Nuclear Decay Graphic www labinitio com Nuclear Symbols

Nuclear Decay Graphic: www. lab-initio. com
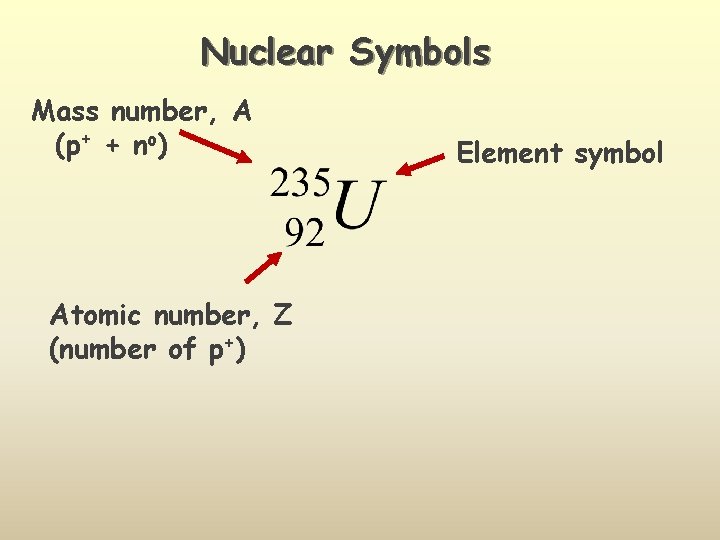
Nuclear Symbols Mass number, A (p+ + no) Atomic number, Z (number of p+) Element symbol
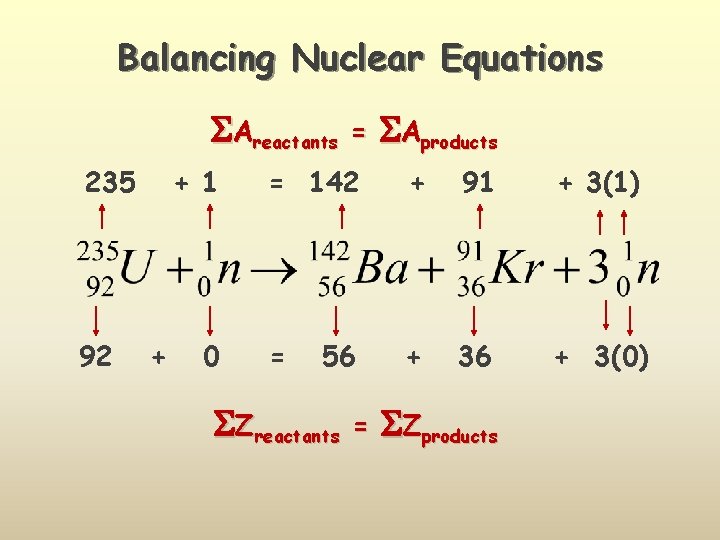
Balancing Nuclear Equations Areactants 235 92 + 1 + 0 = Aproducts = 142 + 91 + 3(1) = + 36 + 3(0) 56 Zreactants = Zproducts
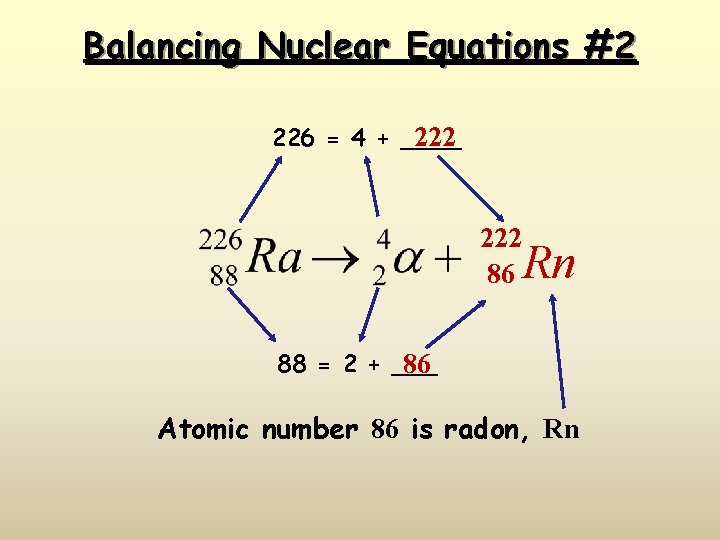
Balancing Nuclear Equations #2 226 = 4 + ____ 222 86 Rn 88 = 2 + ___ 86 Atomic number 86 is radon, Rn
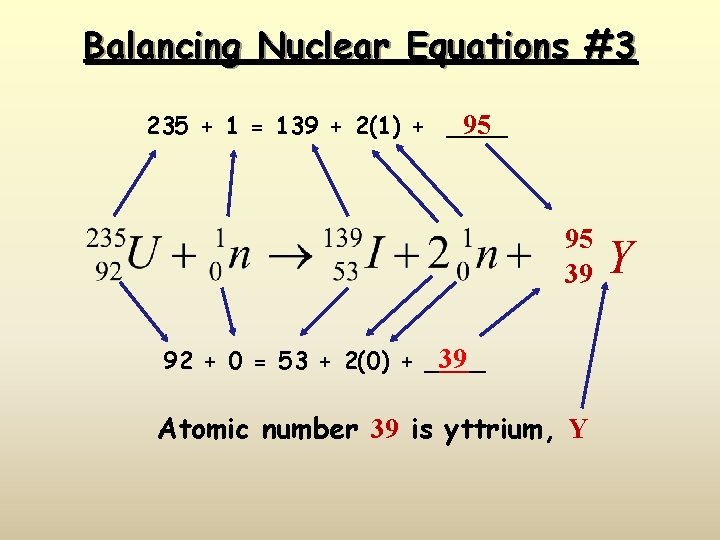
Balancing Nuclear Equations #3 95 235 + 1 = 139 + 2(1) + ____ 95 39 39 92 + 0 = 53 + 2(0) + ____ Atomic number 39 is yttrium, Y Y

