Nuclear Fission and Fusion Webquest Use the links

Nuclear Fission and Fusion Webquest ● Use the links and the slides below to help you answer the questions in the Webquest ● When you are ready to complete your Nuclear Energy Ad check in FIRST with Mrs. Gaines : )

Fission, Fusion, Binding Energy, and Half Life
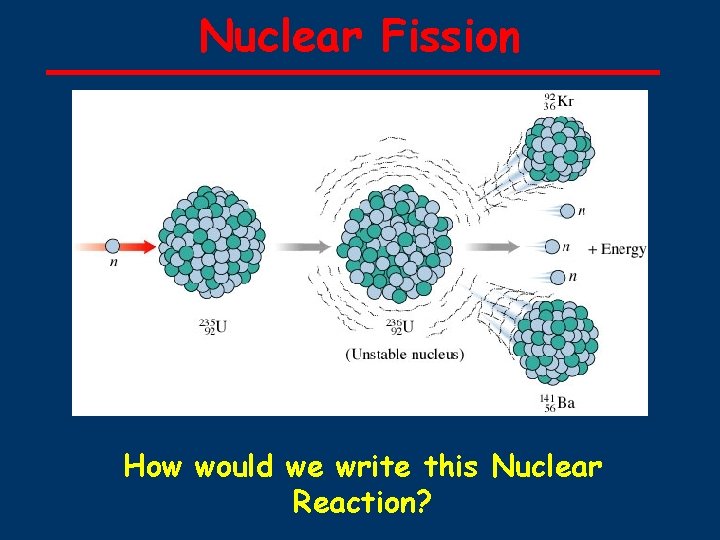
Nuclear Fission How would we write this Nuclear Reaction?

Uranium Isotopes ■ ■ ■ Naturally occurring Uranium contains two major isotopes Uranium-238 (99. 3%) Uranium-235 (0. 7%) As it turns out the only isotope of Uranium that undergoes fission is Uranium-235 Why do you think U-235 can undergo fission?

235 U ■ ■ ■ ■ Fission 1 n → 236 U* U + 92 0 92 and 10 -14 seconds later. . . 236 U* → 92 Kr + 141 Ba + 3 1 n + ENERGY 92 36 56 0 50 possible sets of fission products (sum of atomic numbers = 92) 3 neutrons released for ONE 23592 U each neutron can split another 23592 U CHAIN REACTION POSSIBLE If amount of 23592 U is sufficient (CRITICAL MASS) then the number of neutrons generated is high enough to result in a nuclear explosion ) 235

