Nuclear Energy 2 mc E Eenergy m mass

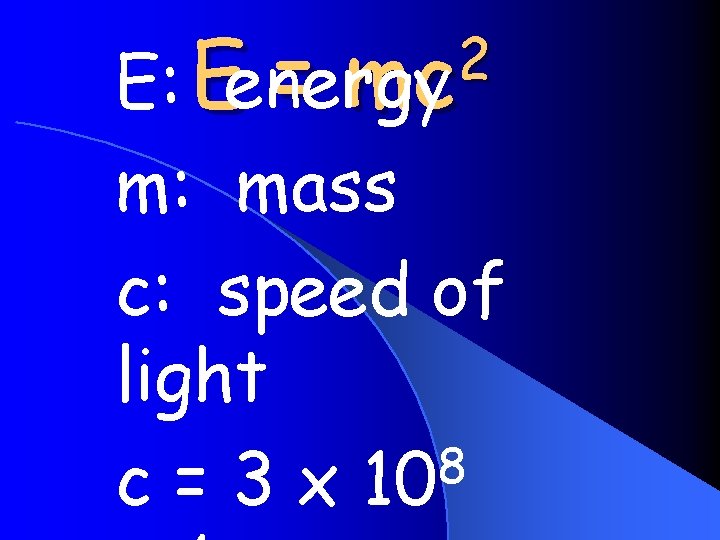






















- Slides: 35

Nuclear Energy
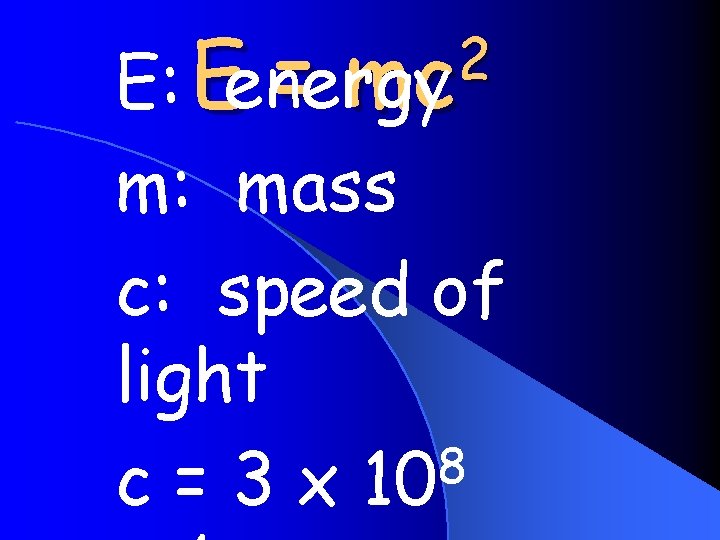
2 = mc E: Eenergy m: mass c: speed of light 8 c = 3 x 10

E= l. Mass 2 mc can be converted to energy l. Energy can be converted to mass l. Mass and energy are the same thing

Mass Defect of Alpha Particle Mass difference = 0. 0304 u Fusion product than the sum of the parts. Binding energyhas = less 28. 3 mass Me. V

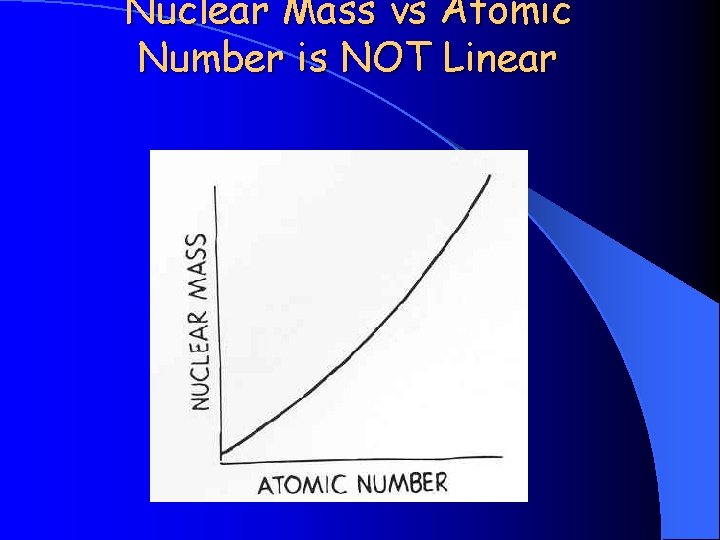
Nuclear Mass vs Atomic Number is NOT Linear

