Nuclear Fusion Before we discuss what nuclear fusion

Nuclear Fusion

Before we discuss what nuclear fusion is, we must first review some concepts about atoms.

Composition of The Atom • The atom is made of three parts: – Protons – Neutrons – Electrons
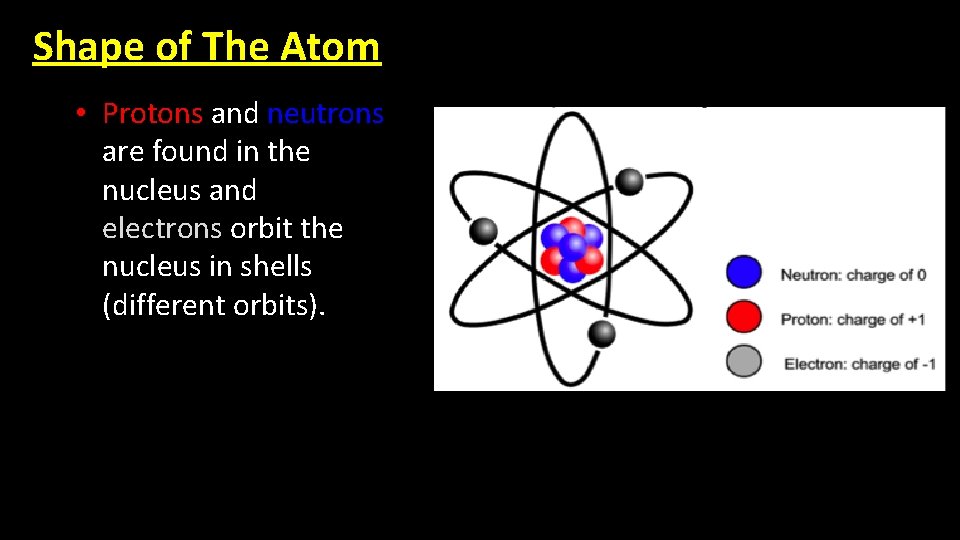
Shape of The Atom • Protons and neutrons are found in the nucleus and electrons orbit the nucleus in shells (different orbits).

Charges of the Different Subatomic Particles • Protons have a positive charge. • Electrons have a negative charge. • Neutrons have no charge.
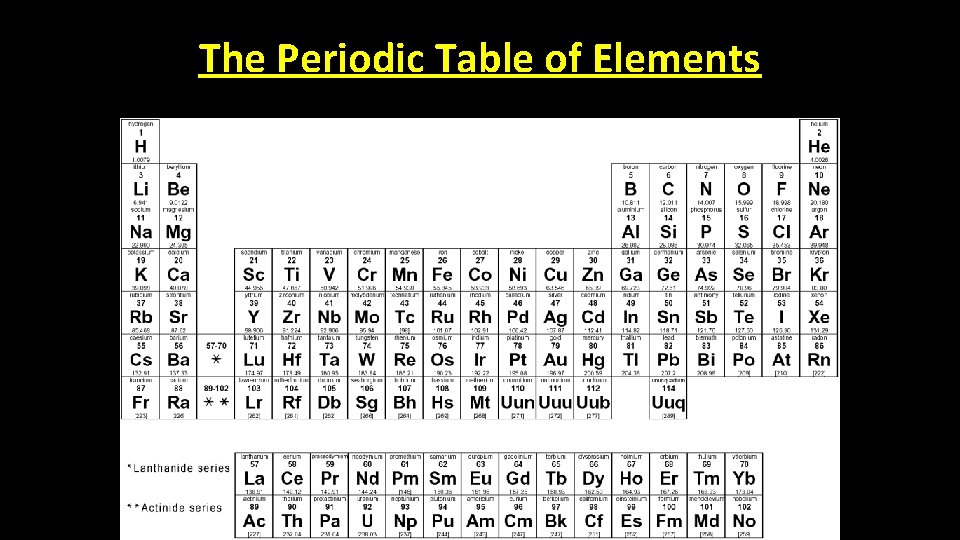
The Periodic Table of Elements

• There is a lot of information on the Periodic Table of Elements. Atomic Number • For this discussion we are only going to focus on one thing, the atomic number.

Atomic Number • The atomic number represents the number of protons found in the atom. Helium has an atomic number of 2 so it has 2 protons The atomic number of Hydrogen is 1 so it has 1 proton How many protons does lithium have?

The Sun • The sun, like all stars, is composed mostly of hydrogen.

Nuclear Fusion • Remember that a hydrogen atom is made of one proton, which has a positive charge • For this reason hydrogen atoms normally repel each other Model of a hydrogen atom.

Nuclear Fusion • However inside of the core of a star the hydrogen atoms are under such extreme pressure and temperature. • Under such extreme conditions the hydrogen atoms are fused (joined) together creating a helium atom and energy (photons).
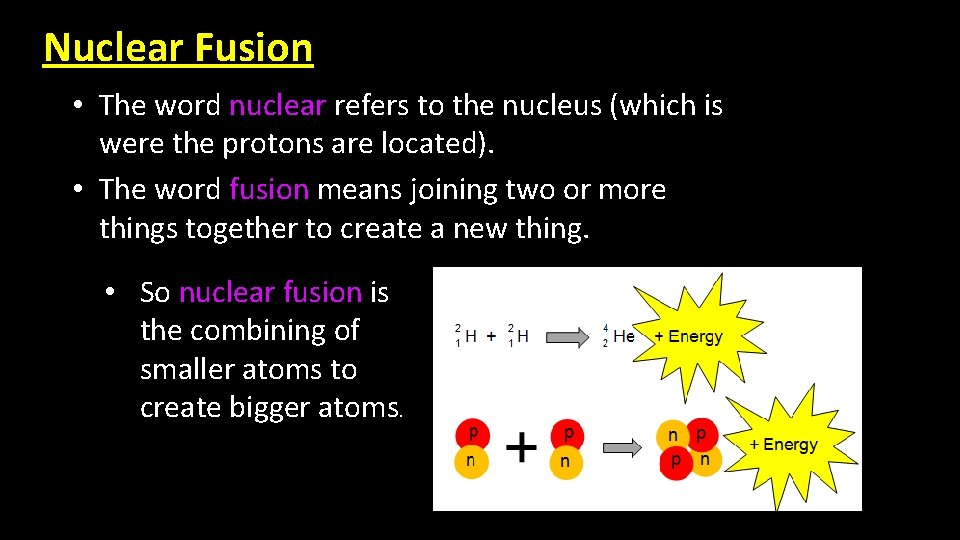
Nuclear Fusion • The word nuclear refers to the nucleus (which is were the protons are located). • The word fusion means joining two or more things together to create a new thing. • So nuclear fusion is the combining of smaller atoms to create bigger atoms.
Nuclear Fusion in Stars • For more than 90% of the life of a star nuclear fusion converts hydrogen into helium. • This process also releases a tremendous amount of energy called photons. • Photons travel through electromagnetic waves as heat and light.
- Slides: 13