Alpha Decay Alpha production (a): an alpha particle is a helium nucleus Alpha decay is limited to heavy, radioactive nuclei
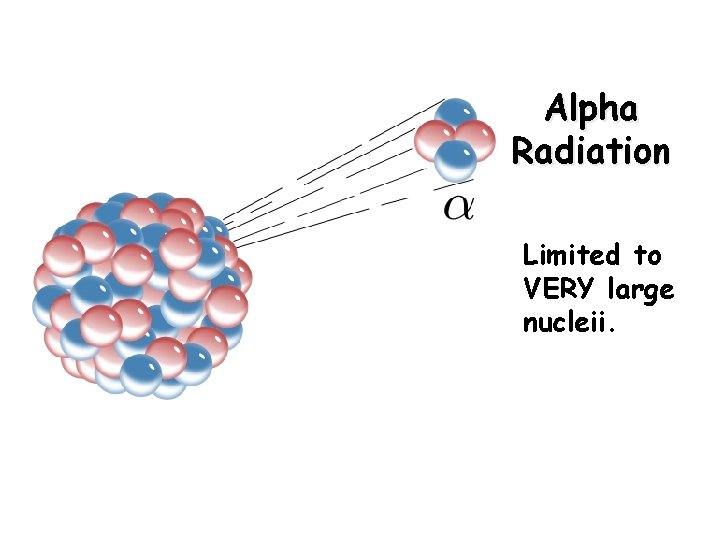
Alpha Radiation Limited to VERY large nucleii.

Beta Decay Beta production (b): A beta particle is an electron ejected from the nucleus Beta emission converts a neutron to a proton
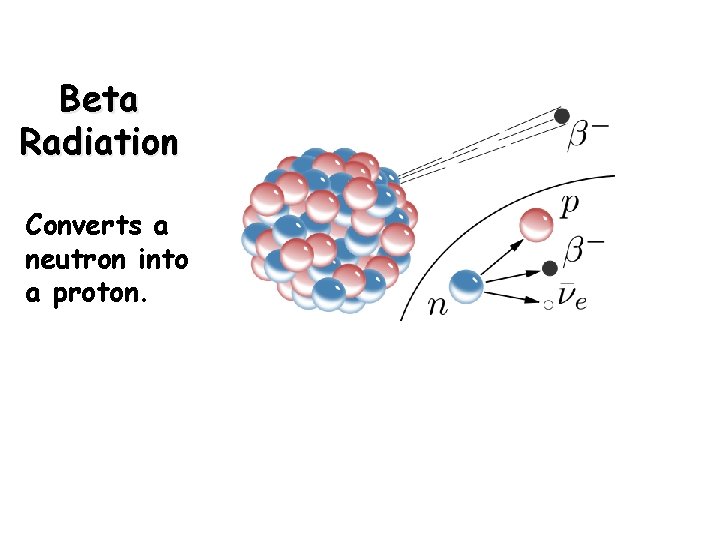
Beta Radiation Converts a neutron into a proton.