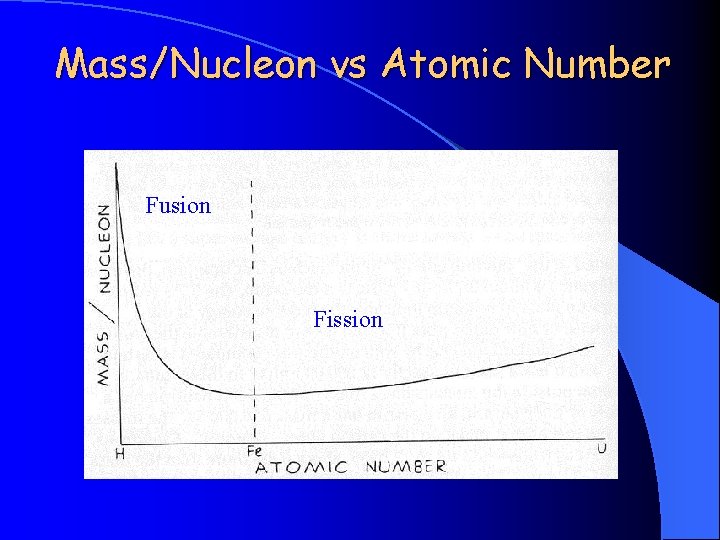
Mass/Nucleon vs Atomic Number Fusion Fission

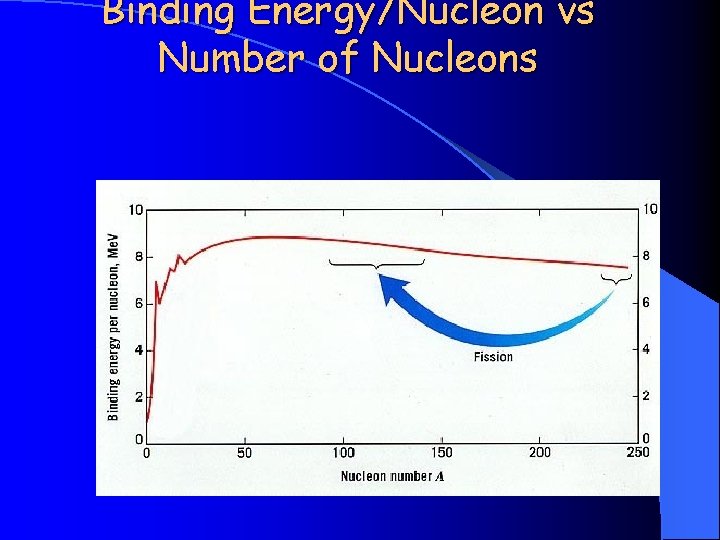
Binding Energy/Nucleon vs Number of Nucleons

Mass Defect in Fission l When a heavy element (one beyond Fe) fissions, the resulting products have a combined mass which is less than that of the original nucleus.

U-235 – Neutron Bombardment

U-235 -- Fissile

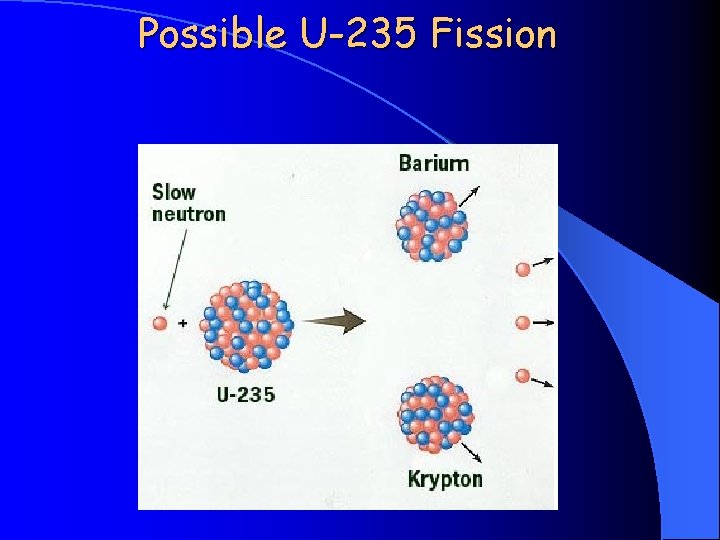
Possible U-235 Fission

How Stuff Works Site l Visit the How Stuff Works Site to learn more details about nuclear energy

