Nuclear Chemistry Topic 1 Natural Radioactivity Objective Identify
Nuclear Chemistry Topic 1: Natural Radioactivity Objective: Identify the 4 modes of decay, write nuclear equations, identify the similarities and differences between physical, chemical, and nuclear reactions, identify what transmutation is

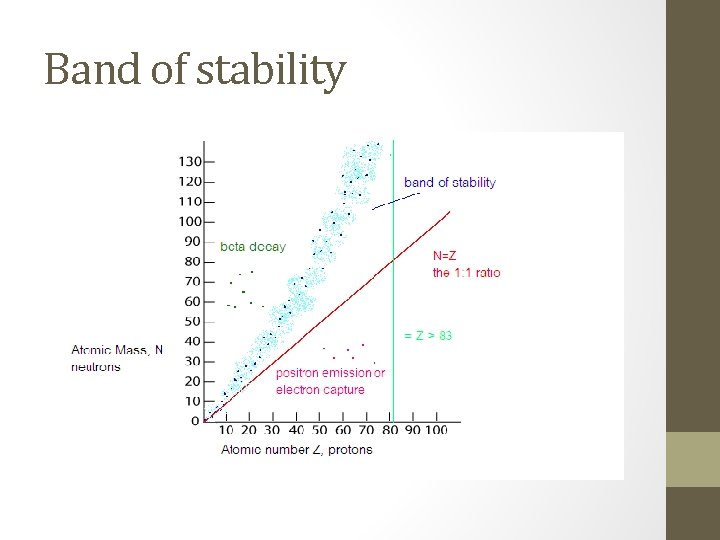
I. Natural Radioactivity and Stability • Nuclear Stability: the larger (more massive) a nucleus is, the harder it is for it to stay together radioactive • When a nucleus is ___________, it gives off decay particles and changes ____________. This is known from one element to another as ______________ Natural decay or natural transmutation • Atoms with an atomic number of 1 through 83 have at least one stable isotope, but… All isotopes of elements above 84 are radioactive ____________________________

• What are two elements that are radioactive? • How do you know? • Which has higher nuclear stability? • Silver or Polonium • Which has higher nuclear stability • Uranium or Iron • Why?

Nuclear Stability • Electrostatic force: The force that causes opposite electrical charges to attract each other. (positive and negative, protons and electrons) • Strong nuclear force: The force between nucleons (protons and neutrons) • Neutrons increase the attraction from the strong force without increasing electrostatic repulsion.
Band of stability

