Nuclear Chemistry Valentim M B Nunes Engineering Unit

Nuclear Chemistry Valentim M. B. Nunes Engineering Unit – Chemistry Section May 2018

Introduction: nuclear reactions Nuclear Chemistry studies the reactions that involves transformations in the atomic nuclei. With the exception of all the nuclei contain two types of fundamental particles, the protons and the neutrons. Some of the nucleus are unstable, and they emit spontaneously particles and/or electromagnetic radiation. This phenomena is called radioactivity. Nuclei can also suffer transformations when they are bombarded by neutrons, electrons or other nucleus. This process is called nuclear transmutation.

Nuclear reactions Chemical reactions Nuclear reactions Break or formation of chemical Elements (or isotopes of the bonds same element) are converted in each other Only electrons from atomic orbitals are involved It can be involved protons, neutrons, electrons and other elementary particles Absorption or release of low quantities of energy Absorption or release of enormous quantities of energy Velocity of reaction depends on p and T. Velocities does not depend on p and T.
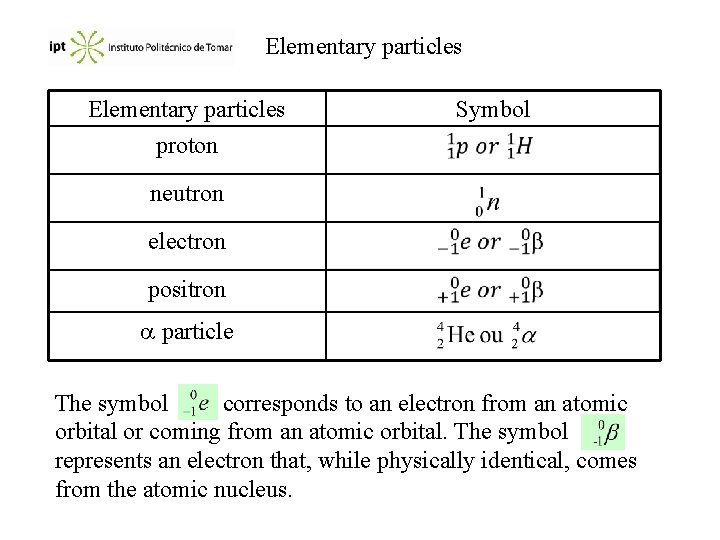
Elementary particles proton Symbol neutron electron positron particle The symbol corresponds to an electron from an atomic orbital or coming from an atomic orbital. The symbol represents an electron that, while physically identical, comes from the atomic nucleus.

Balance of nuclear reactions When balancing nuclear reactions we must obey to the following rules: >> The total number of protons + neutrons in products and reagents must be the same (conservation of the mass number) >> The total number of nuclear charges must be the same (conservation of the atomic number)

Examples

Nuclear stability As we know, the nucleus occupy a very small part of the size of the atom, but contains almost all the mass (neutrons + protons). Considering a typical nucleus with r = 5 10 -3 pm and m = 1 10 -22 g, we obtain: 2 1011 kg of H 2 O are approximately 2 1011 L or 2 108 m 3. An Olympic swimming pool (50 x 16 x 2) takes about 1600 m 3 of water. So the density of a nucleus its equivalent to put all the water of 125 000 Olympic swimming pools in a volume of 1 cm 3 !!!

Nuclear forces What are the forces that maintain the particles so strongly linked? It exists repulsion (Coulombic forces) p-p, but also attractive short range forces p-p, p-n e n-n (beyond the scope of this course). The main factor that determines the nucleus stability is the n/p ratio. In stable elements with low atomic number the ratio is n/p 1. As the atomic number increases the ratio is n/p > 1.
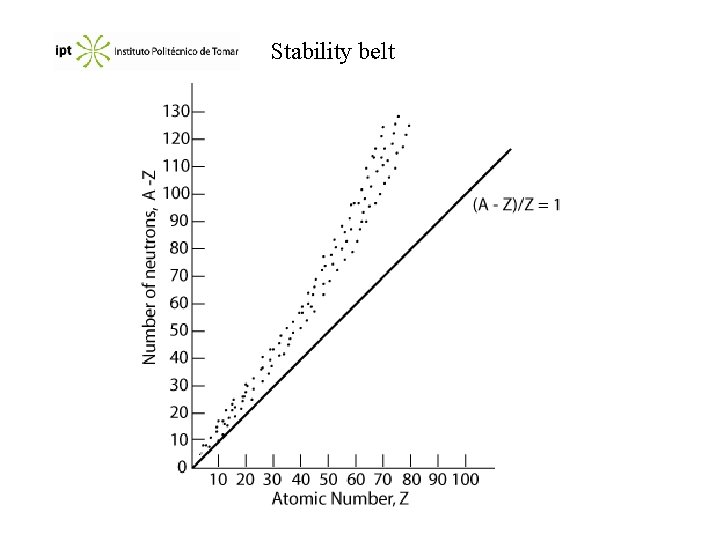
Stability belt

