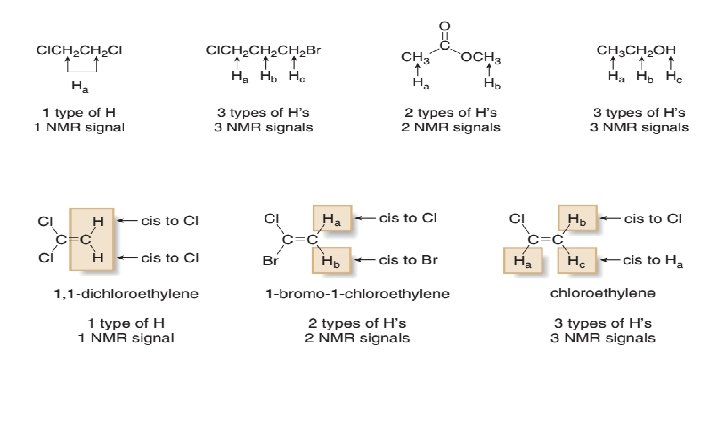
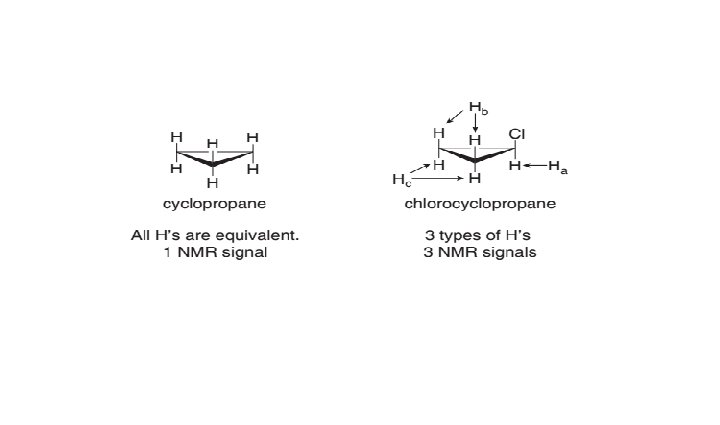
H NMR: Number of Signals 1 • The number of NMR signals equals the number of different types of protons in a compound.




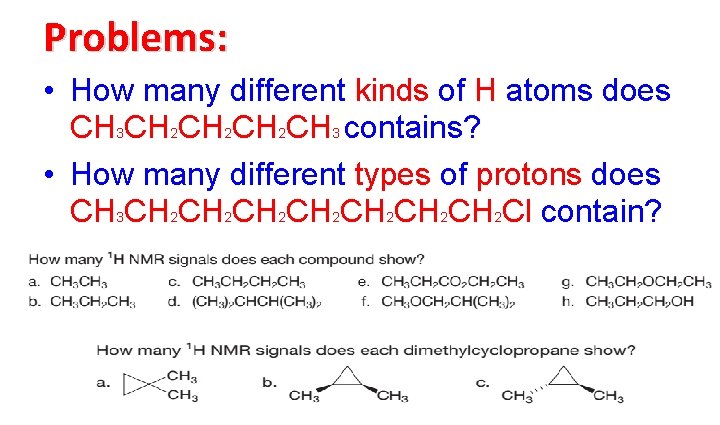
Problems: • How many different kinds of H atoms does CH 3 CH 2 CH 2 CH 3 contains? • How many different types of protons does CH 3 CH 2 CH 2 CH 2 CH 2 Cl contain?

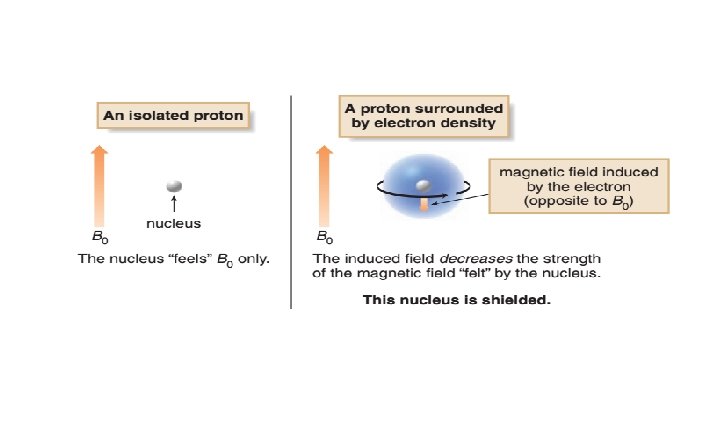
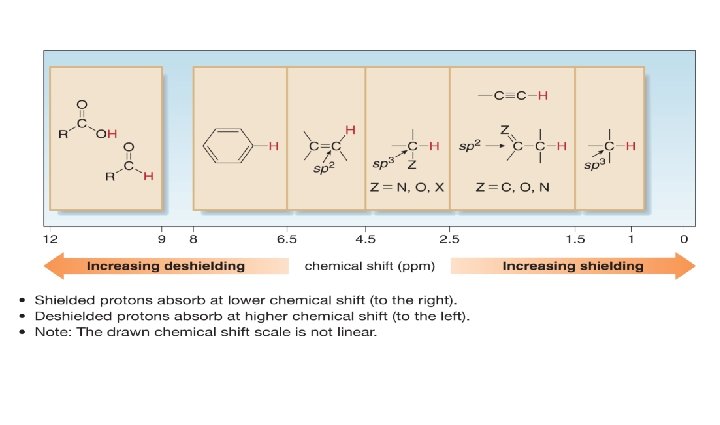
H NMR: Position of Signals 1 • Where a particular proton absorbs depends on its electronic environment. • To understand how the electronic environment around a nucleus affects its chemical shift, recall that in a magnetic field, an electron creates a small magnetic field that opposes the applied magnetic field, B 0. • Electrons are said to shield the nucleus from B 0.