235 U 235 92 U + 1 Fission 92 Kr + n → 0 36 141 1 n Ba + 3 56 0 + E
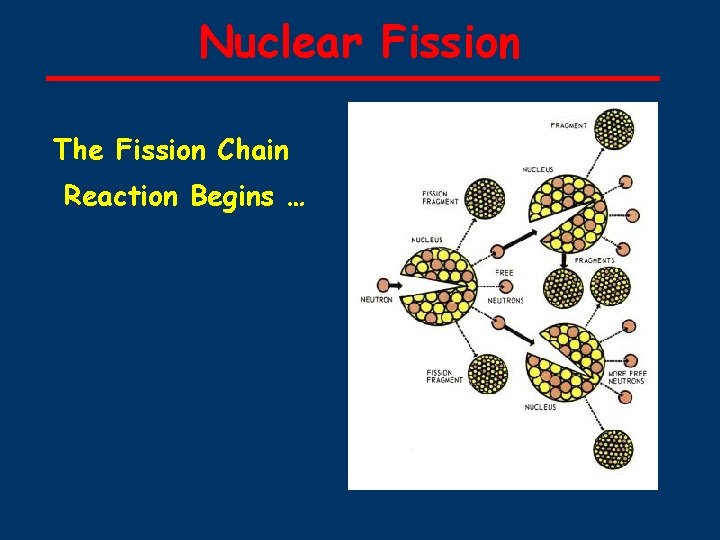
Nuclear Fission The Fission Chain Reaction Begins …
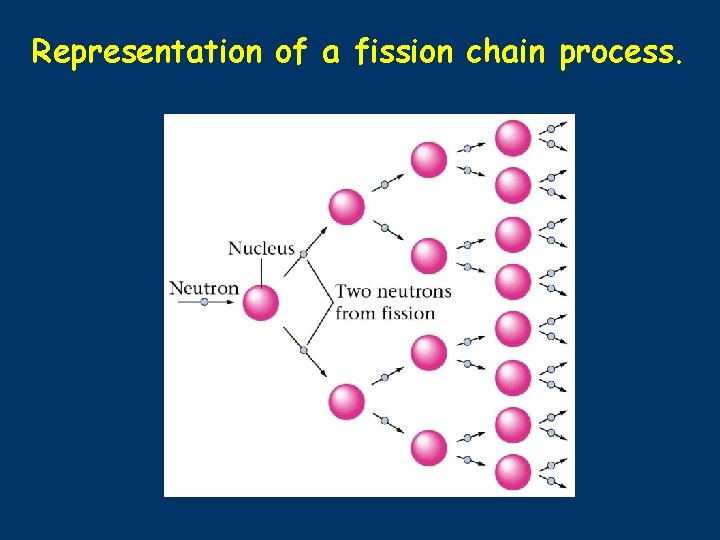
Representation of a fission chain process.
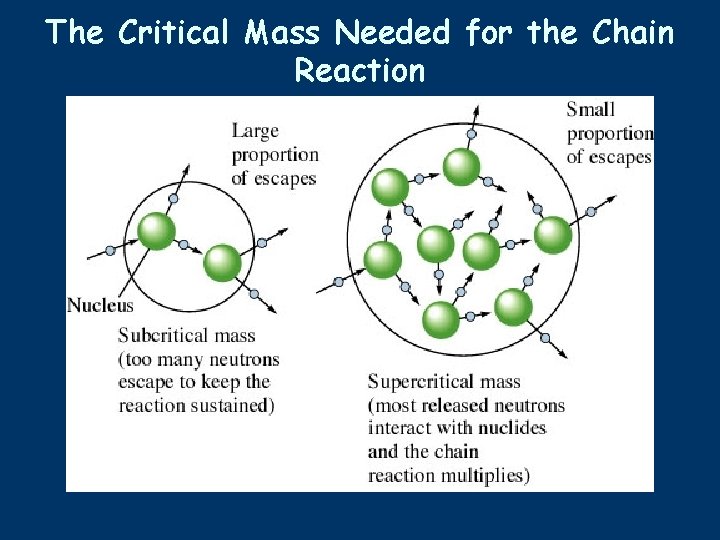
The Critical Mass Needed for the Chain Reaction

Are there other Fuels for the Fission Chain Reaction? ■ Uranium-235. (Keep in mind, the abundant form of Uranium found in nature is Uranium-238 (~99%). ■ Plutonium-239 … but Plutonium-239 is scarce … now what? ■ Here’s what scientists discovered during World War II: 238 92 U + 239 92 U 239 93 Np 1 0 n ---> 239 93 Np ---> 239 92 U + 0 + γ -1β 94 Pu + 0 -1β

Development of the Atomic Bomb • During WW 2 the American set up a secret project called the “Manhattan Project” recruiting all the best physicists and nuclear chemists available • By 1945 they developed two types of nuclear bombs • A U-235 fission bomb • and a Pu-239 fission bomb

Little Boy and Fat Man 10 feet

The Arrangement of Uranium in the First Atomic Bombs

GHS Honors Chem