Consequences Isotopes above the stability belt have tendency to emit a beta particle: Below the stability belt they either emit a positron or capture an electron.

Nuclear cohesion energy A quantitative measure of the nuclear stability is the nuclear cohesion energy, that is, the energy necessary to decompose the nucleus in its components, the protons and neutrons. The mass of a nucleus is always less than the sum of the mass of protons + neutrons. Example: This difference is the mass defect.

Theory of relativity - Einstein The difference appears as energy released to the exterior, E = m c 2 1. 4 1010 k. J (!!!) The cohesion energy is 2. 37 10 -11 J

Natural radioactivity The nuclei apart from the stability belt, and also all the nucleus with more than 83 protons, tends to be unstable. The radioactivity is the spontaneous emission of particles and/or electromagnetic radiation by instable nucleus. The principal types of radiation are particles , rays (electromagnetic waves of short , from 0. 1 to 10 -4 nm, highly energetic), positron emission or electronic capture. The sequence of disintegration steps of an original radioactive nucleus is a decay series.
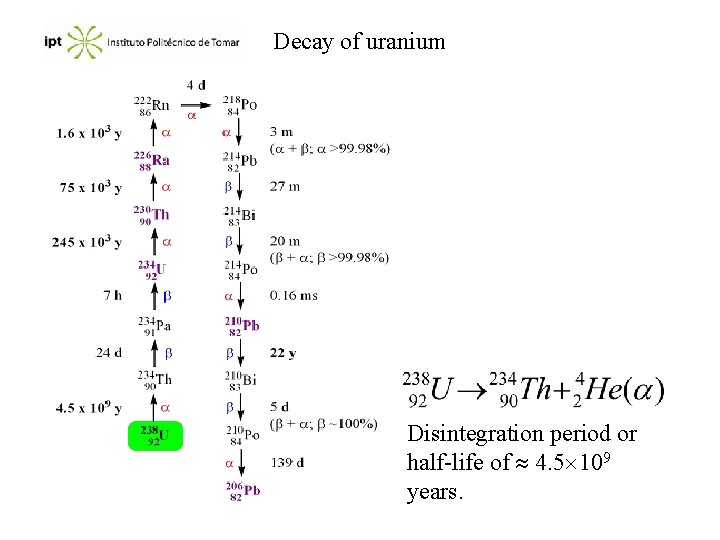
Decay of uranium Disintegration period or half-life of 4. 5 109 years.
At the home for old atoms!

Kinetics of radioactive decay All radioactive decays obey to the equation: k is a velocity constant and N is the number of radioactive nuclei at instant t.

Semi - disintegration period The semi - disintegration period (or half life period) is the period of time t = t 1/2 in witch N = N 0/2. Then, ln 2 = k t 1/2 or:
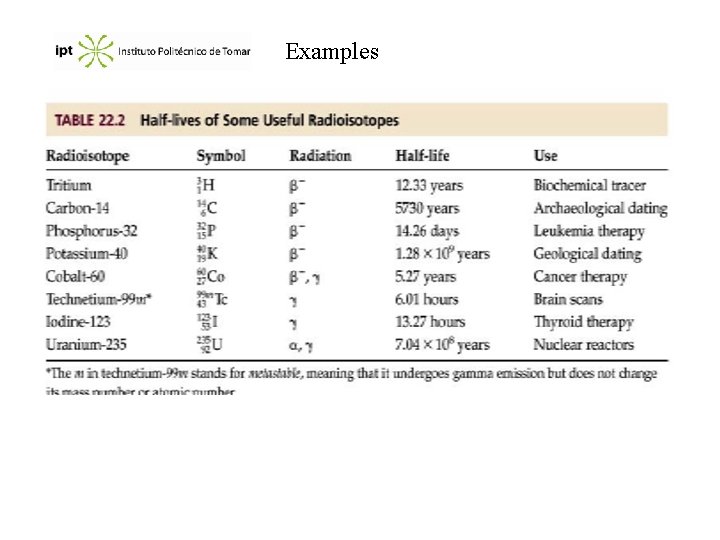
Examples