• Because the shielded proton experiences a lower magnetic field strength, it needs a lower frequency to achieve resonance. • Lower frequency is to the right in an NMR spectrum, toward lower chemical shift, so shielding shifts an absorption upfield.


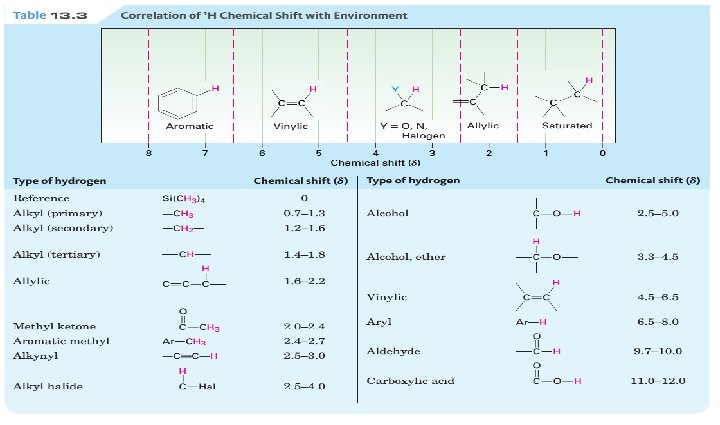
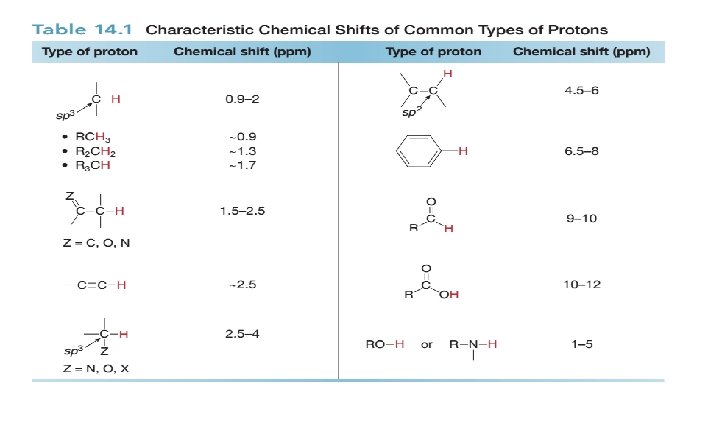
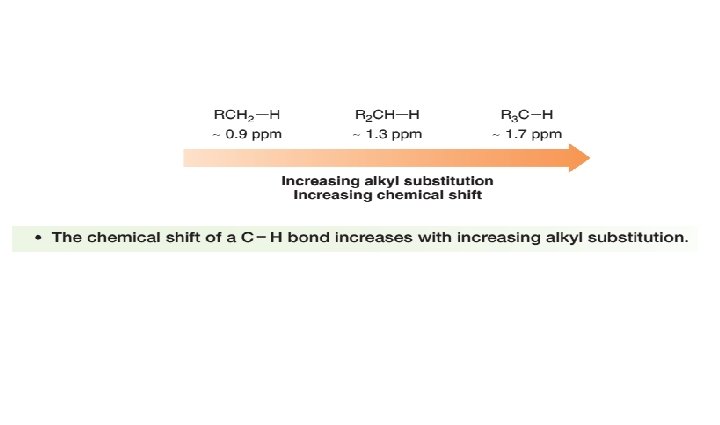
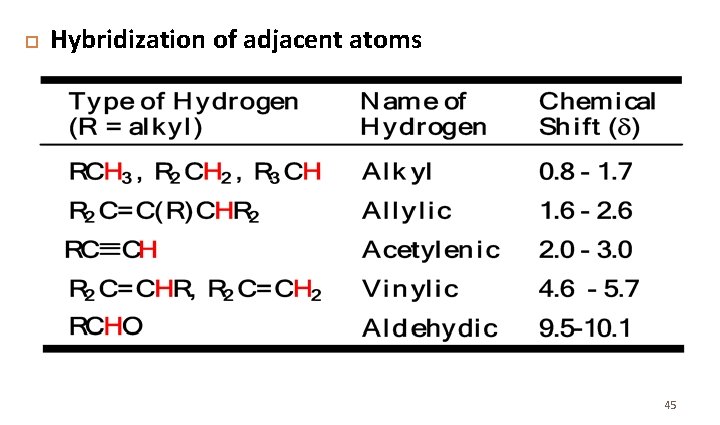
Chemical Shift Values • Not only is the relative position of NMR absorptions predictable, but it is also possible to predict the approximate chemical shift value for a given type of proton.