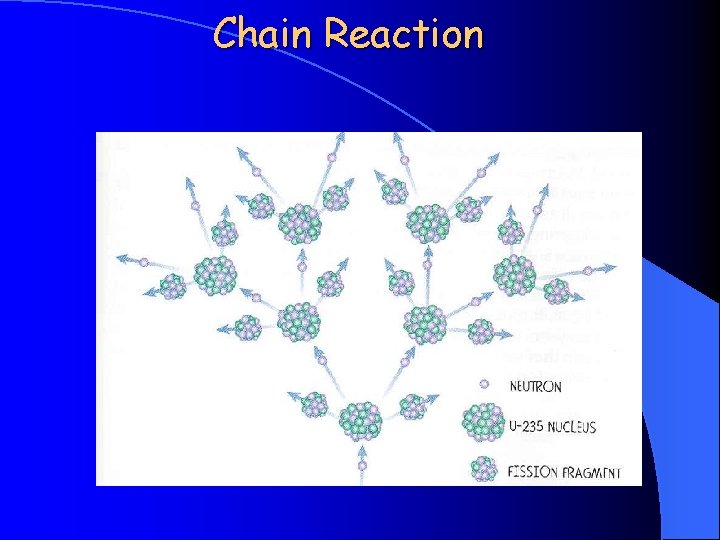
Chain Reaction

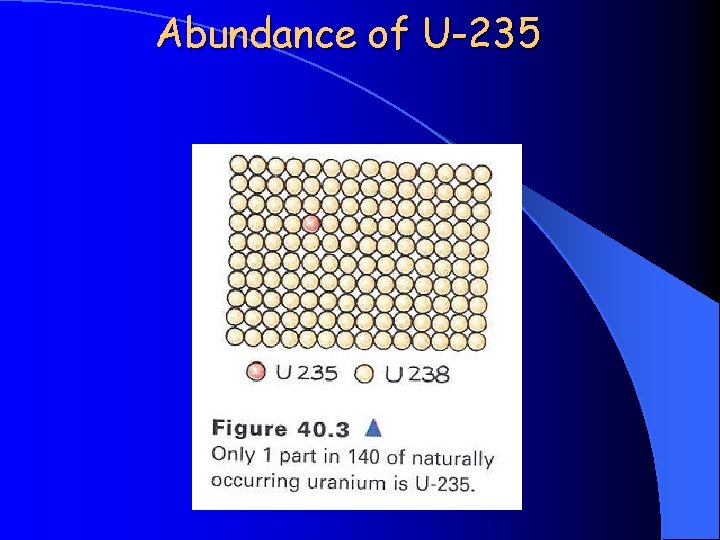
Abundance of U-235

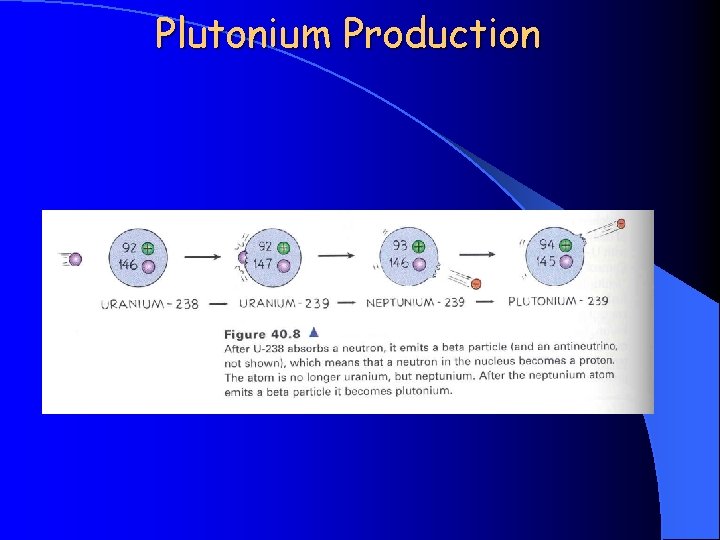
Plutonium Production

U-238 – Not Fissile

Breeder Reaction

Breeder Reactor l Small amounts of Pu-239 combined with U-238 l Fission of Pu frees neutrons l These neutrons bombard U-238 and produce more Pu-239 in addition to energy