Decay of 14 C The radioactive decay has been used to determine the age of certain objects. Cosmic radiation gives origin to t 1/2 = 5. 73 103 years (Cambridge time) k = 0. 693/5. 73 103 1. 21 10 -4 y-1 The 14 C/12 C ration remains constant in a living organism. When he dies the 14 C loss by radioactivity is no longer substituted.

Dating based on decay Willard F. Libby (Nobel Prize of Chemistry in 1960) suggested that the 14 C decay could be used to date the age of archeologic findings. t 1/2 5568 anos (Chicago time) k = 0. 693/5568 1. 245 10 -4 y-1 A 0 is the initial activity of sample, A is the activity after a period of time t. The year 1950 is considered the 0 BP (zero before present). This method is able to date objects until approximately 60000 years ago.

Example A sample of a wood Thracian car, found in excavations in Bulgaria, shown a carbon 14 activity of 11. 2 dpm/g (decompositions per minute and per gram). Considering that the activity of a sample of living material is 14 dpm/g, calculate the age of the car and the year in which it was built (t 1/2 = 5730 years) The car was built around 106 D. C.

Science enough? the Holy Shroud of Turin The evangelist John recounts that Nicodemus and Joseph of Arimathea took Jesus body and covered it with perfumes, in linen, like the way the Jews used to bury (John 19, 38 -40). Matthew says that Joseph of Arimathea took Jesus body and involved it in a clean sheet by placing it in a new Tomb (Mt -60 27. 29). Mark and Luke refer to the fact that, "wrapped him in a sheet" (Mc 15. 46; LC 23. 53). The holy shroud of Turin, for almost 2000 years, undergone several vicissitudes. Of Jerusalem, fleeing persecution, the early Christians taken it to the city of Edessa, today southern Turkey. After several centuries went to Constantinople across the inside of the Turkey until 1204, being so taken by the Crusaders until Paris. Currently is in the Cathedral of Turin.

Nuclear transmutation The work of Rutherford in 1919 permitted to observe, for the first time, artificial radioactivity. When bombarding a sample of nitrogen with particle he obtained the following reaction: This reaction shown, for the first time, that it is possible to convert an element into another, in a process called nuclear transmutation. Another example:

Synthetic isotopes Many synthetic isotopes are prepared by similar processes, using particles accelerators where it is possible to increase the velocity (or kinetic energy) of charged particles (like the particles) allowing nuclear reactions.

Transuranium elements All the isotopes of the transuranium elements (atomic number higher than 92) are radioactive.

Nuclear fission In fission or nuclear fission a nucleus of an heavy element (mass number > 200) divides to form smaller nucleus , with intermediate mass, and one or more neutrons, releasing enormous quantities of energy.
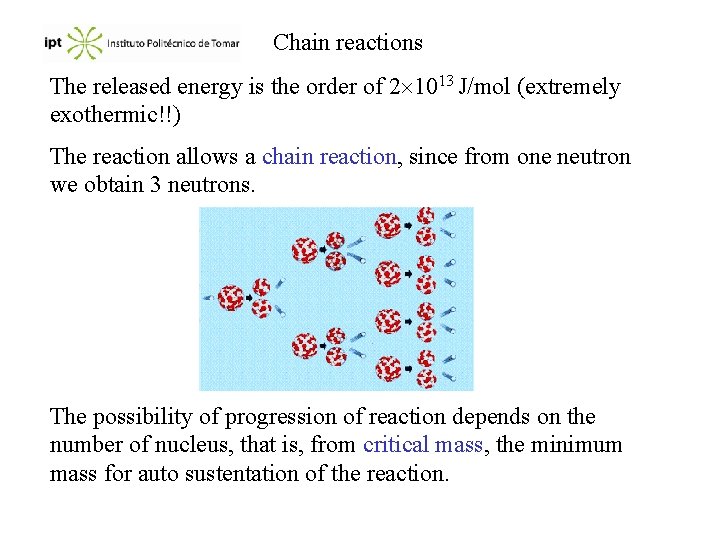
Chain reactions The released energy is the order of 2 1013 J/mol (extremely exothermic!!) The reaction allows a chain reaction, since from one neutron we obtain 3 neutrons. The possibility of progression of reaction depends on the number of nucleus, that is, from critical mass, the minimum mass for auto sustentation of the reaction.