II. Three Modes of Decay spontaneous disintegration • Natural Radioactivity: _________________ of the _________ of an atom, with the emission of nucleus _________________ Particles and/or energy
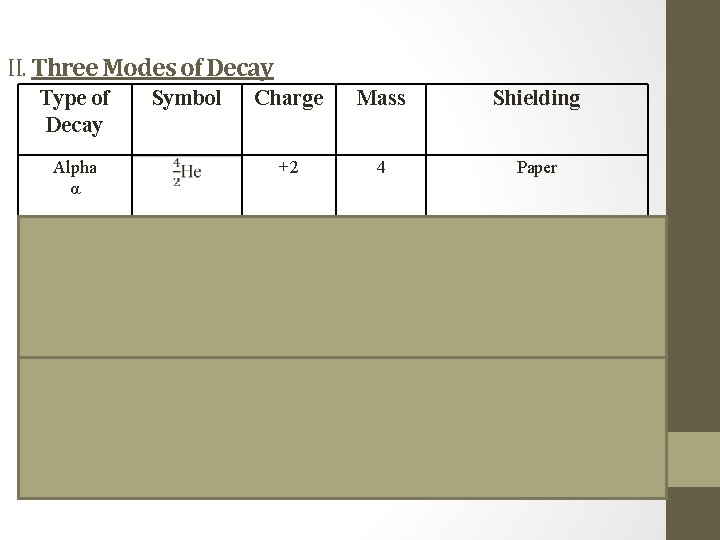
II. Three Modes of Decay Type of Decay Symbol Alpha α Charge Mass Shielding +2 4 Paper Essentially zero Aluminum Foil Beta β Gamma γ γ -1 0 0 0 (pure energy) Lead or Concrete
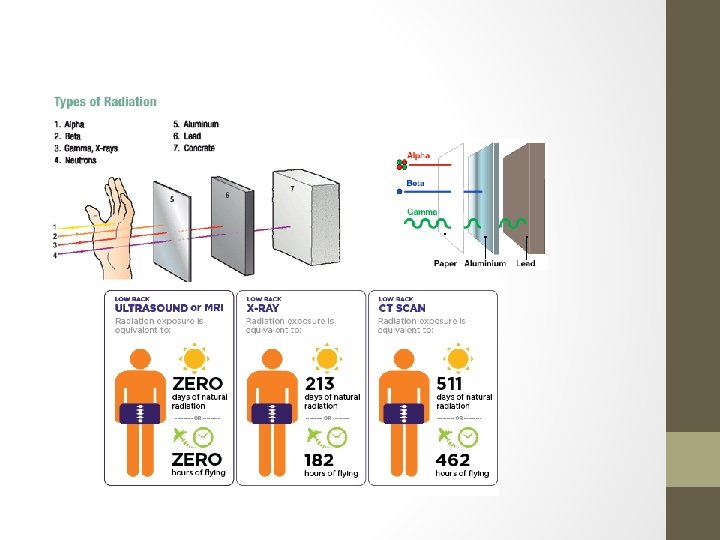

• Which type of radiation is most dangerous? • Which type of radioactive particle is largest?

Alpha Decay • Alpha decay – emission of an alpha particle ( ), 4 denoted by the symbol , because an α has 2 2 protons and 2 neutrons, just like the He nucleus. Charge is because of the 2 . • Alpha decay causes the number to decrease by and the number to decrease by . • determines the element. All nuclear equations are .

• Show the radioactive decay of Radium-226 through alpha decay. • • Show the radioactive decay of Polonium-210 through alpha decay

Beta decay β • Beta decay – emission of a beta particle ( ), a fast moving electron 0 , denoted by the symbol or . β has insignificant mass -1 ( ) and the charge is because it’s an . -1 0 e e electron • Beta decay causes change in number and causes the mass no. number to increase by . 1 atomic

• • • Show the radioactive decay of carbon-14 through beta decay. Show the radioactive decay of iodine-131 through beta decay.

Gamma decay • Gamma rays – high-energy electromagnetic radiation, denoted by the symbol . γ • γ has no mass ( ) and no charge ( ). Thus, it 0 0 no causes change in or mass atomic numbers. Gamma rays almost always accompany alpha and beta radiation. However, since there is effect on mass number or no atomic number, they are usually from omitted nuclear equations.
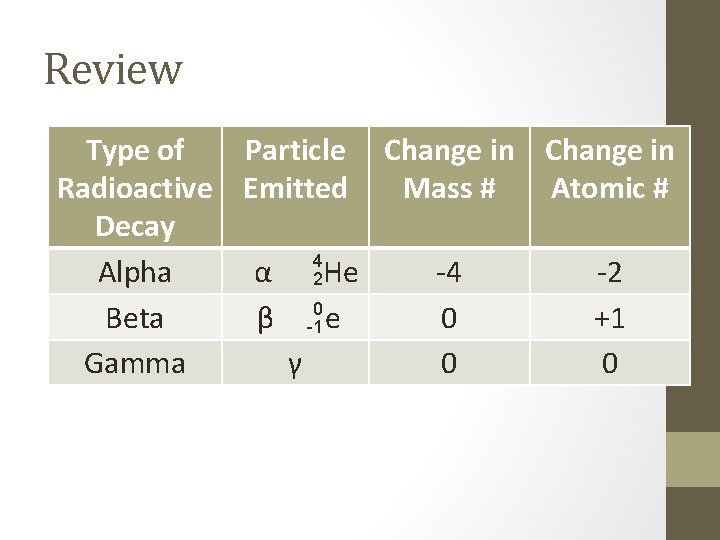
Review Type of Radioactive Decay Alpha Beta Gamma Particle Change in Emitted Mass # Atomic # 4 2 α He 0 β e -1 γ -4 0 0 -2 +1 0