Plutonium l Plutonium is fissile l Half-life of Plutonium is 24, 000 years l Plutonium can be chemically separated from uranium – easier than separating U-235 from U-238 l Plutonium is chemically poisonous (just like lead and arsenic)

l Plutonium attacks the nervous system and can cause paralysis (or death in sufficient doses) l Plutonium combines rapidly with oxygen to form compounds which are chemically biologically harmless l Plutonium is always radioactively toxic – worse than uranium but not as bad as radium l Plutonium emits high energy alphas

Einstein Prediction l "I do not know with what weapons World War 3 will be fought, but World War 4 will be fought with sticks and stones. " A. Einstein l

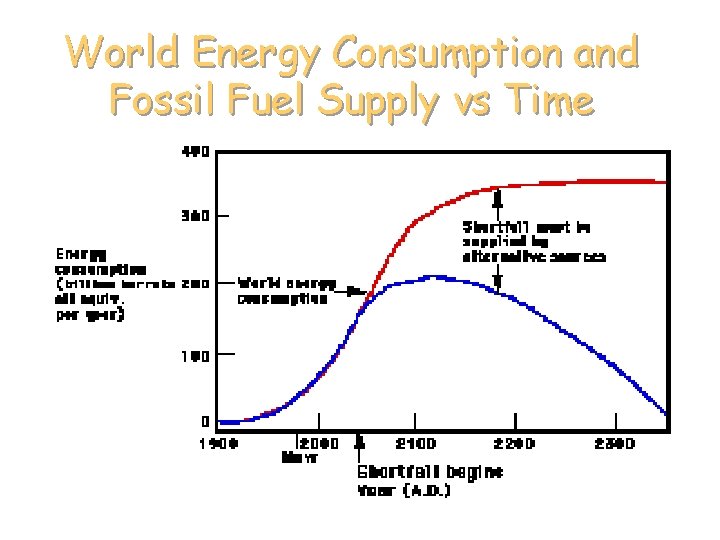
World Energy Consumption and Fossil Fuel Supply vs Time

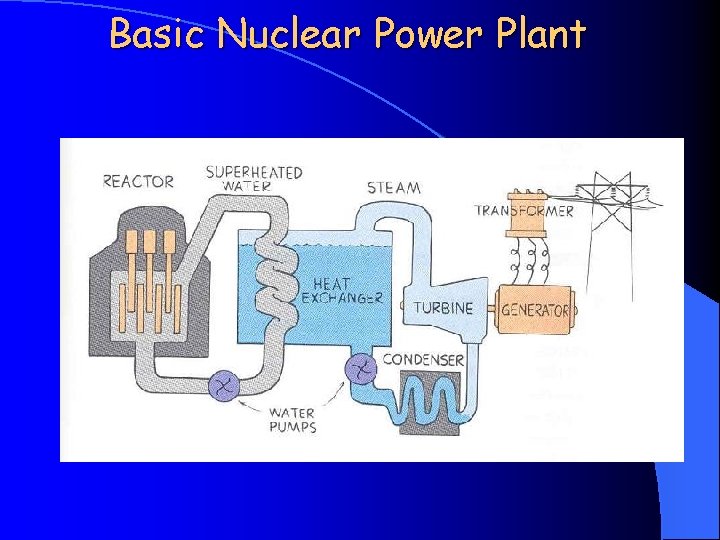
Basic Nuclear Power Plant

Fission Power Advantages l Supplies plentiful electricity l Conserves coal, oil, and natural gas l Reduces pollution from burning fossil fuel – sulfur oxides and carbon oxides

Fission Power Disadvantages l Storage of radioactive wastes l Production of plutonium l Danger of nuclear weapon proliferation l Low-level release of radioactive materials into air and groundwater l Risk of accidental release of large amounts of radioactivity