The atomic bomb The first application of nuclear fission was the atomic bomb. The Hiroshima was released in the 6 August 1945. Three days later it was released the second in Nagasaki!!! A warm wind began to blow. Here and there in the distance I saw many small fires, like elf-fires, smoldering. Nagasaki had been completely destroyed – Yosuke Yamahata

Nuclear reactors The nuclear reactors produce electrical energy using heat generated from a controlled nuclear chain reaction. Most of the reactors use water as the working fluid. Slow neutrons are more effective in a division of nuclei, and therefore we must have moderator substances to reduce the kinetic energy. Water is adequate as it is a fluid (refrigeration liquid), has a high specific heat, and its non toxic and cheap. In Portugal it only exists one nuclear reactor at ITN (Sacavém) for scientific purposes.
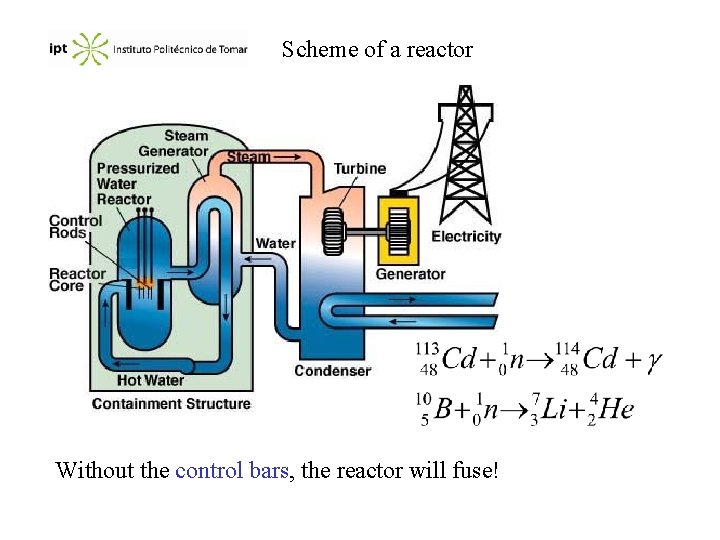
Scheme of a reactor Without the control bars, the reactor will fuse!

The Chernobyl accident In the early hours of 26 April 1986, one of four nuclear reactors at the Chernobyl power station exploded. The Chernobyl disaster triggered the release of substantial amounts of radiation into the atmosphere in the form of both particle and gaseous radioisotopes, and is the most significant unintentional release of radiation into the environment to date.

Nuclear fusion Contrarily to fission, nuclear fusion consists in the combination of lighter elements to give origin to heavy elements. This reactions occurs natural in the Sun. Origins temperatures of the order of millions of ºC. These reactions are called thermonuclear reactions.

ITER (Caradache-France) The ITER (International Thermonuclear Experimental Reactor) is in construction in Caradache (France), it will cost 10 000 millions USD and it will start to produce electrical energy in a clean and safe way in ~ 2050. Plasma: mixture of ions and electrons in gaseous phase at millions of ºC

Units of measure for radiation In the USA the radioactivity degree is measured in curies (Ci). In SI the unit is the becquerel (Bq) 1 Ci = 3. 7 1010 dps (disintegrations per second) 1 Bq = 1 dps The intensity of radiation depends on the number of disintegrations and the type of radiation. A common unit for absorbed radiation is the rad (radiation absorbed dose), the quantity of radiation absorbed of 1 10 -5 J per gram of irradiated material.

Biological effects The biological effect of radiation depends on what part of the body is irradiated and the type of radiation. So the rad is multiplied by a factor RBE (relative biological effectiveness) 1 rem = 1 rad 1 RBE The result is the roentgen equivalent for men.

- Slides: 36