Problems:

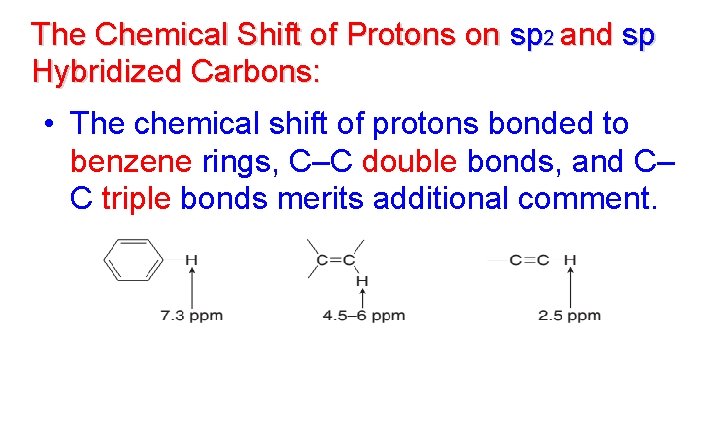
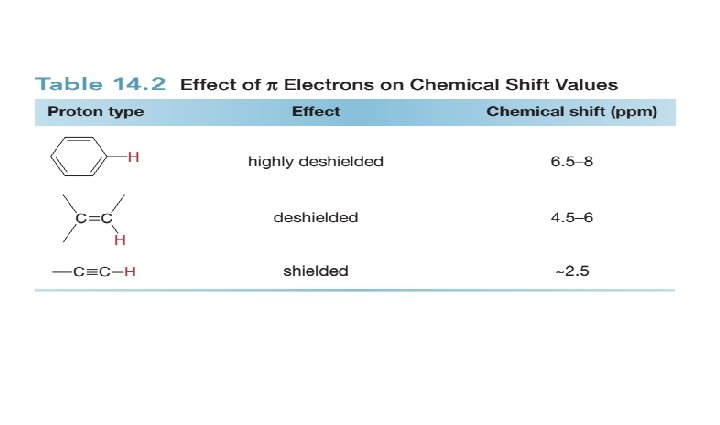
The Chemical Shift of Protons on sp 2 and sp Hybridized Carbons: • The chemical shift of protons bonded to benzene rings, C–C double bonds, and C– C triple bonds merits additional comment.

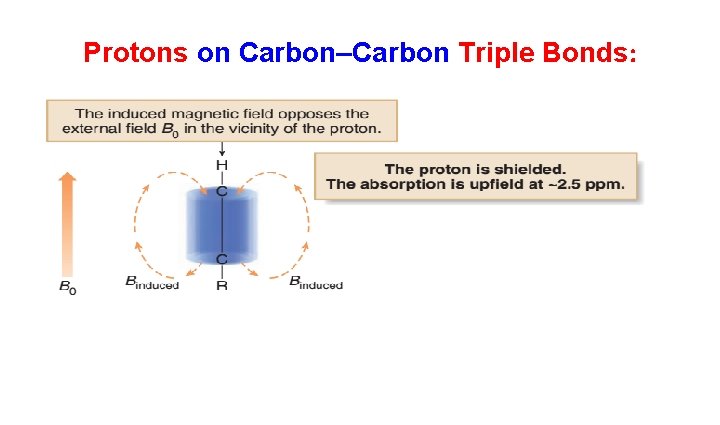
• Each of these functional groups contains bonds with loosely held electrons. • When placed in a magnetic field, these electrons move in a circular path, inducing a new magnetic field. • How this induced magnetic field affects the chemical shift of a proton depends on the direction of the induced field in the vicinity of the absorbing proton.