Gamma Ray Production Gamma ray production (g): Gamma rays are high energy photons produced in association with other forms of decay. Gamma rays are massless and do not, by themselves, change the nucleus
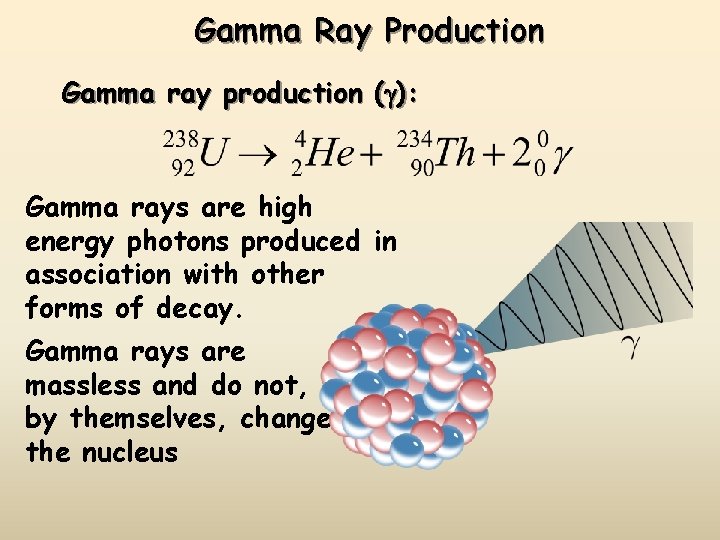
Gamma Ray Production Gamma ray production (g): Gamma rays are high energy photons produced in association with other forms of decay. Gamma rays are massless and do not, by themselves, change the nucleus

Positron Production Positron emission: Positrons are the antiparticle of the electron Positron emission converts a proton to a neutron