Arranging U-235 to get a Controlled Nuclear Reaction ■ ■ ■ Arrangement of U-235 in a critical mass, where there’s enough U-235 in a small space so that the neutrons cause a chain reaction, would be difficult to control in a nuclear power plant. How can this conversion of small amounts of mass into great amounts of energy be used to produce electricity? In the 1930, Enrico Fermi, an Italian Physicist, discovered that Uranium can be arranged in what he called a Fission Pile, where the Uranium would be arranged in rows, or in a lattice, so that the fission reaction could be controlled.
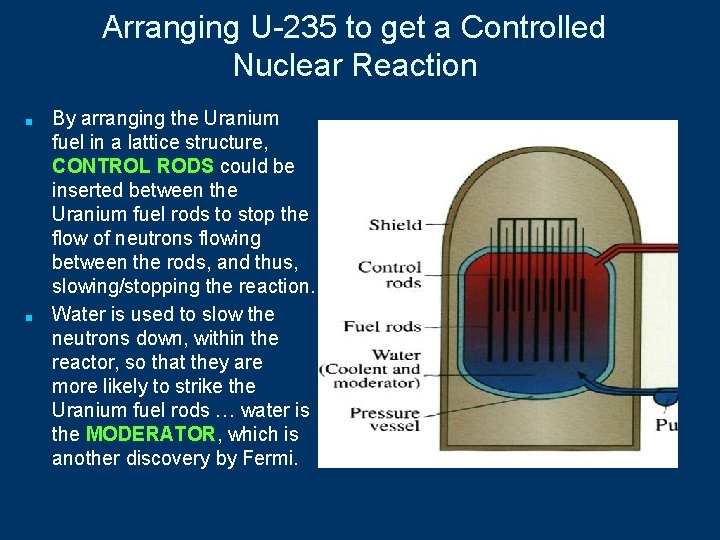
Arranging U-235 to get a Controlled Nuclear Reaction ■ ■ By arranging the Uranium fuel in a lattice structure, CONTROL RODS could be inserted between the Uranium fuel rods to stop the flow of neutrons flowing between the rods, and thus, slowing/stopping the reaction. Water is used to slow the neutrons down, within the reactor, so that they are more likely to strike the Uranium fuel rods … water is the MODERATOR, which is another discovery by Fermi.