III. Nuclear Equations • Show the natural radioactivity decay of: 1. Francium – 220 2. Gold – 198 3. Neon – 19
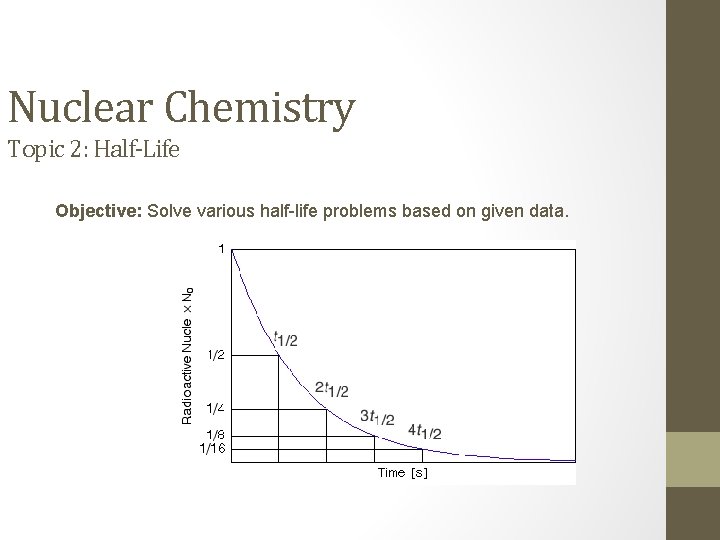
Nuclear Chemistry Topic 2: Half-Life Objective: Solve various half-life problems based on given data.

IV. Half-Life Problems • Half-Life: the period of time that must go by for half of the nuclei in the sample to undergo decay. • During a half-life period: half of the radioactive nuclei in a sample decays to a new, more stable nuclei _____________ • Solving half-life problems strategies: 1. Identify what nuclide (isotope) is the problem asking for? 2. What is the half-life of the nuclide? 3. How much time has passed? How many half lives does this represent? How many cuts in half were there? 4. Cut mass in half the required number of times

IV. Half-Life Problems • Examples: 1. What mass of I-131 remains 32 days after a 100 gram sample is obtained? (I-131 has a half life of 8 days) 2. What fraction of 1 gram sample of carbon – 14 would remain after 17190 years? (carbon-14 has a half life of 5730 years)

IV. Half-Life Problems • Examples: 3. Analysis of charred wood at a prehistoric campsite reveals that it contains 1/4 of the amount of carbon-14 that is found in living tissues. How old is the campsite? 4. The half-life of Rn-222 (a carcinogenic house pollutant) is 3. 8 days. If today your basement contains 20 grams of Rn-222, how much will remain after 19 days, assuming no more leaks in?

IV. Half-Life Problems • Examples: 5. The half-life of Tc-99 m* (used to locate brain tumors) is 6. 0 hours. If 10. Micrograms are left after 24 hours, how much Tc-99 m was administered?