Electron Capture Electron capture: (inner-orbital electron is captured by the nucleus) Electron capture converts a proton to a neutron
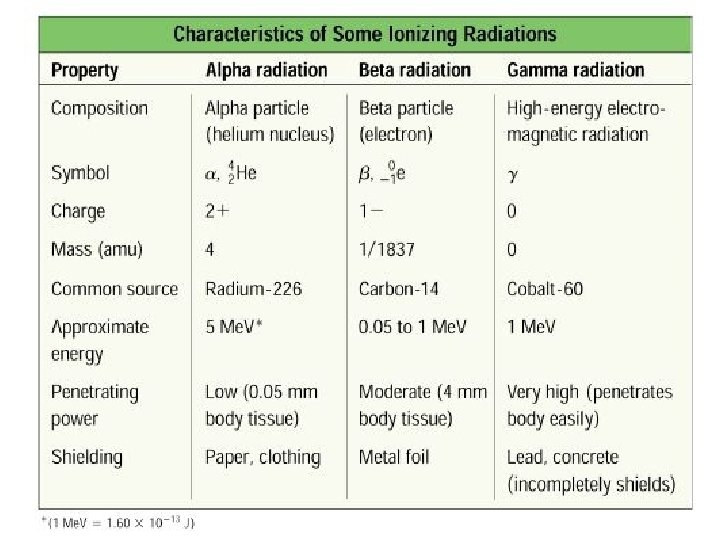
Types of Radiation
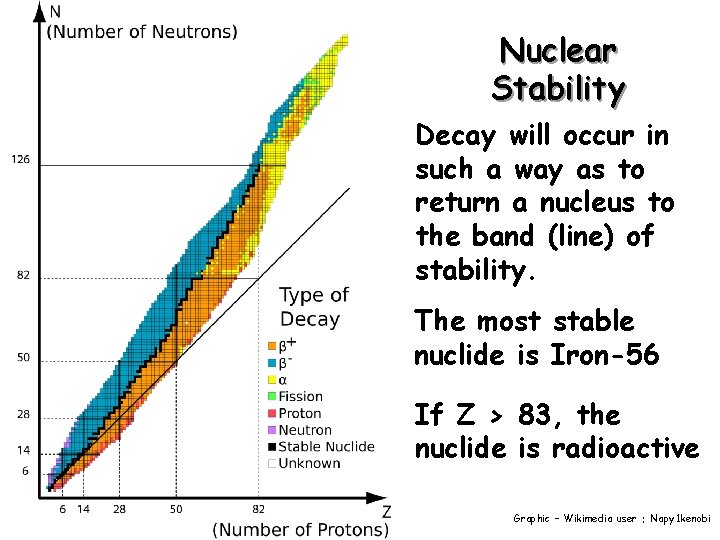
Nuclear Stability Decay will occur in such a way as to return a nucleus to the band (line) of stability. The most stable nuclide is Iron-56 If Z > 83, the nuclide is radioactive Graphic – Wikimedia user : Napy 1 kenobi
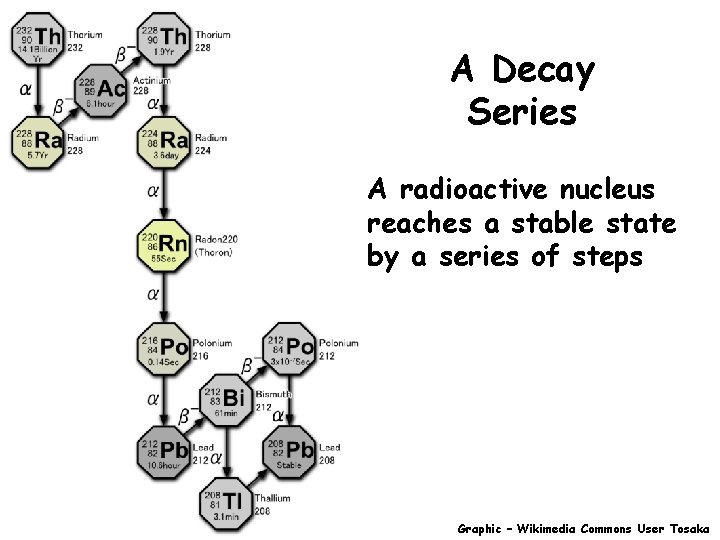
A Decay Series A radioactive nucleus reaches a stable state by a series of steps Graphic – Wikimedia Commons User Tosaka
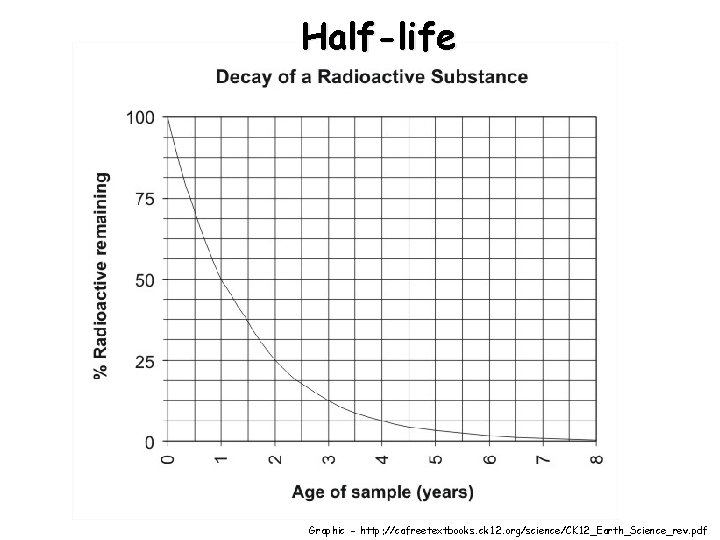
Half-life Graphic - http: //cafreetextbooks. ck 12. org/science/CK 12_Earth_Science_rev. pdf

Decay Kinetics Decay occurs by first order kinetics (the rate of decay is proportional to the number of nuclides present) N 0 = number of nuclides present initially N = number of nuclides remaining at time t k = rate constant t = elapsed time

Calculating Half-life t 1/2 = Half-life (units dependent on rate constant, k)

Sample Half-Lives
- Slides: 20