Arranging U-235 to get a Controlled Nuclear Reaction ■ ■ By arranging the Uranium fuel in a lattice structure, there is NO POSSIBILITY of an uncontrolled nuclear explosion, like in a nuclear warhead or bomb. Simply stated, there Uranium is not in a close-enough proximity for an uncontrolled chain reaction, or blast. So, this is an important point: the threat of a nuclear explosion, within a nuclear power plant, is an imagined danger.
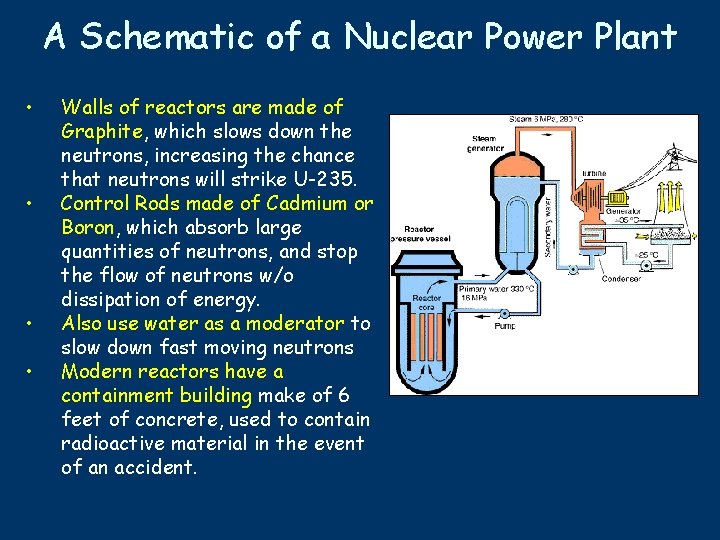
A Schematic of a Nuclear Power Plant • • Walls of reactors are made of Graphite, which slows down the neutrons, increasing the chance that neutrons will strike U-235. Control Rods made of Cadmium or Boron, which absorb large quantities of neutrons, and stop the flow of neutrons w/o dissipation of energy. Also use water as a moderator to slow down fast moving neutrons Modern reactors have a containment building make of 6 feet of concrete, used to contain radioactive material in the event of an accident.
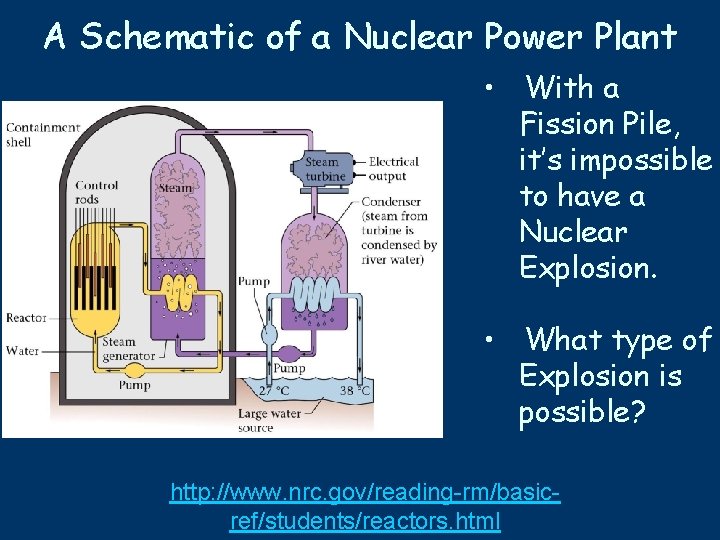
A Schematic of a Nuclear Power Plant • With a Fission Pile, it’s impossible to have a Nuclear Explosion. • What type of Explosion is possible? http: //www. nrc. gov/reading-rm/basicref/students/reactors. html
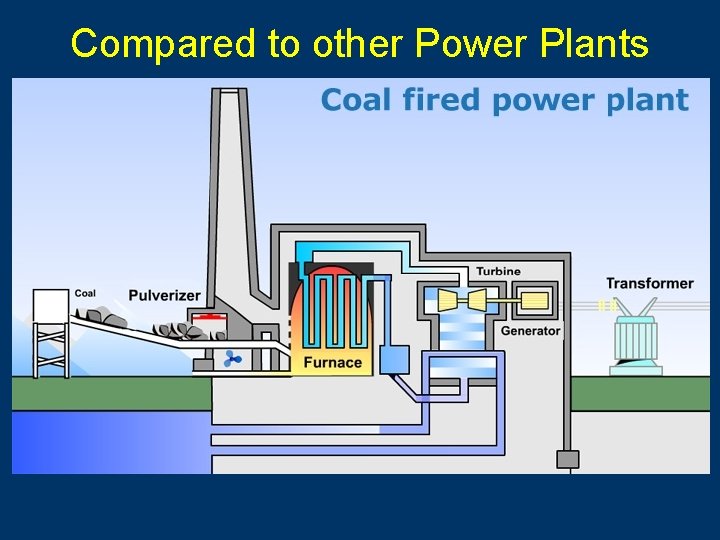
Compared to other Power Plants
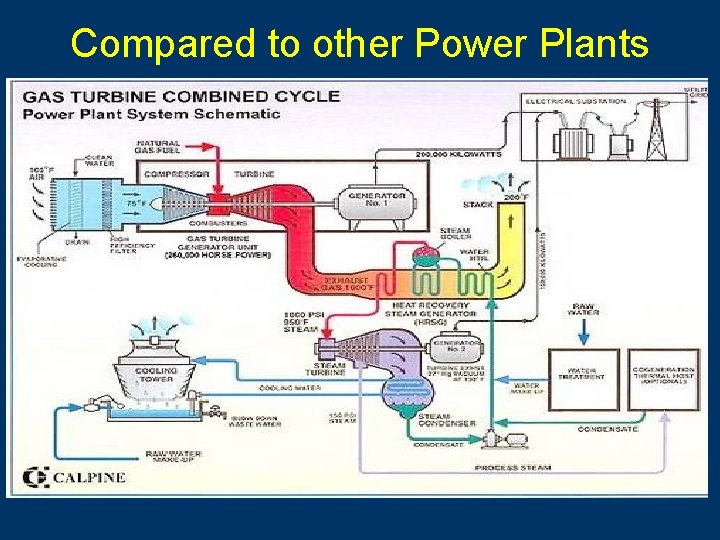
Compared to other Power Plants