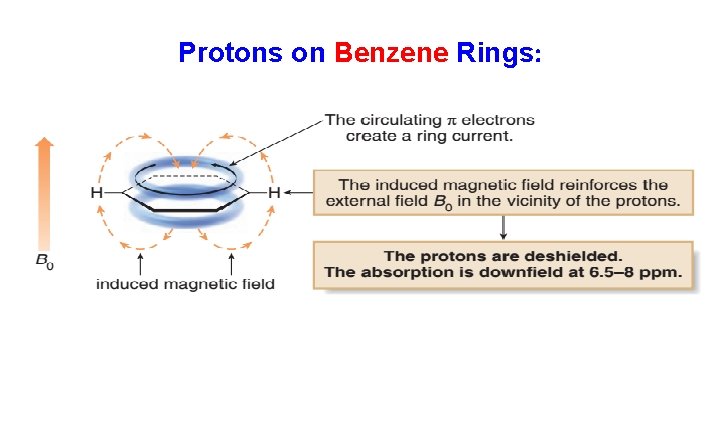
Protons on Benzene Rings:

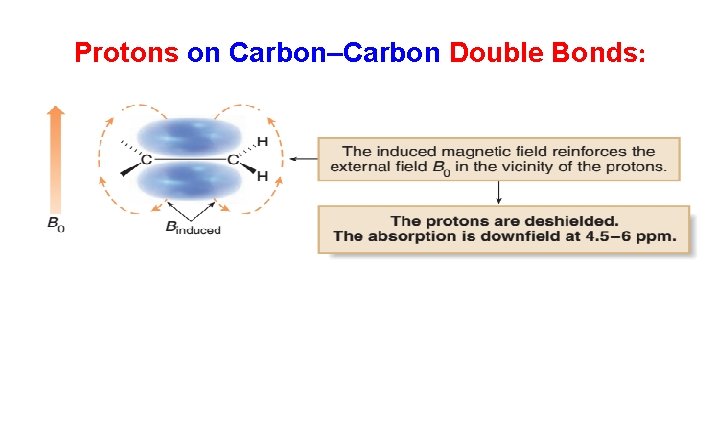
Protons on Carbon–Carbon Double Bonds:

Protons on Carbon–Carbon Triple Bonds:



Therefore in conclusion

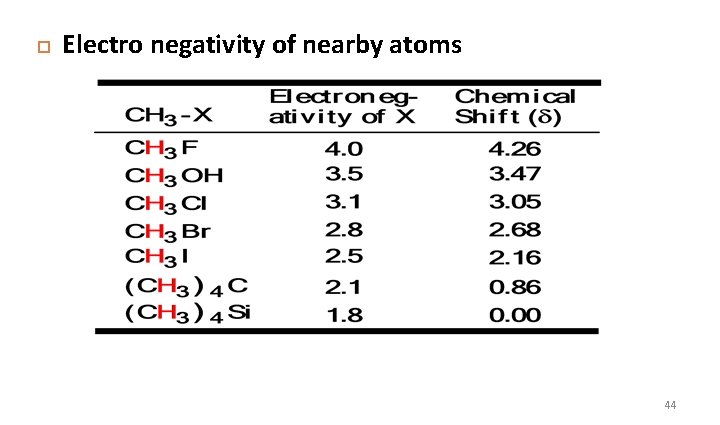
Chemical shift depends upon following parameters: • Electro negativity of nearby atoms • Hybridization of adjacent atoms • Diamagnetic effects from adjacent pi bonds 43

Electro negativity of nearby atoms 44

Hybridization of adjacent atoms 45

Diamagnetic effects from adjacent pi bonds 46

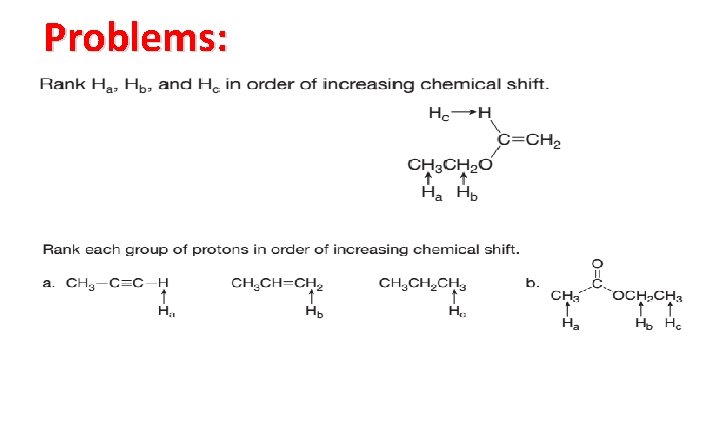
Problems:

Questions !

H NMR: Intensity of Signals 1 • To be continued next lecture.