Controlling Nuclear Fusion l Fusion occurs at very high temperatures (on the order of 108 degrees) & densities l Problem? – All materials melt and vaporize at the temperature required for fusion l Solution – magnetic bottle or laser confinement

Fusion Benefits l No danger of runaway reactions l No air pollution – only helium is produced l By-products of fusion are not radioactive l Fuel is hydrogen (deuterium and tritium) – 30 kilograms of seawater contain 1 gram of deuterium

Nuclear Links l http: //www. ccnr. org/nuclear_primer. html l http: //www. energy. ca. gov/education/sto ry/story-html/chapter 07. html l http: //www. howstuffworks. com/nuclearpower 1. htm l http: //wwwformal. stanford. edu/jmc/progress/nucle ar-faq. html l http: //sol. sci. uop. edu/~jfalward/nuclear reactions/nuclearreactions. html

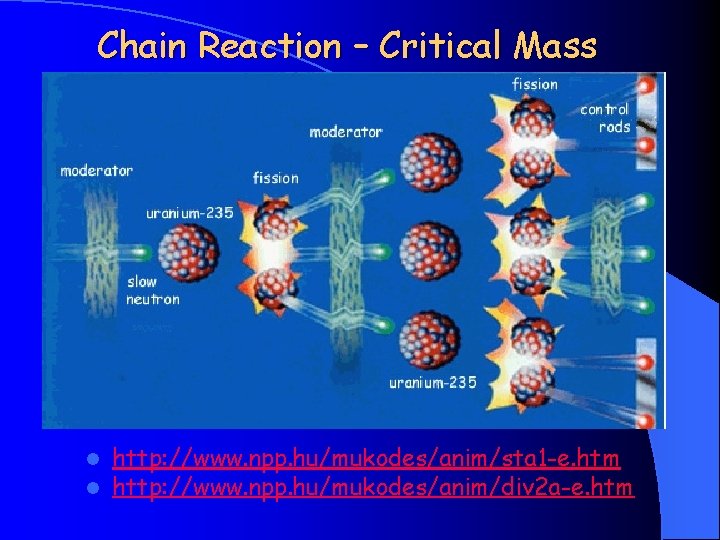
Chain Reaction – Critical Mass l l http: //www. npp. hu/mukodes/anim/sta 1 -e. htm http: //www. npp. hu/mukodes/anim/div 2 a-e. htm

Critical Mass l critical mass a self sustaining reaction starts l for pure metal U 235 (without moderator) the critical mass is somewhat less than 50 kg l Because of the very high density (19. 2 g/cm 3) this amount of uranium makes a sphere whose diameter is about 17 cm.

Moderator l should have as low atomic mass number as possible l its neutron absorbing ability should be as low as possible l water (H 2 O), heavy-water (D 2 O), graphite (C) and beryllium (Be) l

Cherenkov Radiation l Bluish glow when charged particles travel faster than the speed of light in the medium

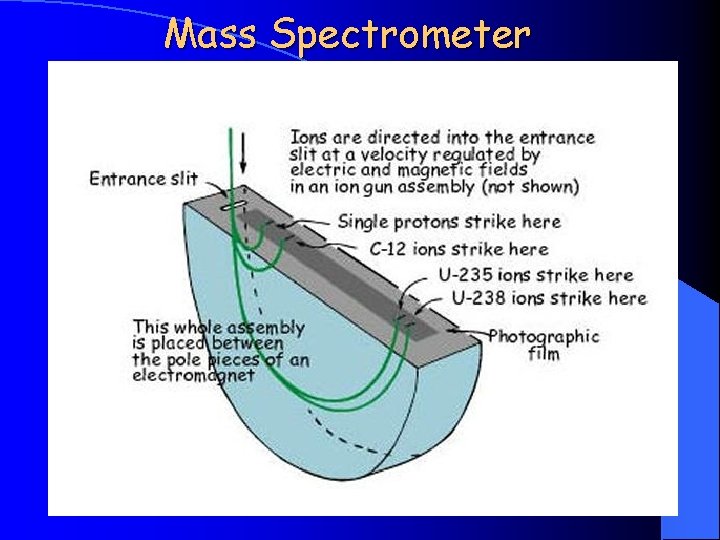
Mass Spectrometer