V. Artificial Transmutations • Similarities and Differences between Nuclear Fission and Fusion Unique to Common to Both Nuclear Fission - SPLITS nucleus to -Both generate - Smaller particles energy the same way (Convert mass - Used to produce energy) energy, by humans. - Produces radioactive waste -Less Energy More energy Unique to Nuclear Fusion - COMBINES two small nuclei to form a larger one - Used by the sun - Produces essentially no radioactive waste

Nuclear Chemistry Topic 4: Uses and Dangers of Radioisotopes Objective: Understand the benefits and risks of nuclear reactions, identify specific uses of common isotopes
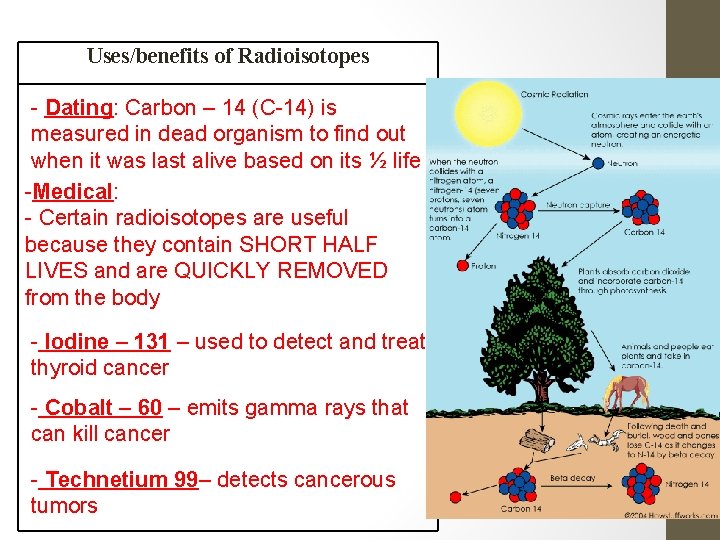
Uses/benefits of Radioisotopes - Dating: Carbon – 14 (C-14) is measured in dead organism to find out when it was last alive based on its ½ life -Medical: - Certain radioisotopes are useful because they contain SHORT HALF LIVES and are QUICKLY REMOVED from the body - Iodine – 131 – used to detect and treat thyroid cancer - Cobalt – 60 – emits gamma rays that can kill cancer - Technetium 99– detects cancerous tumors

Radiation Risks (WRITE SMALL) - Large amounts of radiation given off by isotopes can cause serious illness and death and environmental damage. -Nuclear Power Plant: - decay products have LONG half-lives and are difficult to store and dispose of - accidents can release harmful radioactive waste into air and water Chernobyl, Ukraine (1986)

• HOW MUCH RADIATION IS DANGEROUS? • It depends who you ask. Some people will say no amount is safe, but that doesn’t really help us understand the relative dangers. Here are some basic numbers to use as a guide (μSv means micro. Sieverts): • 10 μSv – The average radiation you received today • 40 μSv – The radiation you receive by taking a flight from New York to L. A. • 100 μSv – The radiation you receive during a dental x-ray • 800 μSv – Total radiation dose at Three-Mile Island for the duration of the accident • 3, 000 μSv – Radiation dose from a mammogram • 3, 600 μSv – Average radiation a US citizen receives in a year from all sources • 50, 000 μSv – Maximum allowable yearly occupational dose (USA) • 100, 000 μSv – Lowest yearly dose likely linked to increased cancer risk • 2, 000 μSv – Severe radiation poisoning (sometimes fatal) • http: //www. informationisbeautiful. net/visualizations/radiation-dosage-chart/

Your front porch may incorporate cinder blocks or granite. blocks that both contain uranium. A smoke detector owes its effectiveness to the constant source of alpha particle emissions from Americium-241. As long as the gases remain ionized within the shielded container, electricity flows, and all is calm. When smoke enters the chamber, it neutralizes the charges on these ions. In the absence of these ions, the circuit breaks and the alarm goes off. Indicator lights on your appliances may use Krypton-85; Electric blankets can have promethium-147 Fluorescent lights, thorium-229. Even the food we eat is radioactive. The more potassium-rich the food source, the more potassium -40—a radioactive isotope that makes up about 0. 01% of the natural supply of this mineral—is present. Thus, brazil nuts, peanuts, bananas, potatoes, and flour, all rich in potassium, are radiation sources.
- Slides: 27