Nuclear Fission & POWER ■ Currently about 103 nuclear power plants in the U. S. and about 435 worldwide. ■ 17% of the world’s energy comes from nuclear.

We’ll take a closer look at Nuclear Power Plant Mishaps in a bit …

Nuclear Fusion small nuclei combine 2 H 1 + 3 H 4 He 1 2 + 1 n + 0 Occurs in the sun and other stars Energy

Nuclear Fusion ■ Excessive heat can not be contained ■ Attempts at “cold” fusion have FAILED. ■ “Hot” fusion is difficult to contain

Recall Mass Defect It is experimentally observed that the mass of an atom (containing neutrons) is always slightly less than the sum of the masses of its component particles. The difference between the atomic mass and the sum of the masses of its protons, neutrons, and electrons is called the mass defect.

Can we calculate the Energy given off by a nuclear reaction based on the weight difference from reactants to products based on Einstein’s Equation, E = mc 2?

Calculation of Nuclear Energy Released Calculate the energy released (per mole of tritium consumed) for the fusion reaction of tritium and deuterium, given the following molar masses of nucleons and nuclei. (c = 3. 00 × 108 m/s) Mass Reactants = 3. 01605 + 2. 0140 g particle mass (g/mol) proton 1. 007825 neutron 1. 008665 deuterium 2. 0140 tritium 3. 01605 helium 4. 00260 Mass Reactants = 5. 03005 g Mass Products = 4. 00260 + 1. 008665 g Mass Products = 5. 011265 g Mass Diff. = Mass. Reactants – Mass. Products Mass Difference = 0. 018785 g Mass Difference = 1. 8785 x 10 -5 kg E = mc 2 = 1. 8785 x 10 -5 kgx(3 x 108 m/s)2 E = 1. 69 x 1012 kg-m 2/s 2 = 1. 69 x 1012 J

Half-Life ■ ■ HALF-LIFE is the time that it takes for 1/2 a sample to decompose. The rate of a nuclear transformation depends only on the “reactant” concentration.
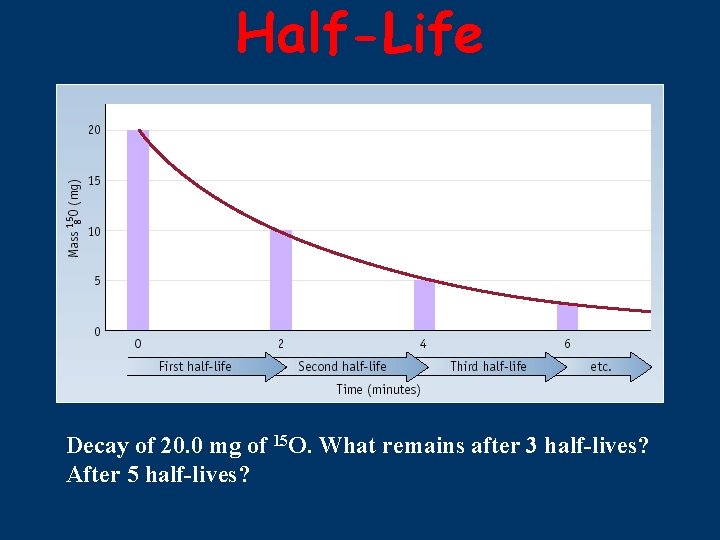
Half-Life Decay of 20. 0 mg of 15 O. What remains after 3 half-lives? After 5 half-lives?

Kinetics of Radioactive Decay For each duration (half-life), one half of the substance decomposes. For example: Ra-234 has a half-life of 3. 6 days If you start with 50 grams of Ra-234 After 3. 6 days > 25 grams After 7. 2 days > 12. 5 grams After 10. 8 days > 6. 25 grams

Half-Life of Radioisotopes Half–life is the time required for one-half of any given quantity of isotope to decay

Learning Check! The half life of I-123 is 13 hr. How much of a 64 mg sample of I-123 is left after 39 hours?

Learning Check! The half life of I-123 is 13 hr. Only 4 mg are present from a sample that had originally contained 128 mg sample of I 123. How old is the sample?

Learning Check! The half life of I-123 is 13 hr. Only 2 mg are present after 65 days have passed. What was the original amount of sample present?

How Old is Oetzi, the Iceman? Oetzi the Iceman was discovered on the border between Italy and Austria in 1991, in the Otztal Alps, partially submerged in a melting glacier. • It’s estimated that he is from the Neolithic age, some 5300 years ago.

How Old is Oetzi, the Iceman? • Oetzi was discovered in an extraordinarily warm summer season, which led to abnormal melting of glacier ice. • It’s estimated that Oetzi, along with animals and plants from his time, were covered by a sudden, deep snow, followed by a prolonged cold period. • Examination of the body suggests that Oetzi died from an arrow wound to his back/shoulder. How can we estimate his age … using the Half life of C 14.

Radiocarbon (C-14) Dating ■ Radioactive C-14 is formed in the upper atmosphere by nuclear reactions initiated by neutrons in cosmic radiation 14 N + 1 n ---> 14 C + 1 H 7 o 6 1 ■ The C-14 is oxidized to CO 2, which circulates through the biosphere. When a plant dies, the C-14 is not replenished. But the C-14 continues to decay with t 1/2 = 5730 years. Activity of a sample can be used to date the sample. ■ ■ ■

How Old is Oetzi, the Iceman? • How much C-14 would be remaining in Oetzi’s bones, given his estimated age of 5300 years? The T 1/2 of C-14 is 5730 years.

C-14 Dating Solves The Shroud of Turin (Torino) Question …

The Shroud of Turin (Torino) ■ ■ ■ The Shroud bearing the image of Jesus should be approximately 2000 years old C-14 analysis revealed the presence of 92% of the original amount of C-14 This decrease of 8% corresponds to a time period of about 700 years

Learning Check! The C-14 content of a fossilized bone is 12. 5% of that of a live bone. Knowing that the t 1/2 of C-14 is 5730 years, how old is the artifact?
- Slides: 42